Working Group 2 - Chapter 4: Ecosystems, their Properties, Goods and Services - (AR4-WG2-4)
Original at: http://www.ipcc.ch/publications_and_data/ar4/wg2/en/ch4.html
Main AR4 Index | Working Group WG2 Index | Table of Contents | Authors | Executive Summary | Annotated Text | References | Reviewer Comments
With the exception of Chapter and Section headings, all coloured text has been inserted by AccessIPCC. The non-coloured text is the IPCC original.
A number of emails from the Climate Research Unit (CRU) of the University of East Anglia were published on the Internet in November 2009. This has provided a window into the world of climate science.
We have identified a number of key individuals involved in the emails whom we have designated as Persons of Concern [PoC]; a Journal in which a PoC has published has been designated as a Journal of Concern [JoC].
This is not to suggest that we believe such papers are necessarily flawed, but rather that, as Joseph Alcamo noted at Bali in October 2009, "as policymakers and the public begin to grasp the multi-billion dollar price tag for mitigating and adapting to climate change, we should expect a sharper questioning of the science behind climate policy".
References occur in a list at the end of each chapter. Citations are within the normal text of sections and paragraphs.
| Tag | Explanation | Where Used | References | Citations |
|---|---|---|---|---|
| PoC |
Person of Concern Key individual involved in CRU emails as defined in this spreadsheet. |
References, Citations, IPCC Roles | 16 | 36 |
| JoC |
Journal of Concern A Journal which has published articles by one or more PoCs (Person of Concern) |
References, Citations | 273 | 469 |
| MoS |
Model or Simulation Reference appears to be a model or simulation, not observation or experiment |
References, Citations | 130 | 208 |
| NPR |
Non Peer Reviewed Reference has no Journal or no Volume or no Pages or it has Editors. |
References, Citations | 174 | 348 |
| SRC |
Self Reference Concern Author of a chapter containing references to own work. |
References, Citations, IPCC Roles | 148 | 311 |
| ARC |
Paper authored or co-authored by person who is also in list of Authors of another chapter. |
References, Citations | 113 | 218 |
| 2007 |
Paper dated 2007, when IPCC policy stated cutoff was December 2005 |
References, Citations | 18 | 53 |
| Ambiguous |
The short inline citation matched with more than one reference; however, AccessIPCC will link to the first reference found. |
Citations | - | 22 |
| NotFound |
The short inline citation was not matched with any reference. Believed to be caused by typing errors. |
Citations | - | 3 |
| Clean |
The reference was probably peer reviewed. |
References, Citations | 303 | 352 |
Coordinating Lead Authors:
Andreas Fischlin (Switzerland) [SRC:9], Guy F. Midgley (South Africa) [SRC:23],
| Concern | Occurrence |
|---|---|
| SRC >= 5 | 2 |
| Potentially Biased Authors | 2 |
Lead Authors:
Jeff Price (USA) [SRC:6], Rik Leemans (The Netherlands) [SRC:11], Brij Gopal (India) [SRC:2], Carol Turley (UK) [SRC:3], Mark Rounsevell (Belgium) [SRC:7], Pauline Dube (Botswana), Juan Tarazona (Peru), Andrei Velichko (Russia) [SRC:5],
| Concern | Occurrence |
|---|---|
| SRC >= 5 | 4 |
| SRC 1-4 | 2 |
| Potentially Biased Authors | 6 |
| Impartial Authors | 2 |
Contributing Authors:
Julius Atlhopheng (Botswana), Martin Beniston (Switzerland) [SRC:9], William J. Bond (South Africa) [SRC:7], Keith Brander (Denmark) [SRC:1], Harald Bugmann (Switzerland) [SRC:10], Terry V. Callaghan (UK) [SRC:13], Jacqueline de Chazal (Belgium), Oagile Dikinya (Australia), Antoine Guisan (Switzerland) [SRC:5], Dimitrios Gyalistras (Switzerland) [SRC:4], Lesley Hughes (Australia) [SRC:6], Barney S. Kgope (South Africa) [SRC:1], Christian Körner (Switzerland) [SRC:15], Wolfgang Lucht (Germany) [SRC:10], Nick J. Lunn (Canada) [SRC:2], Ronald P. Neilson (USA) [SRC:12], Martin Pêcheux (France), Wilfried Thuiller (France) [SRC:12], Rachel Warren (UK) [SRC:1],
| Concern | Occurrence |
|---|---|
| SRC >= 5 | 10 |
| SRC 1-4 | 5 |
| Potentially Biased Authors | 15 |
| Impartial Authors | 4 |
Review Editors:
Wolfgang Cramer (Germany) [SRC:11], Sandra Myrna Diaz (Argentina),
| Concern | Occurrence |
|---|---|
| SRC >= 5 | 1 |
| Potentially Biased Authors | 1 |
| Impartial Authors | 1 |
This chapter should be cited as:
Fischlin, A., G.F. Midgley, J.T. Price, R. Leemans, B. Gopal, C. Turley, M.D.A. Rounsevell, O.P. Dube, J. Tarazona, A.A. Velichko, 2007: Ecosystems, their properties, goods, and services. Climate Change 2007: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, M.L. Parry, O.F. Canziani, J.P. Palutikof, P.J. van der Linden and C.E. Hanson, Eds., Cambridge University Press, Cambridge, 211-272.
4.1 Introduction
An ecosystem can be practically defined as a dynamic complex of plant, animal and micro-organism communities, and the non-living environment, interacting as a functional unit (Millennium Ecosystem Assessment Reid et al., 2005 [NPR, SRC] ). Ecosystems may be usefully identified through having strong interactions between components within their boundaries and weak interactions across boundaries ( Reid et al., 2005 [NPR, SRC] , part 2). Ecosystems are well recognised as critical in supporting human well-being ( Reid et al., 2005 [NPR, SRC] ), and the importance of their preservation under anthropogenic climate change is explicitly highlighted in Article 2 (The Objective) of the United Nations Framework Convention on Climate Change (UNFCCC).
In this chapter the focus is on the properties, goods and services of non-intensively managed and unmanaged ecosystems and their components (as grouped by widely accepted functional and structural classifications, Figure 4.1 ), and their potential vulnerability to climate change as based on scenarios mainly from IPCC (see Chapter 2 and IPCC, 2007 [NPR, 2007] ). Certain ecosystem goods and services are treated in detail in other sectoral chapters (this volume): chapters 3 (water), 5 (food, fibre, fisheries), 6 (coasts) and 8 (health). Key findings from this chapter are further developed in the synthesis chapters 17 to 20 (this volume). Region-specific aspects of ecosystems are discussed in chapters 9 to 16 (this volume). This chapter is based on work published since the Third Assessment Report of the IPCC (TAR) ( Gitay et al., 2001 [NPR, ARC] ). We do not summarise TAR findings here, but refer back to relevant TAR results, where appropriate, to indicate confirmation or revision of major findings.
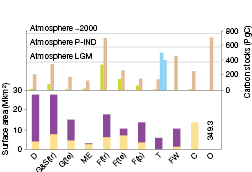
Figure 4.1. Major ecosystems addressed in this report, with their global areal extent (lower panel, Mkm2), transformed by land use in yellow, untransformed in purple, from Hassan et al. 2005 [NPR] ), except for mediterranean-climate ecosystems, where transformation impact is from Myers et al. 2000 [JoC] ), and total carbon stores (upper panel, PgC) in plant biomass (green), soil (brown), yedoma/permafrost (light blue). D = deserts, G&S(tr) = tropical grasslands and savannas, G(te) = temperate grasslands, ME = mediterranean ecosystems, F(tr) = tropical forests, F(te) = temperate forests, F(b) = boreal forests, T = tundra, FW = freshwater lakes and wetlands, C = croplands, O = oceans. Data are from Sabine et al. 2004 [NPR, ARC] , Table 2.2 , p. 23), except for carbon content of yedoma permafrost and permafrost (light blue columns, left and right, respectively, Zimov et al., 2006 [JoC] ), ocean organic carbon content (dissolved plus particulate organic; Denman et al., 2007 [NPR, 2007] , Section 7.3.4.1), and ocean surface area from Hassan et al. 2005 [NPR] , Summary, Table C2, p. 15, inserted as a number). Figures here update the TAR ( Prentice et al., 2001 [NPR] ), especially through considering soil C to 3 m depth ( (Jobbagy and Jackson, 2000 ) ), as opposed to 1 m. Approximate carbon content of the atmosphere (PgC) is indicated by the dotted lines for last glacial maximum (LGM), pre-industrial (P-IND) and current (about 2000 ).
Projecting the impacts of climate change on ecosystems is complicated by an uneven understanding of the interlinked temporal and spatial scales of ecosystem responses. Processes at large spatial scales, i.e., the biosphere at the global scale, are generally characterised by slow response times on the order of centuries, and even up to millennia ( Jansen et al., 2007 [NPR, PoC, 2007] ). However, it is also important to note that some large-scale responses in the palaeorecord ( Jansen et al., 2007 [NPR, PoC, 2007] ) and to current climate anomalies such as El Niño events may emerge at much shorter time-scales ( Holmgren et al., 2001 [Ambiguous] ;( Sarmiento and Gruber, 2002; ) Stenseth et al., 2002 [JoC] ; van der Werf et al., 2004 [JoC] ). At continental scales, biomes (see Glossary) respond at decadal to millennial time-scales (e.g., Davis, 1989 [JoC] ;( Prentice et al., 1991; ) Lischke et al., 2002 [MoS, SRC] ; Neilson et al., 2005 [MoS, SRC] ), and groups of organisms forming ecological communities at the regional scale have shorter response times of years to centuries. Responses of populations (i.e., interbreeding individuals of the same species) occur at intermediate temporal scales of months to centuries, and underpin changes in biodiversity. These include changes at the genetic level that may be adaptive, as demonstrated for example for trees ( Jump et al., 2006 [NPR] ) and corals ( Coles and Brown, 2003 [ARC] ). Fast physiological response times (i.e., seconds, hours, days, months) of micro-organisms, plants and animals operate at small scales from a leaf or organ to the cellular level; they underlie organism responses to environmental conditions, and are assessed here if they scale up to have a significant impact at broader spatial scales, or where the mechanistic understanding assists in assessing key thresholds in higher level responses.
The spatial distribution of ecosystems at biome scale has traditionally been explained only in terms of climate control ( Schimper, 1903 [NPR] ), but it is increasingly apparent that disturbance regimes such as fire or insects may strongly influence vegetation structure somewhat independently of climate (e.g., Andrew and Hughes, 2005 [SRC] ; Bond et al., 2005 [Ambiguous] ). Biomes are differentially sensitive to climatic change (e.g., Kirschbaum and Fischlin, 1996 [NPR, SRC] ; Sala et al., 2000 [JoC, SRC] ; Gitay et al., 2001 [NPR, ARC] ), with temperature-limited biomes prone to impacts of warming, and water-limited biomes prone to increasing levels of drought. Some, such as fire-dependent biomes, may be in a meta-stable state that can change rapidly under climate and other environmental changes ( Scheffer et al., 2001 [JoC] ;( Sankaran et al., 2005 ) ). Marine biome responses, too, have been shown at decadal scales ( Beaugrand et al., 2002 [JoC, ARC] ), with more rapid regime shifts within decades ( Edwards et al., 2002 [ARC] ; Richardson and Schoeman, 2004 [JoC] ; Edwards et al., 2006 [ARC] ). Biomes therefore provide a useful level of ecological organisation at which to summarise climate change impacts, being of large enough extent to conduct a global synthesis, yet having a response time relevant to anthropogenic climate change.
4.1.1 Ecosystem goods and services
Ecosystems provide many goods and services that are of vital importance for the functioning of the biosphere, and provide the basis for the delivery of tangible benefits to human society. Hassan et al. 2005 [NPR] ) define these to include supporting, provisioning, regulating and cultural services. In this chapter we divide services into four categories.
i. Supporting services, such as primary and secondary production, and biodiversity, a resource that is increasingly recognised to sustain many of the goods and services that humans enjoy from ecosystems. These provide a basis for three higher-level categories of services.
ii. Provisioning services, such as products (cf. Gitay et al., 2001 [NPR, ARC] ), i.e., food (including game, roots, seeds, nuts and other fruit, spices, fodder), fibre (including wood, textiles) and medicinal and cosmetic products (including aromatic plants, pigments; see Chapter 5 ).
iii. Regulating services, which are of paramount importance for human society such as (a) carbon sequestration, (b) climate and water regulation, (c) protection from natural hazards such as floods, avalanches or rock-fall, (d) water and air purification, and (e) disease and pest regulation.
iv. Cultural services, which satisfy human spiritual and aesthetic appreciation of ecosystems and their components.
4.1.2 Key issues
Based on new findings for ecosystems since the TAR, we highlight here five overarching key issues pertinent to assessing the vulnerability of ecosystems to anthropogenic climate change, and related adaptation responses.
Firstly, ecosystems are expected to tolerate some level of future climate change and, in some form or another, will continue to persist (e.g., Kirschbaum and Fischlin, 1996 [NPR, SRC] ; Gitay et al., 2001 [NPR, ARC] ), as they have done repeatedly with palaeoclimatic changes ( Jansen et al., 2007 [NPR, PoC, 2007] ). A primary key issue, however, is whether ecosystem resilience (understood as the disturbance an ecosystem can tolerate before it shifts into a different state, e.g., Scheffer et al., 2001 [JoC] ;( Cropp and Gabrica, 2002; ) ( Folke et al., 2004 ) ) inferred from these responses (e.g., Harrison and Prentice, 2003 [JoC, MoS] ) will be sufficient to tolerate future anthropogenic climate change (e.g., Chapin et al., 2004 [SRC] ;( Jump and Peñuelas, 2005 ) ). The implications of possibly transient increases in productivity for resilience are also very relevant. These may occur in certain terrestrial ecosystems through likely atmospheric CO2-fertilisation effects and/or modest warming (e.g., ( Baker et al., 2004; ) ( Lewis et al., 2004b; ) ( Malhi and Phillips, 2004 ) ), and demonstrated consequences of increased radiation due to reduced tropical cloudiness ( Nemani et al., 2003 [JoC, ARC] ). Ecosystem resilience thus seems usefully equivalent to the critical ecosystem property highlighted in Article 2 of the UNFCCC, i.e., an “ability to adapt naturally”.
Secondly, ecosystems are increasingly being subjected to other human-induced pressures, such as extractive use of goods, and increasing fragmentation and degradation of natural habitats (e.g., Bush et al., 2004 [JoC] ). In the medium term (i.e., decades) especially, climate change will increasingly exacerbate these human-induced pressures, causing a progressive decline in biodiversity ( Lovejoy and Hannah, 2005 [NPR] ). However, this is likely to be a complex relationship that may also include some region-specific reductions in land-use pressures on ecosystems (e.g., ( Goklany, 2005; ) Rounsevell et al., 2006 [MoS, SRC] ).
A third key issue involves exceeding critical thresholds and triggering non-linear responses in the biosphere that could lead via positive feedback to novel states that are poorly understood. Projected future climate change and other human-induced pressures are virtually certain to be unprecedented ( Forster et al., 2007 [NPR, ARC, 2007] ) compared with the past several hundred millennia (e.g., Petit et al., 1999 [JoC] ; Augustin et al., 2004 [JoC] ; Siegenthaler et al., 2005 [JoC] ).
Fourthly, the understanding of time-lags in ecosystem responses is still developing, including, for example, broad-scale biospheric responses or shifting species geographical ranges. Many ecosystems may take several centuries (vegetation) or even possibly millennia (where soil formation is involved) before responses to a changed climate are played out (e.g., Lischke et al., 2002 [MoS, SRC] ). A better understanding of transient responses and the functioning of ecosystems under continuously changing conditions is needed to narrow uncertainties about critical effects and to develop effective adaptation responses at the time-scale of interest to human society.
A fifth key issue relates to species extinctions, and especially global extinction as distinct from local extinctions, since the former represents irreversible change. This is crucial, especially because of a very likely link between biodiversity and ecosystem functioning in the maintenance of ecosystem services ( Duraiappah et al., 2005 [NPR] ;( Hooper et al., 2005; ) ( Diaz et al., 2006; ) Worm et al., 2006 [JoC] ), and thus extinctions critical for ecosystem functioning, be they global or local, are virtually certain to reduce societal options for adaptation responses.
4.2 Current sensitivities
4.2.1 Climatic variability and extremes
The biosphere has been exposed to large variability and extremes of CO2 and climate throughout geological history ( Augustin et al., 2004 [JoC] ; Siegenthaler et al., 2005 [JoC] ; Jansen et al., 2007 [NPR, PoC, 2007] ), and this provides some insight into the current sensitivities of ecosystems even though it is not possible to match past climate analogues precisely with future warming, due to differences in forcing factors ( Overpeck et al., 2006 [PoC, JoC, MoS] ), dominant ecosystems, and species (e.g., Velichko et al., 2002 [Ambiguous] ). What can be learned is that, firstly, significant biological changes including species extinctions have accompanied large climate perturbations of the past (e.g., Overpeck et al., 2005 [NPR, PoC] ). Secondly, endemic biodiversity is concentrated in regions that have experienced lower variability during the Pleistocene (from about 2 million years ago) ( (Jansson, 2003 ) ), during which glacial and inter-glacial conditions have alternated for roughly the past 2 million years. Thirdly, range shifts have been a major species response ( Lovejoy and Hannah, 2005 [NPR] ), although genetic and physiological responses ( Davis and Shaw, 2001 [JoC] ) have also occurred, which can be broadly defined as ‘natural adaptation’ at species level, and by aggregation, at the ecosystem level.
While earlier IPCC reports described several ecosystems to be resilient to warming up to 1°C (e.g., Kirschbaum and Fischlin, 1996 [NPR, SRC] ), recent studies provide a more differentiated view of ecosystem sensitivity (e.g., Walther et al., 2002 [JoC, ARC] ) that includes understanding of the role of climatic variability and extremes. Knowledge about climate variability and natural ecosystems has improved with better understanding of the behaviour of decadal-scale climatic oscillations and their impacts, including ENSO (El Niño/Southern Oscillation) and the NAO (North Atlantic Oscillation) ( Trenberth et al., 2007 [NPR, PoC, 2007] , Section 3.6 ). These low-frequency phenomena indirectly determine vegetation responses, notably through shifts in major controls (temperature, precipitation, snow cover). For example, the European Alps show changes in regional climates that can partly be attributed to NAO variability ( Hurrell and van Loon, 1997 [JoC] ; Serreze et al., 1997 [ARC] ;( Wanner et al., 1997; ) Beniston and Jungo, 2002 [SRC] ) such as the lack of snow in the late 1980 s and early 1990 s ( Beniston, 2003 [JoC, SRC] ). Disruptions of precipitation regimes in the Pacific region and beyond during ENSO events can disrupt vegetation through drought, heat stress, spread of parasites and disease, and more frequent fire (e.g., Diaz and Markgraf, 1992 [NPR] ). Similar effects have been reported for NAO ( Edwards and Richardson, 2004 [JoC, ARC] ;( Sims et al., 2004; ) Balzter et al., 2005 [JoC, ARC] ). Sea surface temperature increases associated with ENSO events have been implicated in reproductive failure in seabirds ( Wingfield et al., 1999 [NPR] ), reduced survival and reduced size in iguanas ( Wikelski and Thom, 2000 [JoC] ) and major shifts in island food webs ( Stapp et al., 1999 [JoC] ).
Many significant impacts of climate change may emerge through shifts in the intensity and the frequency of extreme weather events. Extreme events can cause mass mortality of individuals and contribute significantly to determining which species occur in ecosystems ( Parmesan et al., 2000 [JoC, ARC] ). Drought plays an important role in forest dynamics, driving pulses of tree mortality in the Argentinean Andes ( (Villalba and Veblen, 1997 ) ), North American woodlands ( (Breshears and Allen, 2002; ) Breshears et al., 2005 [JoC] ), and in the eastern Mediterranean ( Körner et al., 2005b [SRC] ). In both the Canadian Rockies ( Luckman, 1994 [NPR] ) and European Alps ( Bugmann and Pfister, 2000 [MoS, SRC] ) extreme cold through a period of cold summers from 1696 to 1701 caused extensive tree mortality. Heatwaves such as the recent 2003 event in Europe ( Beniston, 2004 [JoC, MoS, SRC] ; Schär et al., 2004 [JoC] ; Box 4.1 ) have both short-term and long-term implications for vegetation, particularly if accompanied by drought conditions.
Box 4.1. Ecological impacts of the European heatwave 2003
Anomalous hot and dry conditions affected Europe between June and mid-August, 2003 ( (Fink et al., 2004; ) Luterbacher et al., 2004 [JoC] ; Schär et al., 2004 [JoC] ). Since similarly warm summers may occur at least every second year by 2080 in a Special Report on Emissions Scenario (SRES; Naki´cenovi´c et al, 2000 [NotFound] ) A2 world, for example ( Beniston, 2004 [JoC, MoS, SRC] ; Schär et al., 2004 [JoC] ), effects on ecosystems observed in 2003 provide a conservative analogue of future impacts. The major effects of the 2003 heatwave on vegetation and ecosystems appear to have been through heat and drought stress, and wildfires.
Drought stress impacts on vegetation ( (Gobron et al., 2005; ) ( Lobo and Maisongrande, 2006 ) ) reduced gross primary production (GPP) in Europe by 30% and respiration to a lesser degree, overall resulting in a net carbon source of 0.5 PgC/yr ( Ciais et al., 2005 [JoC, ARC] ). However, vegetation responses to the heat varied along environmental gradients such as altitude, e.g., by prolonging the growing season at high elevations ( Jolly et al., 2005 [JoC] ). Some vegetation types, as monitored by remote sensing, were found to recover to a normal state by 2004 (e.g., ( Gobron et al., 2005 ) ), but enhanced crown damage of dominant forest trees in 2004, for example, indicates complex delayed impacts ( Fischer, 2005 [NPR] ). Freshwater ecosystems experienced prolonged depletion of oxygen in deeper layers of lakes during the heatwave ( (Jankowski et al., 2006 ) ), and there was a significant decline and subsequent poor recovery in species richness of molluscs in the River Saône ( Mouthon and Daufresne, 2006 [JoC] ). Taken together, this suggests quite variable resilience across ecosystems of different types, with very likely progressive impairment of ecosystem composition and function if such events increase in frequency (e.g., Lloret et al., 2004 [JoC] ;( Rebetez and Dobbertin, 2004; ) Jolly et al., 2005 [JoC] ; Fuhrer et al., 2006 [JoC, SRC] ).
High temperatures and greater dry spell durations increase vegetation flammability (e.g., ( Burgan et al., 1997 ) ), and during the 2003 heatwave a record-breaking incidence of spatially extensive wildfires was observed in European countries ( Barbosa et al., 2003 [NPR] ), with roughly 650,000 ha of forest burned across the continent ( De Bono et al., 2004 [NPR] ). Fire extent (area burned), although not fire incidence, was exceptional in Europe in 2003, as found for the extraordinary 2000 fire season in the USA ( Brown and Hall, 2001 [NPR] ), and noted as an increasing trend in the USA since the 1980 s ( Westerling et al., 2006 [JoC, ARC] ). In Portugal, area burned was more than twice the previous extreme ( 1998 ) and four times the 1980 - 2004 average ( (Trigo et al., 2005, ) 2006 ). Over 5% of the total forest area of Portugal burned, with an economic impact exceeding ¤1 billion ( De Bono et al., 2004 [NPR] ).
Long-term impacts of more frequent similar events are very likely to cause changes in biome type, particularly by promoting highly flammable, shrubby vegetation that burns more frequently than less flammable vegetation types such as forests ( (Nunes et al., 2005 ) ), and as seen in the tendency of burned woodlands to reburn at shorter intervals ( Vazquez and Moreno, 2001 [ARC] ; Salvador et al., 2005 ). The conversion of vegetation structure in this way on a large enough scale may even cause accelerated climate change through losses of carbon from biospheric stocks ( Cox et al., 2000 [JoC, MoS, ARC] ). Future projections for Europe suggest significant reductions in species richness even under mean climate change conditions ( Thuiller et al., 2005b [JoC, SRC] ), and an increased frequency of such extremes (as indicated e.g., by Schär et al., 2004 [JoC] ) is likely to exacerbate overall biodiversity losses ( Thuiller et al., 2005b [JoC, SRC] ).
Hurricanes can cause widespread mortality of wild organisms, and their aftermath may cause declines due to the loss of resources required for foraging and breeding ( (Wiley and Wunderle, 1994 ) ). The December 1999 ‘storm-of-the-century’ that affected western and central Europe destroyed trees at a rate of up to ten times the background rate (Anonymous, 2001 ). Loss of habitat due to hurricanes can also lead to greater conflict with humans. For example, fruit bats (Pteropus spp.) declined recently on American Samoa due to a combination of direct mortality events and increased hunting pressure ( Craig et al., 1994 [ARC] ). Greater storminess and higher return of extreme events will also alter disturbance regimes in coastal ecosystems, leading to changes in diversity and hence ecosystem functioning. Saltmarshes, mangroves and coral reefs are likely to be particularly vulnerable (e.g. ( Bertness and Ewanchuk, 2002; ) Hughes et al., 2003 [MoS, SRC] ).
Assessment of the impacts of climate variability, their trends, and the development of early warning systems has been strongly advanced since the TAR by satellite-based remote sensing efforts. Notable contributions have included insights into phenological shifts in response to warming (e.g., Badeck et al., 2004 [SRC] ) and other environmental trends (e.g., Nemani et al., 2003 [JoC, ARC] ), complex Sahelian vegetation changes (e.g., Prince et al., 1998 [JoC] ; Rasmussen et al., 2001 [JoC] ;( Anyamba and Tucker, 2005; ) Hein and Ridder, 2006 [JoC] ), wildfire impacts (e.g., Isaev et al., 2002 [JoC, ARC] ; Barbosa et al., 2003 [NPR] ; Hicke et al., 2003 [JoC] ; Kasischke et al., 2003 [JoC, MoS] ), coral bleaching events (e.g., Yamano and Tamura, 2004 [MoS] ), cryosphere changes ( Walsh, 1995 [JoC, ARC] ; Lemke et al., 2007 [NPR, ARC, 2007] ), ecotone (see Glossary) responses to climate (e.g., ( Masek, 2001 ) ), deforestation (e.g., Asner et al., 2005 [JoC] ), and even feedbacks to regional climate (e.g., ( Durieux et al., 2003 ) ), the impacts of extreme climate events (e.g., ( Gobron et al., 2005; ) ( Lobo and Maisongrande, 2006 ) ) and monitoring of soil water ( Wagner et al., 2003 [MoS, SRC] ).
4.2.2 Other ecosystem change drivers
Ecosystems are sensitive not only to changes in climate and atmospheric trace gas concentrations but also to other anthropogenic changes such as land use, nitrogen deposition, pollution and invasive species ( Vitousek et al., 1997 [JoC] ;( Mack et al., 2000; ) Sala et al., 2000 [JoC, SRC] ; Hansen et al., 2001 [SRC] ; Lelieveld et al., 2002 [JoC] ; Körner, 2003b [SRC] ;( Lambin et al., 2003; ) Reid et al., 2005 [NPR, SRC] ). In the recent past, these pressures have significantly increased due to human activity ( Gitay et al., 2001 [NPR, ARC] ). Natural disturbance regimes (e.g., wildfire and insect outbreaks) are also important climate-sensitive drivers of ecosystem change. Projecting the impacts of the synergistic effects of these drivers presents a major challenge, due to the potential for non-linear, rapid, threshold-type responses in ecological systems ( Burkett et al., 2005 [SRC] ).
Land-use change represents the anthropogenic replacement of one land use type by another, e.g., forest to cultivated land (or the reverse), as well as subtle changes of management practices within a given land use type, e.g., intensification of agricultural practices, both of which are affecting 40% of the terrestrial surface (reviewed by Foley et al., 2005 [JoC, ARC] ). Land-use change and related habitat loss and fragmentation have long been recognised as important drivers of past and present ecosystem change, particularly of biodiversity ( Heywood and Watson, 1995 [NPR] ;( Fahrig, 2003 ) ).
Fire influences community structure by favouring species that tolerate fire or even enhance fire spread, resulting in a relationship between the relative flammability of a species and its relative abundance in a particular community ( Bond and Keeley, 2005 [SRC] ). As a result, many vegetation types are far from the maximum biomass predicted by regional climate alone ( Bond et al., 2005 [Ambiguous] ). Geographical shifts in key species or fire may therefore cause fundamental community shifts ( (Brooks et al., 2004; ) Schumacher and Bugmann, 2006 [JoC, MoS, SRC] ). Fire-prone vegetation types cover a total of 40% of the world’s land surface ( Chapin et al., 2002 [NPR] ), and are common in tropical and sub-tropical regions ( Bond et al., 2005 [Ambiguous] ), and the boreal region ( Harden et al., 2000 [JoC] ) in particular. Intensified wildfire regimes driven at least partly by 20th century climate change ( Gillett et al., 2004 [JoC] ; Westerling et al., 2006 [JoC, ARC] ), appear to be changing vegetation structure and composition with shifts from Picea- to Pinus-dominated communities and 75-95% reductions in tree densities observed in forest-tundra transition in eastern Canada ( (Lavoie and Sirois, 1998 ) ). By contrast, in Quebec, fire frequency appears to have dropped during the 20th century ( (Bergeron et al., 2001 ) ), a trend projected to continue (see Section 4.4.5 Bergeron et al., 2004 [MoS] ). Across the entire North American boreal region, however, total burned area from fires increased by a factor of 2.5 between the 1960 s and 1990 s, while the area burned from human-ignited fires remained constant ( Kasischke and Turetsky, 2006 [JoC] ). In South-East Asia, by contrast, human activities have significantly altered fire regimes in ways that may be detrimental to the affected ecosystems ( Murdiyarso and Lebel, 2007 [NPR, ARC, 2007] ).
Drought facilitated the spread of human-caused fire in tropical regions during the 1997 /98 El Niño ( (Randerson et al., 2005 ) ), affecting atmospheric trace gas concentrations such as CO, CH4 and H2( (Langenfelds et al., 2002; ) ( Novelli et al., 2003; ) ( Kasischke et al., 2005 ) ), and CO2 emissions ( van der Werf et al., 2004 [JoC] ) at hemispheric and global scales. Drought conditions increase Amazon forest flammability ( Nepstad et al., 2004 [JoC] ). Tropical forest fires are becoming more common ( Cochrane, 2003 [JoC] ), and have strong negative effects on Amazonian vegetation ( (Cochrane and Laurance, 2002; ) ( Haugaasen et al., 2003 ) ), possibly even intensifying rainfall events ( Andreae et al., 2004 [JoC] , but see Sections 4.4.1 and 4.4.5 on forest productivity trends).
Significant progress on globally applicable models of fire has been made since the TAR ( Thonicke et al., 2001 [MoS, SRC] ). Modelling suggests increases in wildfire impacts (see Sections 4.4.1 and 4.4.5 ) during the 21st century under a wide range of scenarios (e.g., Scholze et al., 2006 [JoC, ARC] ). The implications of the regional and global importance of fire are manifold ( Bond et al., 2005 [Ambiguous] ). Firstly, fire suppression strategies often have limited impact ( (Keeley, 2002; ) ( Schoennagel et al., 2004; ) ( van Wilgen et al., 2004 ) ), and the enhancement of vegetation flammability through more prevalent fire weather ( Brown et al., 2004 [JoC] ) and the resulting big wildfires threatens human settlements, infrastructure and livelihoods (e.g., Allen Consulting Group, 2005 ). Secondly, in some ecosystems, including islands, human-caused fires have transformed forests into more flammable shrublands and grasslands ( (Ogden et al., 1998 ) ). Thirdly, the drivers of flammability, such as ecosystem productivity, fuel accumulation and environmental fire risk conditions, are all influenced by climate change ( Williams et al., 2001 [JoC, ARC] ; see Sections 4.4.3 , 4.4.4 and 4.4.5 ).
The spatial impact of insect damage is significant and exceeds that of fire in some ecosystems, but especially in boreal forests ( (Logan et al., 2003 ) ). Spruce bud worm (SBW), for example, defoliated over 20 times the area burned in eastern Ontario between 1941 and 1996 ( Fleming et al., 2002 [JoC] ). Furthermore, fires tended to occur 3 to 9 years after a SBW outbreak ( Fleming et al., 2002 [JoC] ), suggesting a greater interaction between these disturbances with further warming. Disturbance by forest tent caterpillar has also increased in western Canada in the past 25 years ( (Timoney, 2003 ) ). In the Mediterranean region, the defoliation of Scots Pine shows a significant association with previous warm winters, implying that future climatic warming may intensify insect damage ( (Hodar and Zamora, 2004; ) see Section 4.4.5 ).
Invasive alien species (IAS) ( Chornesky and Randall, 2003 [MoS] ) represent a major threat to endemic or native biodiversity in terrestrial and aquatic systems ( Sala et al., 2000 [JoC, SRC] ; Scavia et al., 2002 [ARC] ;( Occhipinti-Ambrogi and Savini, 2003 ) ). Causes of biological invasions are multiple and complex ( (Dukes and Mooney, 1999 ) ), yet some simple models have been developed ( Crawley, 1989 [NPR] ;( Deutschewitz et al., 2003; ) ( Chytry et al., 2005; ) ( Facon et al., 2006 ) ). Alien species invasions also interact with other drivers, sometimes resulting in some unexpected outcomes ( (Chapuis et al., 2004 ) ). Changes in biotic and/or abiotic disturbance regimes are recognised as primary drivers of IAS ( (Le Maitre et al., 2004 ) ), with communities often becoming more susceptible to invasion following extreme events ( (Smith and Knapp, 1999 ) ), such as are projected under future climate change. IAS can also change disturbance regimes through increasing vegetation flammability ( (Brooks et al., 2004 ) ). Overall, ongoing shifts in human-mediated disturbances, insect pests, IAS and fire regimes are very likely to be important in altering regional ecosystem structure, diversity and function (e.g., ( Timoney, 2003 ) ).
4.3 Assumptions about future trends
The work reviewed in this chapter is dependent on assumptions of various types that are important in assessing the level of confidence that can be associated with its results ( Moss and Schneider, 2000 [NPR, PoC, ARC] ), but can be challenging to quantify and aggregate. Assumptions and uncertainties associated with climate scenarios ( Randall et al., 2007 [NPR, 2007] ) are not considered here, other than to identify the greenhouse gas emission trends or socio-economic development pathways (e.g., SRES, Naki?enovi? et al., 2000 ) assumed in the literature we review (see also Table 4.1 , especially scaling methodology and associated uncertainties). Since the TAR, many global or regional scenarios have become available to quantify future impacts ( Christensen et al., 2002 [ARC] , 2007; Meehl et al., 2007 [NPR, ARC, 2007] ), and confidence in future climate projections has increased recently (Naki?enovi? et al., 2000; Randall et al., 2007 [NPR, 2007] ). However, many assumptions must be made, due to imperfect knowledge, in order to project ecosystem responses to climate scenarios. We provide here a brief outline and guide to the literature of those that are most relevant.
To project impacts of climate change on ecosystems there are basically three approaches: (i) correlative, (ii) mechanistic, and (iii) analogue approaches. For the correlative and mechanistic approaches, studies and insights from the present give rise to the assumption that the same relationships will hold in the future. Three modelling approaches in particular have provided relevant results since the TAR. Firstly, correlative models use knowledge of the spatial distribution of species to derive functions ( Guisan and Thuiller, 2005 [MoS, SRC] ) or algorithms ( Pearson et al., 2004 [MoS, ARC] ) that relate the probability of their occurrence to climatic and other factors ( Guisan and Zimmermann, 2000 [MoS, SRC] ). Criticised for assumptions of equilibrium between species and current climate, an inability to account for species interactions, lack of a physiological mechanism, and inability to account for population processes and migration (see Pearson and Dawson, 2003 [MoS] ;( Pearson, 2006 ) ), these methods have nonetheless proved capable of simulating known species range shifts in the distant ( Martinez-Meyer et al., 2004 [MoS] ) and recent ( Araújo et al., 2005 [MoS, ARC] ) past, and provide a pragmatic first-cut assessment of risk to species decline and extinction ( Thomas et al., 2004a [JoC, SRC] ). Secondly, mechanistic models include the modelling of terrestrial ecosystem structure and function. They are based on current understanding of energy, biomass, carbon, nutrient and water relations, and their interacting dynamics with and among species such as primary producers. Such approaches generate projections of future vegetation structure, e.g., as the likely balance of plant functional types (PFTs) after permitting competitive interaction and accounting for wildfire ( Woodward and Lomas, 2004b [MoS] ; Lucht et al., 2006 [SRC] ; Prentice et al., 2007 [NPR, SRC, 2007] ; but see Betts and Shugart, 2005 [NPR, ARC] , for a more complete discussion). Extrapolated to global scale, these are termed Dynamic Global Vegetation Models (DGVMs, see Glossary). An equivalent approach for oceans is lacking (but see Field et al., 1998 [JoC, ARC] ). Thirdly, Earth system models have begun to incorporate more realistic and dynamic vegetation components, which quantify positive and negative biotic feedbacks by coupling a dynamic biosphere to atmospheric circulations with a focus on the global carbon cycle ( (Friedlingstein et al., 2003, ) 2006; Cox et al., 2004 [MoS, ARC] , 2006 ).
Ecosystem- and species-based models are typically applied at scales much finer than are resolved or reliably represented in global climate models. The requisite downscaling techniques of various types (statistical, dynamic) have matured and are increasingly used to provide the necessary spatio-temporal detail ( IPCC-TGCIA, 1999 [NPR] ; Mearns et al., 2003 [NPR, MoS, ARC] ; Wilby et al., 2004 [NPR, ARC] ; Christensen et al., 2007 [NPR, ARC, 2007] ). Physically consistent bioclimatic scenarios can now be derived for almost any region, including developing countries (e.g., Jones et al., 2005 [NPR, SRC] ) and complex, mountainous terrain (e.g., Gyalistras and Fischlin, 1999 [MoS, SRC] ; Hayhoe et al., 2004 [PoC, JoC, SRC] ). However, major uncertainties relating to downscaling remain in the impact projections presented in this chapter, centring mainly on soil water balance and weather extremes which are key to many ecosystem impacts, yet suffer from low confidence in scenarios for precipitation and climate variability, despite recent improvements ( Randall et al., 2007 [NPR, 2007] ).
Despite the recognised importance of multiple drivers of ecosystem change, they are rarely all included in current climate and ecosystem models used for assessing climate change impacts on ecosystems ( Hansen et al., 2001 [SRC] ; Levy et al., 2004 [JoC, MoS] ;( Zebisch et al., 2004; ) Feddema et al., 2005 [JoC, MoS, ARC] ; Holman et al., 2005b [JoC, SRC] ; Pielke, 2005 [JoC] ). The explicit inclusion of non-climatic drivers and their associated interactions in analyses of future climate change impacts could lead to unexpected outcomes ( Hansen et al., 2001 [SRC] ; Sala, 2005 [NPR] ). Consequently, many impact studies of climate change that ignore land-use and other global change trends (see also Section 4.2.2 ) may represent inadequate estimates of projected ecosystem responses.
4.4 Key future impacts and vulnerabilities
The scope of this section satisfies that required by the IPCC plenary in relation to future impacts on properties, goods and services of major ecosystems and on biodiversity. However, to assess ecosystem goods and services more completely, issues relating to biogeochemical cycling and other supporting or regulating services are also deemed appropriate for consideration under this heading. Following reviews of impacts on individual ecosystems, impacts that cut across ecosystems (such as large-scale vegetation shifts and migratory species) are elaborated. Finally the overall implications for biodiversity are highlighted in a global synthesis. Within the relevant sub-sections, we describe briefly ecosystem properties, goods and services, we summarise key vulnerabilities as identified by the TAR, and then review what new information is available on impacts, focusing on supporting and regulating services (for provisioning services see Chapters 3 , 5 and 6 ).
4.4.1 Biogeochemical cycles and biotic feedback
The cycling of chemical elements and compounds sustains the function of the biosphere and links ecosystems and climate by regulating chemical concentrations in soil, biota, atmosphere and ocean. Substantial progress has been made since the TAR in understanding the interactive responses of terrestrial ecosystems and the climate system, as determined by plant physiological responses, interactions with the soil, and their scaled-up effects on regional and global biogeochemical cycles ( (Buchmann, 2002; ) Cox et al., 2006 [NPR] ; Friedlingstein et al., 2006 [PoC, JoC, MoS, ARC] ; Gedney et al., 2006 [JoC, ARC] ). Interactions between ocean and atmosphere and land and oceans are also critical for the future evolution of climate (see Section 4.4.9 , but mainly Denman et al., 2007 [NPR, 2007] , e.g., Section 7.3.5.4).
Among the most advanced tools to achieve scaling-up of terrestrial systems to the global scale are Dynamic Global Vegetation Models (DGVMs), which simulate time-dependent changes in vegetation distribution and properties, and allow mapping of changes in ecosystem function and services ( Schröter et al., 2005 [JoC, SRC] ; Metzger et al., 2006 [SRC] ). Testing at hierarchical levels from leaf to biome and over relevant time-scales has shown encouraging agreement with observations ( Lucht et al., 2002 [JoC, SRC] ; Bachelet et al., 2003 [MoS, SRC] ; Harrison and Prentice, 2003 [JoC, MoS] ; Gerten et al., 2004 [MoS, SRC] ; Joos and Prentice, 2004 [NPR, PoC] ; Kohler et al., 2005 [PoC, JoC, MoS] ;( Peylin et al., 2005 ) ), and validation is ongoing (e.g., Woodward and Lomas, 2004b [MoS] ; Prentice et al., 2007 [NPR, SRC, 2007] ). Recently, full coupling between DGVMs and climate models has progressed from earlier work (e.g., ( Woodward and Lomas, 2001 ) ) to explore feedback effects between biosphere and atmospheric processes ( Cox et al., 2006 [NPR] ; Friedlingstein et al., 2006 [PoC, JoC, MoS, ARC] ), that were initially reported as having significant implications for the carbon cycle ( Cox et al., 2000 [JoC, MoS, ARC] ).
Key vulnerabilities
Ecosystems are likely to respond to increasing external forcing in a non-linear manner. Most initial ecosystem responses appear to dampen change ( Aber et al., 2001 [MoS, SRC] ), but amplify it if thresholds in magnitude or rate of change are surpassed. Transitions between states may be triggered, or the ecosystem may even ‘collapse’ i.e., show a rapid transition to a much less productive and/or species-poor assemblage with lower biomass and other impairments such as degrading soils (e.g., Scheffer et al., 2001 [JoC] ; Rietkerk et al., 2004 [JoC] ;( Schröder et al., 2005 ) ). Changing fire regimes provide an important example (see Section 4.2.2 for a more complete treatment), as these are of significant concern for the terrestrial carbon balance ( Schimel and Baker, 2002 [JoC] ; van der Werf et al., 2004 [JoC] ; Westerling et al., 2006 [JoC, ARC] ), especially because they can be self-reinforcing ( Bond and Keeley, 2005 [SRC] ). However, even less extreme responses of ecosystems are likely to have important ramifications for the biosphere because of their spatial extent.
Based on early versions of DGVMs (equilibrium biogeography models or global biogeochemical models – Neilson et al., 1998 [Ambiguous] ), the world’s terrestrial ecosystems were projected to continue as a net carbon sink for a number of decades and possibly throughout the 21st century, with an initially ‘greening’ world due to longer growing seasons, more precipitation and CO2-fertilisation benefits. Substantial structural changes in biomes were projected towards 2100, with ecosystem shifts towards higher latitudes and altitudes. A reversal of initial carbon sequestration gains was projected during the 21st century, as CO2-fertilisation benefits approach saturation and temperature effects on respiration and transpiration increase, potentially resulting in net global ecosystem carbon losses relative to today (e.g., Cramer et al., 2001 [JoC, MoS, SRC] ). With feedback from the global carbon cycle to the atmosphere accounted for, dieback of much of the Amazon rainforest due to desiccation was an identified major vulnerability, but with a high degree of uncertainty ( Cox et al., 2000 [JoC, MoS, ARC] ). The TAR concluded that the net global terrestrial carbon exchange would be between -6.7 PgC/yr (uptake 1 PgC) and +0.4 PgC/yr, and that anthropogenic CO2 emissions would remain the dominant determinant of atmospheric CO2 concentration during the 21st century. Key ecosystem forecasting needs identified in the TAR were for spatially and temporally dynamic models to simulate processes that produce inertia and lags in ecosystem responses. Progress on this issue has now allowed initial assessments of the potential for feedbacks from ecosystems to atmospheric composition and climate change.
Impacts
Observations for global net primary productivity (NPP) from 1982 to 1999 show an increase of 6%, concentrated in the tropics and due virtually certainly to greater solar radiation with reduced cloud cover ( Nemani et al., 2003 [JoC, ARC] ), broadly concurring with the projection in the TAR of an increasing biospheric sink in the initial stages of climate change. Scaled-up effects of direct atmospheric CO2 enrichment on plant and ecosystem biomass accumulation (CO2-fertilisation) are largely responsible for the projected continued enhancement of NPP in current global models ( Leemans et al., 2002 [SRC] ). By contrast, impacts in oceans, especially through acidification, have been largely negative (see Section 4.4.9 ).
Despite improved experiments, the magnitude of the terrestrial CO2-fertilisation effect remains uncertain, although improved simulation of major vegetation types (particularly forests and savannas) at the last glacial maximum by incorporating CO2 effects ( Harrison and Prentice, 2003 [JoC, MoS] ) are encouraging. The three main constraints that have been observed to limit the fertilisation effect are element stoichiometry (nutrient balance), forest tree dynamics, and secondary effects of CO2 on water relations and biodiversity. Trends in some empirical data suggest caution when estimating future carbon sequestration potentials of the biosphere as a contribution to mitigating climate change, in particular as these benefits may be smaller than the counteracting impacts of land-use change. Persistent grassland responses to elevated CO2, which range from 0 to 40% biomass gain per season, mainly reflect CO2-induced water savings induced by scaled-up impacts of reduced stomatal conductance ( Morgan et al., 2004 [SRC] ; Gerten et al., 2005 [JoC, SRC] ), and thus rely on current moisture regimes and lack the realistic atmospheric feedback of the future that may negate this benefit. The only replicated test of multiple CO2 * climate/environment interactions (water, temperature, nutrient supply) yielded no overall CO2 biomass signal in a grassland system ( Shaw et al., 2002 [JoC, ARC] ), highlighting the significant influence of co-limiting environmental variables.
Similar trends are emerging for forests, although the interpretation is complicated by time-lags in biomass response to the artifactual step-change when initiating CO2 treatments, requiring longer observation periods before a new steady state (e.g., in terms of leaf area index, fine root dynamics and nutrient cycling) is reached. Three tall forest test systems, loblolly pine plantation ( Oren et al., 2001 [JoC] ; Schäfer et al., 2003 [JoC] ), sweet gum plantation ( (Norby et al., 2002; ) ( Norby and Luo, 2004 ) ), and mixed deciduous forest ( Körner et al., 2005a [JoC, SRC] ) exhibit significant initial biomass stimulation that diminishes with time except for one of the four pairs of test plots (treatment versus control) in the joint Duke pine experiments ( Schäfer et al., 2003 [JoC] ). A European boreal forest system also showed smaller CO2 growth stimulation in mature trees under field conditions than expected from results for saplings ( Rasmussen et al., 2002 [MoS] ). A recent analysis ( Norby et al., 2005 [JoC] ) suggests that the NPP response of trees to elevated CO2 is relatively predictable across a broad range of sites, with a stimulation of 23 ± 2% at a median CO2 of double the pre-industrial level. The logarithmic biotic growth factor derived from this is 0.60 (?-factor, expressing the response as a function of the relative CO2 increase). Nonetheless, it is uncertain whether test systems with mostly young growing trees provide valid analogies for biomass responses in mature forests with a steady state nutrient cycle and many other factors moderating the response to elevated CO2 concentrations (e.g., ( Karnosky, 2003 ) ).
It has been suggested that greatest CO2-fertilisation impacts may be seen in savanna systems post-fire ( Bond and Midgley, 2000 [JoC, SRC] ; Bond et al., 2003 [Ambiguous] ), especially where nutrients are less limiting and in systems in which trees require carbon reserves to re-establish after fire (see Section 4.2.2 ). Scrub oak in Florida shows diminishing CO2 responses as treatment proceeds ( (Hungate et al., 2006 ) ), even though this is a post-fire regenerating system. For tropical forests, the planet’s single largest biomass carbon reservoir, post-industrial atmospheric CO2 enrichment seems to have enhanced growth dynamics ( Phillips et al., 2002 [JoC] ; Laurance et al., 2004 [JoC] ;( Wright et al., 2004 ) ). A more dynamic forest might ultimately store less rather than more carbon in future if long-term species compositional changes are realised ( Laurance et al., 2004 [JoC] ; Malhi et al., 2006 [JoC] ), especially given the exceptional CO2 responsiveness of tropical lianas that may increase tree mortalities and population turnover ( Körner, 2004 [SRC] ).
Based on experimental data, best estimates of instantaneous CO2-induced water savings due to reduced stomatal aperture range from 5 to 15% ( (Wullschleger and Norby, 2001; ) Cech et al., 2003 [SRC] ) for humid conditions, diminishing with drying soils. Desert shrub systems increase production in elevated CO2 only during exceptional wet periods and not in dry periods ( (Nowak et al., 2004 ) ), contrasting with earlier expectations ( Morgan et al., 2004 [SRC] ). Evapotranspiration data for temperate zone ecosystems under future CO2 scenarios suggest that this may be reduced by less than 10% across all weather conditions. Water savings through elevated CO2 hold limited benefits for trees during drought, because nutrient availability in drying top soil becomes interrupted, and initial water savings are exhausted ( Leuzinger et al., 2005 [SRC] ). Repeated drought with high temperatures (e.g., Europe in 2003, Box 4.1 ) may reduce landscape-wide carbon stocks ( Ciais et al., 2005 [JoC, ARC] ). Studies using a land-surface model indicate at least for the past century a hydrological response up to the global scale of increasing runoff (e.g., Gedney et al., 2006 [JoC, ARC] ) that is consistent with expected stomatal responses to rising CO2 (e.g., Hetherington and Woodward, 2003 [JoC] ; Gedney et al., 2006 [JoC, ARC] ).
Soil nitrogen availability is key to predicting future carbon sequestration by terrestrial ecosystems ( Reich et al., 2006 [JoC] ), especially in light of global nitrogen-deposition trends (2-10 fold increase in some industrialised areas – ( Matson et al., 2002 ) ). The future ability of ecosystems overall to sequester additional carbon is very likely to be constrained by levels of nitrogen availability and fixation, and other key nutrients such as phosphorus that may also become increasingly limiting ( Hungate et al., 2003 [JoC, ARC] ). Carbon accumulation and sequestration in critical soil stocks (see Figure 4.1 ) has been found to be strongly nitrogen-constrained, both because levels well above typical atmospheric inputs are needed to stimulate soil C-sequestration, and because natural N2-fixation appears to be particularly strongly limited by key nutrients ( van Groenigen et al., 2006 [JoC] ).
Results from a loblolly pine forest ( (Lichter et al., 2005 ) ) and grassland experiments ( Van Kessel et al., 2000 [JoC] ) suggest a reduced likelihood for CO2-fertilisation-driven carbon accumulation in soils, probably because carbon sequestration to humus is more nutrient-demanding (not only nitrogen), than is wood formation, for example ( (Hungate et al., 2006 ) ). Carbon accretion in soil is therefore itself likely to exert negative feedback on plant growth by immobilising soil nutrients (in addition to cation depletion by acidic precipitation), contributing to a faster diminishing of the biospheric sink (see Figure 4.2 Reich et al., 2006 [JoC] ) than implemented in model projections (e.g., Scholze et al., 2006 [JoC, ARC] ; see Figure 4.2 ).
Accumulation of seasonally transitory soil carbon pools such as in fine roots has been found at elevated ambient CO2 concentrations, but the general validity of such enhanced C-fluxes and what fraction of these might be sequestered to recalcitrant (see Glossary) soil carbon stocks remains unresolved ( Norby et al., 2004 [Ambiguous] ). Soil warming may enhance carbon emissions, especially by reducing labile soil organic carbon pools ( Davidson and Janssens, 2006 [JoC] ). This results in the commonly observed short-term (less than decadal) loss of carbon in warming experiments, followed by the re-establishment of a new equilibrium between inputs and losses of soil carbon (e.g., Eliasson et al., 2005 [JoC] ; Knorr et al., 2005 [JoC] ). Recent observations indeed show widespread carbon losses from soils ( Bellamy et al., 2005 [JoC] ; Schulze and Freibauer, 2005 [JoC] ) that are consistent with this formulation. However, in regions with thawing permafrost, a decay of historically accumulated soil carbon stocks (yedoma, >10,000 years old, Figure 4.1 ) due to warming ( Zimov et al., 2006 [JoC] ) and nutrient deposition ( (Blodau, 2002; ) Mack et al., 2004 [JoC] ) could release large amounts of carbon to the atmosphere (see also Section 4.4.6 ). Increased NPP (but see Angert et al., 2005 [JoC] ) and vegetation change (see Section 4.4.5 and, e.g., Sturm et al., 2001 [JoC] ) may partly counterbalance this carbon release (see Section 4.4.6 and Sitch et al., 2007 [MoS, 2007] ), thus complicating projections ( (Blodau, 2002; ) for a full discussion see Section 4.4.6 ).
Ecosystem changes associated with land-use and land-cover change (see Section 4.2.2 ) are complex, involving a number of feedbacks ( Lepers et al., 2005 [ARC] ; Reid et al., 2005 [NPR, SRC] ). For example, conversion of natural vegetation to agricultural land drives climate change by altering regional albedo and latent heat flux, causing additional summer warming in key regions in the boreal and Amazon regions, and winter cooling in the Asian boreal zone ( Chapin et al., 2005b [JoC] ; Feddema et al., 2005 [JoC, MoS, ARC] ), by releasing CO2 via losses of biomass and soil carbon ( Gitz and Ciais, 2003 [MoS] ; Canadell et al., 2004 [JoC, ARC] ; Levy et al., 2004 [JoC, MoS] ) and through a ‘land-use amplifier effect’ ( Gitz and Ciais, 2003 [MoS] ). In contrast, reforestation, and other land-use or land-management changes such as modifications of agricultural practices, can work to mitigate climate change through carbon sequestration ( (Lal, 2003, ) 2004; ( Jones and Donnelly, 2004; ) ( King et al., 2004a; ) Wang et al., 2004a [JoC] ;( de Koning et al., 2005; ) Nabuurs et al., 2007 [NPR, 2007] ). This mitigation potential is probably limited to reducing the ultimate atmospheric CO2 increase by 2100 by between 40 and 70 ppm ( House et al., 2002 [JoC, MoS] ), and by approximately century-long time-lags until mature forests are established (see Sections 4.4.5 , 4.4.6 and 4.4.10 ), and is probably offset by regional warming effects of lower albedo with poleward boreal forest expansion (e.g., Betts, 2000 [JoC, ARC] ; for a full discussion see Section 4.4.6 ).
The sequestration and cycling of carbon in terrestrial ecosystems is a key vulnerability, given the above drivers, their generally global extent, their potential irreversibility, and the likely existence of threshold-type impacts. The extent to which the recently discovered methane release from plant foliage ( Keppler et al., 2006 [JoC] ) can be scaled to biome level is under debate ( Houweling et al., 2006 [JoC] ), and highlights the currently limited understanding of the methane cycle, and its exclusion from Earth system models (e.g., Betts and Shugart, 2005 [NPR, ARC] ). Nonetheless, recent work especially with DGVM approaches, has begun to elucidate the likelihood of occurrence of important thresholds, and positive feedback to the atmosphere through diminishing CO2 sequestration or even net carbon release from ecosystems, thus amplifying climate change (e.g., Friedlingstein et al., 2006 [PoC, JoC, MoS, ARC] ; Lucht et al., 2006 [SRC] ; Scholze et al., 2006 [JoC, ARC] ).
Global estimates (IS92a, HACM2-SUL – Cramer et al., 2001 [JoC, MoS, SRC] ) suggest a reduced global sink relative to that expected under CO2-fertilisation alone, both in 2000 (0.6±3.0 PgCy-1) and 2100 (0.3±6.6 PgCy-1) as a result of climate change impacts on Net Biome Productivity (NBP) of tropical and Southern Hemisphere ecosystems. According to these models, the rate of NBP increase slows by around 2030 as CO2-fertilisation itself saturates, and in four of six models shows further, climate-induced, NBP declines, due to increased heterotrophic respiration and declining tropical NPP after 2050 . These trends are projected to continue until mid-century, even with stabilised atmospheric CO2 concentration and instantaneously stabilised climate beyond 2100 ( Woodward and Lomas, 2004b [MoS] ; see also next paragraph, Figure 4.2 ). More recent modelling based on projected deforestation and climate change (for the IS92a emissions scenario and the CGCM1, CSIRO, ECHAM, HadCM3 climate models) in the tropics alone suggests an additional release of 101 to 367 PgC, adding between 29 and 129 ppm to global atmospheric CO2 by 2100, mainly due to deforestation ( Cramer et al., 2004 [SRC] ).
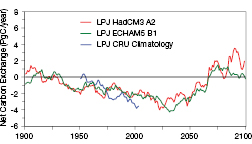
Figure 4.2. Net carbon exchange of all terrestrial ecosystems as simulated by the DGVM LPJ ( Sitch et al., 2003 [JoC, MoS, SRC] ; Gerten et al., 2004 [MoS, SRC] – negative values mean a carbon sink, positive values carbon losses to the atmosphere). Past century data are based on observations and climate model data were normalised to be in accord with these observations for the 1961 - 1990 data (CRU-PIK). Transient future projections are for the SRES A2 and B1 emissions scenarios (Naki´cenovi´c et al., 2000 [NotFound] ), forcing the climate models HadCM3 and ECHAM5, respectively (cf. Lucht et al., 2006 [SRC] ; Schaphoff et al., 2006 [JoC, MoS, SRC] ). In contrast to previous global projections ( Prentice et al., 2001 [NPR] – Figure 3.10), the world’s ecosystems sink service saturates earlier (about 2030 ) and the terrestrial biosphere tends to become a carbon source earlier (about 2070 ) and more consistently, corroborating other projections of increased forcing from biogenic terrestrial sources (e.g., Cox et al., 2000 [JoC, MoS, ARC] , 2004; White et al., 2000a [JoC] ; Lucht et al., 2006 [SRC] ; Schaphoff et al., 2006 [JoC, MoS, SRC] ; Scholze et al., 2006 [JoC, ARC] ; see Figure 4.3 for maps on underlying ecosystem changes). Note that these projections assume an effective CO2-fertilisation (see Section 4.4.1 ).
Climate scenario uncertainty provides a substantial variance in global terrestrial C balance by 2100, even under a single CO2 emissions scenario (IS92a, projected to reach 703 ppm atmospheric CO2 concentration by 2100, excluding vegetation feedback). Using five General Circulation Models (GCMs) to drive DGVMs, global terrestrial C-sequestration is estimated at between -106 and +201 PgC ( Schaphoff et al., 2006 [JoC, MoS, SRC] ), though in four out of five, the sink service decreased well before 2060 . A risk assessment for terrestrial biomes and biogeochemical cycling shows that a terrestrial carbon source is predicted in almost half of 52 GCM * emissions scenario combinations, and that wildfire frequency increases dramatically even for a warming of <2°C by 2100 ( Scholze et al., 2006 [JoC, ARC] ). Here we show model results for the most recent version of the DGVM Lund-Potsdam-Jena Model (LPJ) ( Schaphoff et al., 2006 [JoC, MoS, SRC] ) highlighting changes in biome structure (relative cover of dominant growth forms) and the terrestrial carbon sink under more recent IPCC emissions scenarios SRES A2 and B1 (Naki?enovi? et al., 2000 ). This supports projections of diminishing terrestrial C-sequestration as early as 2030 ( Figure 4.2 ) – earlier than suggested in the TAR ( Prentice et al., 2001 [NPR] , Figure 3.10) – and substantial shifts in biome structure ( Figure 4.3 ); discussed more fully in Sections 4.4.10 and 4.4.11 .
Projections from modelling that dynamically link the physical climate system and vegetation, using Ocean-Atmosphere General Circulation Models (OAGCMs, e.g., Cox et al., 2000 [JoC, MoS, ARC] ), suggest a terrestrial C source that will exacerbate both climate and further vegetation change to at least some degree (e.g., Sarmiento, 2000 [JoC] ; Dufresne et al., 2002 [JoC, MoS] ; Canadell et al., 2004 [JoC, ARC] ). Impacts include the collapse of the Amazon forest (e.g., White et al., 2000a [JoC] ; Cox et al., 2004 [MoS, ARC] ), and an overall C source from the tropics that exceeds the boreal C sink ( Berthelot et al., 2002 [MoS] ), leading to an 18% ( Dufresne et al., 2002 [JoC, MoS] ), 5 to 30% ( Friedlingstein et al., 2006 [PoC, JoC, MoS, ARC] ), and 40% ( Cox et al., 2000 [JoC, MoS, ARC] ) higher atmospheric CO2 concentration by 2100 . Carbon and water cycling, at least, are also affected by shifting biogeographical zones ( Gerten et al., 2005 [JoC, SRC] ) which will be lagged by migration constraints that are not yet incorporated in DGVM approaches (see also Sections 4.4.5 and 4.4.6 ), leading to a potential overestimation of vegetation C-sequestration potential. This is especially so for boreal regions, due to unrealistically high projections of in-migration rates of trees and shrubs ( Neilson et al., 2005 [MoS, SRC] ).
Changes in air-sea fluxes of dimethyl sulphide (DMS) from -15% to 30% caused by global warming of about 2°C are projected to have a regional radiative and related climatic impact ( (Bopp et al., 2003, ) 2004 ), as DMS is a significant source of cloud condensation nuclei. DMS is produced by coccolithophores, which are sensitive to high sea-water CO2 ( Riebesell et al., 2000 [JoC] ). As the largest producers of calcite on the planet ( (Holligan et al., 1993 ) ), reduced calcification by these organisms may also influence the global carbon cycle ( Raven et al., 2005 [NPR, SRC] ) and global albedo ( (Tyrrell et al., 1999 ) ). N2O of marine origin contributes about 33% of total input to the atmosphere ( Enhalt and Prather, 2001 [NPR] ). Changes to the concentration and distribution of oxygen in the oceans, either through increased stratification of the surface waters ( Sarmiento et al., 1998 [JoC, MoS] ) or through a decrease in the strength of the thermohaline circulation ( IPCC, 2001 [NPR] ), will impact the ocean nitrogen cycling, especially the processes of nitrification and denitrification which promote N2O production.
4.4.2 Deserts
Properties, goods and services
One of the largest terrestrial biomes, deserts cover 27.7 Mkm2, comprising extra-polar regions with mean annual precipitation <250 mm and an unfavourable precipitation to potential evaporation ratio ( Nicholson, 2002 [NPR] ; Warner, 2004 [NPR] ; Reid et al., 2005 [NPR, SRC] ). Deserts support on the order of 10 people per km2, in sparse populations with among the lowest gross domestic product (GDP) of all ecoregions ( Reid et al., 2005 [NPR, SRC] ). Recent estimates suggest that between 10 and 20% of deserts and drylands are degraded due to an imbalance between demand for and supply of ecosystem services ( Adeel et al., 2005 [NPR, ARC] ). Critical provisioning goods and services include wild food sources, forage and rangeland grazing, fuel, building materials, and water for humans and livestock, for irrigation and for sanitation, and genetic resources, especially of arid-adapted species ( Adeel et al., 2005 [NPR, ARC] ; Hassan et al., 2005 [NPR] ). Regulating services include air quality, atmosphere composition and climate regulation ( Hassan et al., 2005 [NPR] ), especially through wind-blown dust and desert albedo influences on regional rainfall, and biogeochemistry of remote terrestrial and marine ecosystems ( Warner, 2004 [NPR] ).
Key vulnerabilities
The TAR noted several vulnerabilities in drylands ( Gitay et al., 2001 [NPR, ARC] , p. 239) but chiefly that human overuse and land degradation, exacerbated by an overall lack of infrastructure and investment in resource management, would be very likely to overwhelm climate change impacts, with the exception of impacts of increased dry and wet extremes due to ENSO frequency increase, and negative impacts of projected warming and drying in high biodiversity regions. On the other hand, evidence for region-specific increases in productivity and even community compositional change due to rising atmospheric CO2 was reported, with associated increased biomass and soil organic matter. Overall impacts of elevated CO2 were reported as comparable, though usually opposite in sign, to climate change projections. Since the TAR, further work shows that desert biodiversity is likely to be vulnerable to climate change ( Reid et al., 2005 [NPR, SRC] ), with winter-rainfall desert vegetation and plant and animal species especially vulnerable to drier and warmer conditions ( Lenihan et al., 2003 [SRC] ; Simmons et al., 2004 [SRC] ; Musil et al., 2005 [MoS, SRC] ; Malcolm et al., 2006 [SRC] ), and continental deserts vulnerable to desiccation and even soil mobilisation, especially with human land-use pressures ( (Thomas and Leason, 2005 ) ). However, the potentially positive impact of rising atmospheric CO2 remains a significant uncertainty, especially because it is likely to increase plant productivity, particularly of C3 plants ( Thuiller et al., 2006b [JoC, SRC] ) and, together with rainfall change, could even induce wildfires ( Bachelet et al., 2001 [SRC] ; Hardy, 2003 [NPR] ; Duraiappah et al., 2005 [NPR] ). The uncertain impact of elevated CO2 on vegetation productivity and biogeochemical cycling in deserts is an important source of contrasting projections of impacts and vulnerability for different desert regions and vegetation types. Climate change and direct human land-use pressure are likely to have synergistic impacts on desert ecosystems and species that may be offset, at least partly, by vegetation productivity and carbon sequestration gains due to rising atmospheric CO2. The net effect of these trends is very likely to be region-specific.
Impacts
Deserts are likely to experience more episodic climate events, and interannual variability may increase in future, though there is substantial disagreement between GCM projections and across different regions ( Smith et al., 2000 [JoC] ; Duraiappah et al., 2005 [NPR] ). Continental deserts could experience more severe, persistent droughts ( (Lioubimtseva and Adams, 2004; ) ( Schwinning and Sala, 2004 ) ). Vulnerability to desertification will be enhanced due to the indicated increase in the incidence of severe drought globally ( Burke et al., 2006 [JoC, MoS] ). In the Americas, temperate deserts are projected to expand substantially under doubled CO2 climate scenarios ( (Lauenroth et al., 2004 ) ). However, dry-spell duration and warming trend effects on vegetation productivity may be at least partly offset by rising atmospheric CO2 effects on plants ( Bachelet et al., 2001 [SRC] ; Thuiller et al., 2006b [JoC, SRC] ), leading to sometimes contrasting projections for deserts that are based on different modelling techniques that either incorporate or ignore CO2-fertilisation effects.
Elevated CO2 has been projected to have significant potential impacts on plant growth and productivity in drylands ( (Lioubimtseva and Adams, 2004 ) ). This projection has been confirmed for cool desert shrub species ( Hamerlynck et al., 2002 [NPR] ), and both desert shrubs and invasive (but not indigenous) grasses in wet years only ( Smith et al., 2000 [JoC] ). On the whole, evidence for CO2-fertilisation effects in deserts is conflicting, and species-specific ( (Lioubimtseva and Adams, 2004; ) Morgan et al., 2004 [SRC] ). In the south-western USA the total area covered by deserts may decline by up to 60% if CO2-fertilisation effects are realised ( Bachelet et al., 2001 [SRC] ). Limited direct impacts of atmospheric CO2 on nitrogen-fixation have been found in soil biological crusts ( (Billings et al., 2003 ) ), but soil microbial activity beneath shrubs has been observed to increase, thus reducing plant-available nitrogen ( (Billings et al., 2002 ) ).
Soil vulnerability to climate change is indicated by shallow desert substrates with high soluble salts and the slow recolonisation of disturbed soil surfaces by different algae components ( (Evans and Belnap, 1999; ) Johansen, 2001 [NPR] ; Duraiappah et al., 2005 [NPR] ). Very low biomass (a drop below a 14% cover threshold) is very likely to make the Kalahari desert dune system in southern Africa susceptible to aeolian erosion ( (Thomas and Leason, 2005 ) ) and, with regional warming of between 2.5 and 3.5°C, most dune fields could be reactivated by 2100 ( (Thomas and Leason, 2005 ) ). Increased dust flux may increase aridity and suppress rainfall outside deserts, with opposite effects under wetting scenarios ( Bachelet et al., 2001 [SRC] ; Hardy, 2003 [NPR] ; Prospero and Lamb, 2003 [JoC] ;( Lioubimtseva and Adams, 2004 ) ), leading to indirect effects on the vulnerability of remote regions to climate change. About one-third of the Sahel was projected to aridify with warming of 1.5 to 2°C by about 2050, with a general equatorward shift of vegetation zones ( van den Born et al., 2004 [NPR, SRC] ; Box 4.2 ). Alternative climate scenarios show less pronounced changes ( van den Born et al., 2004 [NPR, SRC] ).
Box 4.2. Vegetation response to rainfall variability in the Sahel
The Sahel falls roughly between the 100-200 mm/year (northern boundary) and 400-600 mm/year rainfall isohyets (southern boundary), and supports dry savanna vegetation forming transition zones with the Sahara and humid tropical savanna ( (Nicholson, 2000; ) Hiernaux and Turner, 2002 [NPR] ;( Anyamba and Tucker, 2005 ) ). These transition zones have historically fluctuated in response to rainfall changes ( Hiernaux and Turner, 2002 [NPR] ), in the clearest example of multi-decadal variability measured during the past century ( Hulme, 2001 [JoC] ). Ecosystem responses to past rainfall variability in the Sahel are potentially useful as an analogue of future climate change impacts, in the light of projections that extreme drought-affected terrestrial areas will increase from 1% to about 30% globally by the 2090 s ( Burke et al., 2006 [JoC, MoS] ).
During the mid-Holocene, conditions supporting mesic vegetation and abundant wildlife deteriorated rapidly ( ECF, 2004 [Ambiguous] ;( Foley et al., 2003 ) ), highlighting the Sahel’s sensitivity to forcing effects. The Sahel has shown the largest negative trends in annual rainfall observed globally in the past century, though these reversed somewhat after the late 1970 s ( Trenberth et al., 2007 [NPR, PoC, 2007] ). Since about 1900, multi-decadal-scale rainfall variability persisted, with drying trends between around 1930 - 1950 and 1960 - 1985 ( Hulme, 2001 [JoC] ;( Nicholson, 2001 ) ). Conditions apparently improved between 1950 and 1960, with limited evidence suggesting increased human and livestock numbers ( (Reij et al., 2005 ) ). Severe drought prevailed in the early 1980 s ( Hulme, 2001 [JoC] ; Trenberth et al., 2007 [NPR, PoC, 2007] ), and groundwater levels declined, species-specific woody plant mortality increased (mainly of smaller plants), and even dominant perennial C4 grasses with high water-use efficiency declined. Exposed soil caused increased atmospheric dust loads ( (Nicholson, 2000, ) 2001 ). These events stimulated the concept of desertification and subsequent debates on its causes ( (Herrmann and Hutchinson, 2005 ) ).
The persistence of drought during the latter part of the 20th century prompted suggestions that land-cover change had exerted a positive feedback to reinforce drought conditions, but the modelled vegetation change necessary to induce this effect does not reflect reality ( Hulme, 2001 [JoC] ). During relatively wet periods ( (Nicholson et al., 2000; ) ( Anyamba and Tucker, 2005; ) Trenberth et al., 2007 [NPR, PoC, 2007] ) spatially variable regeneration in both the herbaceous and the woody layer have been observed ( (Gonzalez, 2001; ) Rasmussen et al., 2001 [JoC] ; Hiernaux and Turner, 2002 [NPR] ). Remote sensing shows the resilience of Sahelian vegetation to drought, with no directional change in either desert transition zone position or vegetation cover ( Nicholson et al., 1998 [JoC] ). Sahel green-up between the years 1982 and 1998 ( Prince et al., 1998 [JoC] ; Hickler et al., 2005 [JoC] ) and between 1994 and 2003 ( (Anyamba and Tucker, 2005 ) ) has been noted, but this interpretation has recently been challenged ( Hein and Ridder, 2006 [JoC] ).
Drivers of Sahel vegetation change remain uncertain ( (Hutchinson et al., 2005 ) ), especially because the correlation between rainfall and Normalised Difference Vegetation Index (NDVI) appear weak, signalling that greening cannot be fully explained by increasing rainfall ( (Olsson et al., 2005 ) ), and greening may not comprise a return to the initial species composition, cover and surface soil conditions ( (Warren, 2005 ) ). Inconclusive interpretations of vegetation dynamics in the Sahel may reflect complex combined effects of human land use and climate variability on arid environments ( Rasmussen et al., 2001 [JoC] ). It is far from clear how the interactive effect of climate change, land-use activities and rising CO2 will influence the Sahel in future. Green-up, or desert amelioration ( Figure 4.3 , vegetation class 4) due to rising CO2 and enhanced water-use efficiency (as observed by Herrmann et al., 2005 [Ambiguous] ) may accrue only in wet years ( Morgan et al., 2004 [SRC] ).
Episodic wet periods may increase vulnerability to invasive alien species and subsequent fire outbreaks and this, combined with land overuse, will increase vulnerability to degradation and desertification ( (Dukes and Mooney, 1999; ) ( Dube and Pickup, 2001; ) ( Holmgren and Scheffer, 2001; ) ( Brooks et al., 2004; ) ( Geist and Lambin, 2004; ) ( Lioubimtseva and Adams, 2004 ) ). Wet spells with elevated humidity and warmer temperatures will increase the prevalence of plant diseases ( Harvell et al., 2002 [JoC] ).
Desert biodiversity is likely to be vulnerable to climate change ( Reid et al., 2005 [NPR, SRC] ), especially in so-called ‘biodiversity hotspots’ ( Myers et al., 2000 [JoC] ). In the Succulent Karoo biome of South Africa, 2,800 plant species face potential extinction as bioclimatically suitable habitat is reduced by 80% with a global warming of 1.5-2.7°C above pre-industrial levels (see Table 4.1 ). Daytime in situ warming experiments suggest high vulnerability of endemic succulent (see Glossary) growth forms of the Succulent Karoo to high-end warming scenarios for 2100 (mean 5.5°C above current ambient temperatures), inducing appreciable mortality in some (but not all) succulent species tested within only a few months ( Musil et al., 2005 [MoS, SRC] ). Desert species that depend on rainfall events to initiate breeding, such as resident birds, and migratory birds whose routes cross deserts, will be severely affected ( (Dukes and Mooney, 1999; ) Hardy, 2003 [NPR] ; Box 4.5 ). The Mountain Wheatear in South Africa was projected to lose 51% of its bioclimatic range by 2050 under an SRES A2 emissions scenario ( Simmons et al., 2004 [SRC] ). In contrast, desert reptile species could be favoured by warming, depending on rainfall scenario ( (Currie, 2001 ) ).
4.4.3 Grasslands and savannas
Properties, goods and services
Dominated by a spatially and temporally variable mix of grass and tree-growth forms ( (Sankaran et al., 2005 ) ), grasslands and savannas include tropical C4 grasslands and savannas (C4 grass-dominated with 10-50% tree cover, about 28 Mkm2) and temperate C4 and/or C3-grass and herb-dominated grasslands (15 million km2 Bonan, 2002 [NPR] ). Generally rich in grazing, browsing and other fauna (especially but not only in Africa), these systems are strongly controlled by fire ( Bond et al., 2005 [Ambiguous] ) and/or grazing regimes ( (Scholes and Archer, 1997; ) ( Fuhlendorf et al., 2001 ) ). Disturbance regimes are often managed (e.g., ( Sankaran, 2005 ) ), although fire regimes depend also on seasonality of ignition events and rainfall-dependent accumulation of flammable material ( Brown et al., 2005b [JoC] ). Temperate and tropical systems provide somewhat distinct goods and services. Temperate grasslands contain a substantial soil carbon pool, are important for maintaining soil stability and provide fodder for wild and domestic animals. Tropical savanna systems possess significant wild faunal diversity that supports nature-based tourism revenue (both extractive and non-extractive) and subsistence livelihoods (food, medicinal plants, and construction material), in addition to cultural, regulating and supporting services.
Key vulnerabilities
The structure, productivity and carbon balance of these systems appear more sensitive than indicated in the TAR to variability of, and changes in, major climate change drivers. The direct CO2-fertilisation impact and warming effect of rising atmospheric CO2 have contrasting effects on their dominant functional types (trees and C3 grasses may benefit from rising CO2 but not from warming; C4 grasses may benefit from warming, but not from CO2-fertilisation), with uncertain, non-linear and rapid changes in ecosystem structure and carbon stocks likely. Carbon stocks are very likely to be strongly reduced under more frequent disturbance, especially by fire, and disturbance and drought impacts on cover may exert regional feedback effects. On balance, savannas and grasslands are likely to show reduced carbon sequestration due to enhanced soil respiratory losses through warming, fire regime changes and increased rainfall variability, but possible regional gains in woody cover through direct CO2-fertilisation, and increased plant carbon stocks, cannot be excluded. Scientific predictive skill is currently limited by very few field-based, multi-factorial experiments, especially in tropical systems. Projected range shifts of mammal species will be limited by fragmented habitats and human pressures, as suggested in the TAR, with declines in species richness likely, especially in protected areas. Because of the important control by disturbance, management options exist to develop adaptive strategies for carbon sequestration and species conservation goals.
Impacts
Ecosystem function and species composition of grasslands and savanna are likely to respond mainly to precipitation change and warming in temperate systems but, in tropical systems, CO2-fertilisation and emergent responses of herbivory and fire regime will also exert strong control. Very few experimental approaches have assessed ecosystem responses to multi-factorial treatments such as listed above ( (Norby and Luo, 2004 ) ), and experiments on warming, rainfall change or atmospheric CO2 level are virtually absent in savannas, with many ecosystem studies confined mainly to temperate grasslands ( (Rustad et al., 2001 ) ).
Rainfall change and variability is very likely to affect vegetation in tropical grassland and savanna systems with, for example, a reduction in cover and productivity simulated along an aridity gradient in southern African savanna in response to the observed drying trend of about 8 mm/yr since 1970 ( Woodward and Lomas, 2004a [JoC, MoS] ). Sahelian woody plants, for example, have shown drought-induced mass mortality and subsequent regeneration during wetter periods ( Hiernaux and Turner, 2002 [NPR] ). Large-scale changes in savanna vegetation cover may also feed back to regional rainfall patterns. Modelled removal of savannas from global vegetation cover has larger effects on global precipitation than for any other biome ( Snyder et al., 2004 [JoC] ) and, in four out of five savannas studied globally, modelled savanna-grassland conversion resulted in 10% lower rainfall, suggesting positive feedback between human impacts and changing climate ( Hoffmann and Jackson, 2000 [JoC] ). At the continental scale, modelled forest-savanna conversion reduced rainfall in tropical African regions, but increased it in central southern Africa ( (Semazzi and Song, 2001 ) ).
Changing amounts and variability of rainfall may also strongly control temperate grassland responses to future climate change ( (Novick et al., 2004; ) ( Zha et al., 2005 ) ). A Canadian grassland fixed roughly five times as much carbon in a year with 30% higher rainfall, while a 15% rainfall reduction led to a net carbon loss ( Flanagan et al., 2002 [JoC] ). Similarly, Mongolian steppe grassland switched from carbon sink to source in response to seasonal water stress, although carbon balance was neutral on an annual basis ( Li et al., 2005 [JoC, ARC] ). Non-linear responses to increasing rainfall variability may be expected, as ecosystem models of mixed C3/C4 grasslands show initially positive NPP relationships with increasing rainfall variability, but greater variability ultimately reduces both NPP and ecosystem stability even if the rainfall total is kept constant ( Mitchell and Csillag, 2001 [MoS] ). Empirical results for C4 grasslands confirm a similar monotonic (hump-backed) relationship between NPP and rainfall variability ( Nippert et al., 2006 [MoS] ). Increased rainfall variability was more significant than rainfall amount for tall-grass prairie productivity ( (Fay et al., 2000, ) 2002 ), with a 50% increase in dry-spell duration causing 10% reduction in NPP ( (Fay et al., 2003 ) ) and a 13% reduction in soil respiration ( Harper et al., 2005 [JoC] ).
The CO2-fertilisation and warming effect of rising atmospheric CO2 have generally opposite effects on savanna- and grassland-dominant functional types, with CO2-fertilisation favouring woody C3 plants ( (Ainsworth and Long, 2005 ) ), and warming favouring C4 herbaceous types ( (Epstein et al., 2002 ) ). Simulated heat-wave events increased C4 dominance in a mixed C3/C4 New Zealand grassland within a single growing season, but reduced productivity by over 60% where C4 plants were absent ( White et al., 2000b [JoC, MoS] ). Some African savanna trees are sensitive to seasonal high air temperature extremes ( (Chidumayo, 2001 ) ). North American forest vegetation types could spread with up to 4°C warming; but with greater warming, forest cover could be reduced by savanna expansion of up to 50%, partly due to the impacts of fire ( Bachelet et al., 2001 [SRC] ).
Elevated CO2 has important effects on production and soil water balance in most grassland types, mediated strongly by reduced stomatal conductance and resulting increases in soil water ( (Leakey et al., 2006 ) ) in many grassland types ( (Nelson et al., 2004; ) Niklaus and Körner, 2004 [SRC] ; Stock et al., 2005 [SRC] ). In short-grass prairie, elevated CO2 and 2.6°C warming increased production by 26-47% , regardless of grass photosynthetic type ( Morgan et al., 2001a [JoC] ). In C4 tropical grassland, no relative increase in herbaceous C3 success occurred in double-ambient CO2 ( Stock et al., 2005 [SRC] ). Regional climate modelling indicates that CO2-fertilisation effects on grasslands may scale-up to affect regional climate ( Eastman et al., 2001 [JoC, MoS] ).
Differential effects of rising atmospheric CO2 on woody relative to herbaceous growth forms are very likely ( Bond and Midgley, 2000 [JoC, SRC] ). Trees and shrubs show higher CO2 responsiveness than do herbaceous forms ( (Ainsworth and Long, 2005 ) ). Savannas may thus be shifting towards greater tree dominance as atmospheric CO2 rises, with diminishing grass suppression of faster-growing tree saplings ( Bond et al., 2003 [Ambiguous] ). Simulations suggest that rising CO2 may favour C3 forms at the expense of African C4 grasses ( Thuiller et al., 2006b [JoC, SRC] ), even under projected warming. Continuing atmospheric CO2 rise could increase the resilience of Sahelian systems to drought ( Wang and Eltahir, 2002 [JoC] ). However, without definitive tests of the CO2-fertilisation effect on savanna trees, other factors can be invoked to explain widely observed woody plant encroachment in grassland systems ( (Van Auken, 2000 ) ).
Above-ground carbon stocks in savannas are strongly contingent on disturbance regimes. Australian savanna systems are currently a net carbon sink of 1-3 t C/ha/yr, depending on fire frequency and extent ( (Williams et al., 2004b ) ). Fire exclusion can transform savannas to forests (e.g., ( Bowman et al., 2001 ) ), with an upper (albeit technically unfeasible) global estimate of potential doubling of closed forest cover ( Bond et al., 2005 [Ambiguous] ). Thus savanna structure and carbon stocks are very likely to be responsive to both individual and interactive effects of the disturbance regime ( Bond et al., 2003 [Ambiguous] ;( Sankaran et al., 2005 ) ) and atmospheric CO2 change ( Bond and Midgley, 2000 [JoC, SRC] ).
There are few factorial experiments on multiple changing factors, but they suggest interactions that are not predictable from single factor experiments – such as the dampening effect of elevated CO2 on California C3 grassland responses to increased rainfall, nitrate and air temperature ( Shaw et al., 2002 [JoC, ARC] ). Increasing temperature and rainfall changes are seen to override the potential benefits of rising CO2 for C3 relative to C4 grasses ( (Winslow et al., 2003 ) ), and European C3 grassland showed minor responses to a 3°C rise in temperature, possibly due to concomitant drying impacts ( Gielen et al., 2005 [MoS, ARC] ). Elevated CO2 impacts on grassland carbon sequestration also seem to be dependent on management practices ( (Harmens et al., 2004; ) ( Jones and Donnelly, 2004 ) ), and are complicated by being species- but not functional-type specific ( Niklaus et al., 2001 [SRC] ; Hanley et al., 2004 [MoS] ).
Soil-mediated responses are important in biogeochemical controls of vegetation response. Long-term CO2 enrichment of southern African C4 grassland revealed limited impacts on nitrogen cycling and soil C sequestration ( Stock et al., 2005 [SRC] ), in contrast to greater C sequestration in short-term studies of grassland ecosystems (e.g., ( Williams et al., 2004a ) ). Likewise, elevated CO2 impacts on litter decomposition and soil fauna seem species-specific and relatively minor ( (Ross et al., 2002; ) ( Hungate et al., 2000 ) ). Warming of a tall-grass prairie showed increased plant growth that supported enhanced soil fungal success ( Zhang et al., 2005 [JoC] ). However, complex interactions between plants and fungal symbionts showed potential impacts on soil structure that may predispose them to accelerated erosion ( Rillig et al., 2002 [ARC] ). Soil respiration shows approximately 20% increase in response to about 2.4°C warming ( Norby et al., 2007 [NPR, 2007] ), although acclimatisation of soil respiration ( Luo et al., 2001 [JoC] ) and root growth ( Edwards et al., 2004 [Ambiguous] ) to moderate warming has also been observed. Soil carbon loss from UK soils, many in grasslands, confirm carbon losses of about 2% per annum in carbon-rich soils, probably related to regional climate change ( Bellamy et al., 2005 [JoC] ). In an African savanna system, rainfall after a dry spell generates substantial soil respiration activity and soil respiratory carbon losses ( Veenendaal et al., 2004 [JoC] ), suggesting strong sensitivity to rainfall variability.
Climate change impact studies for savanna and grassland fauna are few. The proportion of threatened mammal species may increase to between 10 and 40% between 2050 and 2080 ( Thuiller et al., 2006a [JoC, SRC] ). Changing migration routes especially threaten migratory African ungulates and their predators ( (Thirgood et al., 2004 ) ). Observed population declines in three African savanna ungulates suggest that summer rainfall reductions could result in their local extirpation if regional climate change trends are sustained ( (Ogutu and Owen-Smith, 2003 ) ). For an African arid savanna raptor, population declines have been simulated for drier, more variable rainfall scenarios ( (Wichmann et al., 2003 ) ). A 4 to 98% species range reduction for about 80% of mainly savanna and grassland animal species in South Africa is projected under an IS92a emissions scenario ( Erasmus et al., 2002 [JoC] ).
4.4.4 Mediterranean ecosystems
Properties, goods and services
Mediterranean-type ecosystems (MTEs) are located in mid-latitudes on all continents (covering about 3.4 Mkm2), often on nutrient-poor soils and in coastal regions. These biodiverse systems ( (Cowling et al., 1996 ) ) are climatically distinct, with generally wet winters and dry summers ( (Cowling et al., 2005 ) ), and are thus fire-prone ( (Montenegro et al., 2004 ) ). Vegetation structure is mainly shrub-dominated, but woodlands, forests and even grasslands occur in limited regions. Heavily utilised landscapes are dominated by grasses, herbs and annual plant species ( (Lavorel, 1999 ) ). MTEs are valuable for high biodiversity overall ( Myers et al., 2000 [JoC] ) and thus favour nature-based tourism, but many extractive uses include wildflower harvesting in South Africa and Australia, medicinal herbs and spices, and grazing in the Mediterranean Basin and Chile. Water yield for human consumption and agriculture is critical in South Africa, and these systems provide overall soil-protection services on generally unproductive nutrient-poor soils.
Key vulnerabilities
Mediterranean-type ecosystems were not explicitly reviewed in the TAR, but threats from desertification were projected due to expansion of adjacent semi-arid and arid systems under relatively minor warming and drying scenarios. Warming and drying trends are likely to induce substantial species-range shifts, and imply a need for migration rates that will exceed the capacity of many endemic species. Land use, habitat fragmentation and intense human pressures will further limit natural adaptation responses, and fire-regime shifts may threaten specific species and plant functional types. Vegetation structural change driven by dominant, common or invasive species may also threaten rare species. Overall, a loss of biodiversity and carbon sequestration services may be realised over much of these regions.
Impacts
These systems may be among the most impacted by global change drivers ( Sala et al., 2000 [JoC, SRC] ). Diverse Californian vegetation types may show substantial cover change for temperature increases greater than about 2°C, including desert and grassland expansion at the expense of shrublands, and mixed deciduous forest expansion at the expense of evergreen conifer forest ( Hayhoe et al., 2004 [PoC, JoC, SRC] ). The bioclimatic zone of the Cape Fynbos biome could lose 65% of its area under warming of 1.8°C relative to 1961 - 1990 (2.3°C, pre-industrial), with ultimate species extinction of 23% resulting in the long term ( Thomas et al., 2004b [JoC, MoS, SRC] ). For Europe, only minor biome-level shifts are projected for Mediterranean vegetation types ( Parry, 2000 [NPR, ARC] ), contrasting with between 60 and 80% of current species projected not to persist in the southern European Mediterranean region (global mean temperature increase of 1.8°C – Bakkenes et al., 2002 [JoC, MoS, SRC] ). Inclusion of hypothetical and uncertain CO2-fertilisation effects in biome-level modelling may partly explain this contrast. Land abandonment trends facilitate ongoing forest recovery ( Mouillot et al., 2003 [ARC] ) in the Mediterranean Basin, complicating projections. In south-western Australia, substantial vegetation shifts are projected under double CO2 scenarios ( Malcolm et al., 2002b [MoS, SRC] ).
Climate change is likely to increase fire frequency and fire extent. Greater fire frequencies are noted in Mediterranean Basin regions ( Pausas and Abdel Malak, 2004 [NPR] ) with some exceptions ( Mouillot et al., 2003 [ARC] ). Double CO2 climate scenarios increase wildfire events by 40-50% in California ( Fried et al., 2004 [JoC, MoS, ARC] ), and double fire risk in Cape Fynbos ( Midgley et al., 2005 [NPR, SRC] ), favouring re-sprouting plants in Fynbos ( Bond and Midgley, 2003 [SRC] ), fire-tolerant shrub dominance in the Mediterranean Basin ( Mouillot et al., 2002 [JoC, MoS] ), and vegetation structural change in California (needle-leaved to broad-leaved trees, trees to grasses) and reducing productivity and carbon sequestration ( Lenihan et al., 2003 [SRC] ).
Projected rainfall changes are spatially complex (e.g., Sumner et al., 2003 [JoC, MoS] ; Sanchez et al., 2004 [MoS] ;( Vicente-Serrano et al., 2004 ) ). Rainfall frequency reductions projected for some Mediterranean regions (e.g., ( Cheddadi et al., 2001 ) ) will exacerbate drought conditions, and have now been observed in the eastern Mediterranean ( Körner et al., 2005b [SRC] ). Soil water content controls ecosystem water and CO2 flux in the Mediterranean Basin system ( Rambal et al., 2003 [JoC] ), and reductions are very likely to reduce ecosystem carbon and water flux ( Reichstein et al., 2002 [JoC] ). The 2003 European drought had major physiological impacts on Mediterranean vegetation and ecosystems, but most appeared to have recovered from drought by 2004 ( (Gobron et al., 2005; ) Box 4.1 ).
Many MTE species show apparently limited benefits from rising atmospheric CO2 ( Dukes et al., 2005 [ARC] ), with constrained increases in above-ground productivity (e.g., ( Blaschke et al., 2001; ) ( Maroco et al., 2002 ) ). Yet modelling suggests that under all but extremely dry conditions, CO2 increases over the past century have already increased NPP and leaf area index (see Glossary) in the Mediterranean Basin, despite warming and drying trends ( Osborne et al., 2000 [JoC, MoS] ). Rising atmospheric CO2 appears increasingly unlikely to have a major impact in MTEs over the next decades, especially because of consistent projections of reduced rainfall. Elevated CO2 is projected to facilitate forest expansion and greater carbon storage in California if precipitation increases ( Bachelet et al., 2001 [SRC] ). In the Mediterranean Basin, CO2-fertilisation impacts such as increased forest success in the eastern Mediterranean and Turkey and increased shrub cover in northern Africa are simulated if rainfall does not decrease ( (Cheddadi et al., 2001 ) ). There is currently insufficient evidence to project elevated CO2-induced shifts in ecosystem carbon stocks in MTE, but nutrient-limited systems appear relatively unaffected ( de Graaff et al., 2006 [JoC] ). Established Pinus halepensis ( (Borghetti et al., 1998 ) ) show high drought resistance, but Ponderosa pine forests had reduced productivity and water flux during a 1997 heatwave, and did not recover for the rest of the season, indicating threshold responses to extreme events ( (Goldstein et al., 2000 ) ). Mediterranean Basin pines ( (Martinez-Vilalta and Pinol, 2002 ) ) and other woody species ( (Peñuelas et al., 2001 ) ) showed species-specific drought tolerance under field conditions. Experimental drying differentially reduced productivity of Mediterranean Basin shrub species ( (Llorens et al., 2003, ) 2004; ( Ogaya and Peñuelas, 2004 ) ) and tree species ( (Ogaya and Peñuelas, 2003 ) ), but delayed flowering and reduced flower production of Mediterranean Basin shrub species ( Llorens and Peñuelas, 2005 [MoS] ), suggesting complex changes in species relative success under drying scenarios. Drought may also act indirectly on plants by reducing the availability of soil phosphorus ( (Sardans and Peñuelas, 2004 ) ).
Bioclimatic niche-based modelling studies project reduced endemic species’ geographical ranges and species richness in the Cape Floristic region ( Midgley et al., 2002 [SRC] , 2003, 2006 ). Ranges of trees and shrubs may shift unpredictably, and fragment, under IS92a emissions scenarios ( Shafer et al., 2001 [MoS] ). In southern Europe, species composition change may be high under a range of scenarios ( Thuiller et al., 2005b [JoC, SRC] ). Range size reductions increase species’ extinction risks, with up to 30 to 40% facing increased extinction probabilities beyond 2050 ( Thomas et al., 2004a [JoC, SRC] ). Species of lowland plains may be at higher risk than montane species both in California ( Peterson, 2003 [JoC] ) and the Cape Floristic region ( Midgley et al., 2003 [SRC] ), although in the Mediterranean Basin, montane species show high risk ( Thuiller et al., 2005b [JoC, SRC] ).
4.4.5 Forests and woodlands
Properties, goods and services
Forests are ecosystems with a dense tree canopy (woodlands have a largely open canopy), covering a total of 41.6 Mkm2 (about 30% of all land) with 42% in the tropics, 25% in the temperate, and 33% in the boreal zone ( Figure 4.1 , e.g., Sabine et al., 2004 [NPR, ARC] ). Forests require relatively favourable environmental conditions and are among the most productive terrestrial ecosystems ( Figure 4.1 ). This makes them attractive both for climate change mitigation ( Watson et al., 2000 [NPR] ; Nabuurs et al., 2007 [NPR, 2007] ) and agricultural uses. The latter underlies the currently high deforestation and degradation rates in tropical and sub-tropical regions ( Hassan et al., 2005 [NPR] ), leading to about one-quarter of anthropogenic CO2 emissions (e.g., Houghton, 2003a [MoS] ). Nevertheless, forests sequester the largest fraction of terrestrial ecosystem carbon stocks, recently estimated at 1,640 PgC ( Sabine et al., 2004 [NPR, ARC] ; Figure 4.1 ), equivalent to about 220% of atmospheric carbon. In addition to commercial timber goods (see Chapter 5 Shvidenko et al., 2005 [NPR, ARC] , Section 21.5, p. 600-607) forests provide numerous non-timber forest products, important for subsistence livelihoods ( Gitay et al., 2001 [NPR, ARC] ; Shvidenko et al., 2005 [NPR, ARC] ). Key ecosystem services include habitat provision for an increasing fraction of biodiversity (in particular where subject to land-use pressures – Hassan et al., 2005 [NPR] ; Duraiappah et al., 2005 [NPR] ), carbon sequestration, climate regulation, soil and water protection or purification (>75% of globally usable freshwater supplies come from forested catchments – Shvidenko et al., 2005 [NPR, ARC] ), and recreational, cultural and spiritual benefits (Millennium Ecosystem Assessment, 2005 [MoS, ARC] ; Reid et al., 2005 [NPR, SRC] ).
Key vulnerabilities
Forests, especially in the boreal region, have been identified as having a high potential vulnerability to climate change in the long term ( Kirschbaum and Fischlin, 1996 [NPR, SRC] ), but more immediately if disturbance regimes (drought, insects, fire), partly due to climate change, cross critical thresholds ( Gitay et al., 2001 [NPR, ARC] ). Since the TAR, most DGVM models based on A2 emissions scenarios show significant forest dieback towards the end of this century and beyond in tropical, boreal and mountain areas, with a concomitant loss of key services ( Figure 4.3 ). Species-based approaches suggest losses of diversity, in particular in tropical forest diversity hotspots (e.g., north-eastern Amazonia – Miles, 2002 [NPR] ) and tropical Africa ( (McClean et al., 2005 ) ), with medium confidence. Mountain forests are increasingly encroached upon from adjacent lowlands, while simultaneously losing high-altitude habitats due to warming (see also Section 4.4.7 ).
Impacts
Projections for some forests currently limited by their minimum climatic requirements indicate gains from climate change ( Figure 4.3 , vegetation changes 1 and 2), but many may be impacted detrimentally ( Figure 4.3 , vegetation change 6), notably for strong warming and its concomitant effects on water availability ( Bachelet et al., 2001 [SRC] , 2003; Bergengren et al., 2001 [JoC, MoS] ;( Ostendorf et al., 2001; ) Smith and Lazo, 2001 [JoC, ARC] ;( Xu and Yan, 2001; ) Arnell et al., 2002 [JoC, ARC] ; Enquist, 2002 [MoS] ;( Iverson and Prasad, 2002; ) ( Lauenroth et al., 2004; ) Levy et al., 2004 [JoC, MoS] ; Matsui et al., 2004 [MoS] ; Izaurralde et al., 2005 [JoC] ; Fuhrer et al., 2006 [JoC, SRC] ; Lucht et al., 2006 [SRC] ; Schaphoff et al., 2006 [JoC, MoS, SRC] ; Scholze et al., 2006 [JoC, ARC] ; cf. Figure 4.3 a versus b, vegetation change 6). Productivity gains may result through three mechanisms: (i) CO2-fertilisation (although the magnitude of this effect remains uncertain in these long-lived systems, see Section 4.4.1 ); (ii) warming in cold climates, given concomitant precipitation increases to compensate for possibly increasing water vapour pressure deficits; and (iii) precipitation increases under water-limited conditions.
There is growing evidence (see Chapter 5 , Section 5.4.1.1 ) that several factors may moderate direct CO2 or climate-change effects on net ecosystem productivity in particular, namely nutrient dynamics (e.g., either enrichment or leaching resulting from N deposition), species composition, dynamic age structure effects, pollution and biotic interactions, particularly via soil organisms, (e.g., ( Karnosky et al., 2003; ) King et al., 2004b [JoC] ; Heath et al., 2005 [JoC] ; Körner et al., 2005a [JoC, SRC] ; Section 4.4.1 ). Climate change impacts on forests will result not only through changes in mean climate, but also through changes in seasonal and diurnal rainfall and temperature patterns (as influenced by the hydrologically relevant surroundings of a forest stand, e.g., Zierl and Bugmann, 2005 [SRC] ). Recently observed moderate climatic changes have induced forest productivity gains globally (reviewed in Boisvenue and Running, 2006 [JoC, ARC] ) and possibly enhanced carbon sequestration, especially in tropical forests ( (Baker et al., 2004; ) ( Lewis et al., 2004a, ) 2004b; ( Malhi and Phillips, 2004; ) ( Phillips et al., 2004 ) ), where these are not reduced by water limitations (e.g., Boisvenue and Running, 2006 [JoC, ARC] ) or offset by deforestation or novel fire regimes ( Nepstad et al., 1999 [JoC, ARC] , 2004; ( Alencar et al., 2006 ) ) or by hotter and drier summers at mid- and high latitudes ( Angert et al., 2005 [JoC] ).
Potential increases in drought conditions have been quantitatively projected for several regions (e.g., Amazon, Europe) during the critical growing phase, due to increasing summer temperatures and precipitation declines (e.g., Cox et al., 2004 [MoS, ARC] ; Schaphoff et al., 2006 [JoC, MoS, SRC] ; Scholze et al., 2006 [JoC, ARC] ; Figure 4.3 , vegetation change 6). Since all these responses potentially influence forest net ecosystem productivity (NEP), substantive biotic feedbacks may result, either through carbon releases or influences on regional climate, contributing to further major uncertainties (e.g., Betts et al., 2000 [JoC, ARC] ; Peng and Apps, 2000 [MoS] ; Bergengren et al., 2001 [JoC, MoS] ;( Semazzi and Song, 2001; ) Leemans et al., 2002 [SRC] ; Körner, 2003c [JoC, SRC] ; Canadell et al., 2004 [JoC, ARC] ; Cox et al., 2004 [MoS, ARC] ; Gruber et al., 2004 [NPR, ARC] ; Heath et al., 2005 [JoC] ; Section 4.4.1 ). Effects of drought on forests include mortality, a potential reduction in resilience (e.g., Lloret et al., 2004 [JoC] ;( Hogg and Wein, 2005 ) ) and can cause major biotic feedbacks (e.g., Ciais et al., 2005 [JoC, ARC] ; Box 4.1 ). However, these effects remain incompletely understood and vary from site to site (e.g., Reichstein et al., 2002 [JoC] ; Betts et al., 2004 [MoS, ARC] ). For example, drought impacts can be offset by fertile soils ( (Hanson and Weltzin, 2000 ) ), or if due to a heatwave, drought may even be accompanied by enhanced tree growth at cooler high elevation sites due to a longer growing season and enhanced photosynthetic activity ( Jolly et al., 2005 [JoC] ; Box 4.1 ).
Drought conditions further interact with disturbances such as insects ( (Hanson and Weltzin, 2000; ) Fleming et al., 2002 [JoC] ;( Logan et al., 2003; ) ( Schlyter et al., 2006; ) Box 4.1 ) or fire ( (Flannigan et al., 2000 ) ). Tree-defoliating insects, especially in boreal forests, periodically cause substantial damage (e.g., Gitay et al., 2001 [NPR, ARC] , Box 5-10; ( Logan et al., 2003 ) ). Insect pests were found to be at least partly responsible for the decline and ultimate extirpation of stands at the southern margins of the range of their hosts, subjected to warmer and drier conditions ( (Volney and Fleming, 2000; ) see also Section 4.2.2 ). At the poleward ecotone (see Glossary), frosts and general low temperatures appear to limit insect outbreaks ( (Virtanen et al., 1996; ) ( Volney and Fleming, 2000 ) ); thus outbreaks currently constrained from northern ranges could become more frequent in the future ( Carroll et al., 2004 [NPR] ). If climate warms and this ecotone becomes exposed to more droughts, insect outbreaks will become a major factor ( (Logan et al., 2003; ) ( Gan, 2004 ) ). With A2 and B2 emissions scenarios downscaled to regional level in northern Europe, projected climate extremes by 2070 - 2100 will increase the susceptibility of Norway spruce to secondary damage through pests and pathogens, matched by an accelerated life cycle of spruce bark beetle populations ( (Schlyter et al., 2006 ) ).
Climate change is known to alter the likelihood of increased wildfire sizes and frequencies (e.g., Stocks et al., 1998 [JoC, ARC] ;( Podur et al., 2002; ) Brown et al., 2004 [JoC] ; Gillett et al., 2004 [JoC] ), while also inducing stress on trees that indirectly exacerbate disturbances ( Dale et al., 2000 [SRC] ; Fleming et al., 2002 [JoC] ;( Schlyter et al., 2006 ) ). This suggests an increasing likelihood of more prevalent fire disturbances, as has recently been observed ( Gillett et al., 2004 [JoC] ; van der Werf et al., 2004 [JoC] ; Westerling et al., 2006 [JoC, ARC] ; Section 4.2.2 ).
Considerable progress has been made since the TAR in understanding fire regimes and related processes ( Kasischke and Stocks, 2000 [NPR] ;( Skinner et al., 2002; ) ( Stocks et al., 2002; ) Hicke et al., 2003 [JoC] ;( Podur et al., 2003; ) Gillett et al., 2004 [JoC] ) enabling improved projections of future fire regimes ( (Flannigan et al., 2000; ) Li et al., 2000 [ARC] ; de Groot et al., 2003 [MoS] ; Brown et al., 2004 [JoC] ; Fried et al., 2004 [JoC, MoS, ARC] ). Some argue (e.g., Harden et al., 2000 [JoC] ) that the role of fire regimes in the boreal region has previously been underestimated. About 10% of the 2002 / 2003 global carbon emission anomaly can be ascribed to Siberian fires by inverse modelling ( van der Werf et al., 2004 [JoC] ), as supported by remote sensing ( Balzter et al., 2005 [JoC, ARC] ). Climate changes including El Niño events alter fire regimes in fire-prone regions such as Australia ( Hughes, 2003 [MoS, SRC] ;( Williams et al., 2004b; ) Allen Consulting Group, 2005 ), the Mediterranean region (e.g., Mouillot et al., 2002 [JoC, MoS] ; see also Section 4.4.4 ), Indonesia and Alaska ( (Hess et al., 2001 ) ), but also introduce fire into regions where it was previously absent (e.g., Schumacher et al., 2006 [Ambiguous] ). Intensified fire regimes are likely to impact boreal forests at least as much as climate change itself ( (Flannigan et al., 2000 ) ), and may accelerate transitions, e.g., between taiga and tundra, through facilitating the invasion of pioneering trees and shrubs into tundra ( (Landhäusser and Wein, 1993; ) ( Johnstone and Chapin, 2006 ) ).
Will forest expansions be realised as suggested by DGVMs ( Figure 4.3 )? Vegetation models project that forest might eventually replace between 11 and 50% of tundra with a doubling of atmospheric CO2 ( White et al., 2000b [JoC, MoS] ;( Harding et al., 2002; ) Kaplan et al., 2003 [MoS, SRC] ; Callaghan et al., 2005 [NPR, SRC] ; Figure 4.3 , vegetation change 1). However, such transitions are likely to be moderated in reality by many processes not yet considered in the models (e.g., ( Gamache and Payette, 2005; ) see below). Other studies using a wide range of GCMs and forcing scenarios indicate that forests globally face the risk of major change (non-forested to forested and vice-versa within at least 10% of non-cultivated land area) in more than 40% of simulated scenarios if global mean warming remains below 2°C relative to pre-industrial, and in almost 90% of simulated scenarios if global mean warming exceeds 3°C over pre-industrial ( Scholze et al., 2006 [JoC, ARC] ). Those risks have been estimated as especially high for the boreal zone (44% and 88%, respectively) whereas they were estimated as smaller for tropical forests in Latin America (19% and 38%, respectively; see also Figure 4.3 ).
One key process controlling such shifts is migration (e.g., ( Higgins and Harte, 2006 ) ). Estimates for migration rates of tree species from palaeoecological records are on average 200-300 m/yr, which is a rate significantly below that required in response to anticipated future climate change (³1 km/yr, Gitay et al., 2001 [NPR, ARC] , Box 5-2). However, considerable uncertainties remain:
- although not completely quantified, many species can achieve rapid large-scale migrations (Reid’s paradox (see Glossary), e.g., Clark, 1998), but estimates at the low extreme imply a considerable range of lagged responses (Clark et al., 2001; e.g., lag 0-20 years, Tinner and Lotter, 2001; lag several millennia, Johnstone and Chapin, 2003);
- recent genetic analysis (<100 m/yr, McLachlan et al., 2005) indicates that commonly inferred estimates from pollen have overestimated dispersal rates, explaining observed pollen records by multi-front recolonisation from low-density refugees (Pearson, 2006);
- future landscapes will differ substantially from past climate change situations and landscape fragmentation creates major obstacles to migration (e.g., Collingham and Huntley, 2000);
- processes moderating migration such as competition, herbivory and soil formation (land use – Vlassova, 2002; paludification – Crawford et al., 2003; herbivory – Cairns and Moen, 2004; Juday, 2005; pathogens – Moorcroft et al., 2006; Section 4.4.6 );
- tree species do not only respond to a changing climate by migration, but also by local adaptation, including genetic adaptation (Davis and Shaw, 2001; Davis et al., 2005).
Modelling studies reconstructing past (e.g., Lischke et al., 2002 [MoS, SRC] ) or projecting future ( Malcolm et al., 2002b [MoS, SRC] ;( Iverson et al., 2004; ) Neilson et al., 2005 [MoS, SRC] ) dispersal all indicate that more realistic migration rates will result in lagged northward shifts of taiga (lag length 150-250 years, Chapin and Starfield, 1997 [JoC] ; Skre et al., 2002 [SRC] ). While shrubs and the tree line (see Glossary) were found to have advanced polewards in response to recent warming ( Sturm et al., 2001 [JoC] ; Lloyd, 2005 [MoS] ; Tape et al., 2006 [JoC] ; Chapter 1 ), the expected slow encroachment of taiga into tundra is confirmed by satellite data showing no expansion of boreal forest stands ( (Masek, 2001 ) ) indicating century-long time-lags for the forest limit (see Glossary) to move northward ( Lloyd, 2005 [MoS] ). All these findings suggest considerable uncertainties in how fast forests will shift northwards (e.g., Clark et al., 2003 [MoS] ; Higgins et al., 2003 [MoS] ; Chapin et al., 2004 [SRC] ;( Jasinski and Payette, 2005; ) McGuire et al., 2007 [NPR, SRC, 2007] ) and in the resulting consequences for the climate system (discussed in Section 4.4.6 ). Lower rates for the majority of species are probably realistic, also because future conditions comprise both unprecedented climate characteristics, including rapid rates of change (Sections 4.2.1 and 4.4.11 ), and a combination of impediments to local adaptation and migration (with the exception of some generalists).
Compared to the TAR ( Gitay et al., 2001 [NPR, ARC] ), the net global loss due to land-use change in forest cover appears to have slowed further ( Stokstad, 2001 [JoC] ; FAO, 2001 [NPR] ), but in some tropical and sub-tropical regions, notably South-East Asia and similarly the Amazon (e.g., Nepstad et al., 1999 [JoC, ARC] ), deforestation rates are still high (0.01-2.01%/yr, Lepers et al., 2005 [ARC] ; Alcamo et al., 2006 [NPR, SRC] ), while in some northern regions such as Siberia, degradation rates are increasing largely due to unsustainable logging ( Lepers et al., 2005 [ARC] ). Though uncertainties in rate estimates are considerable (e.g., FAO, 2001 [NPR] ; Houghton, 2003b [JoC, MoS] ; Lepers et al., 2005 [ARC] ), current trends in pressures ( Nelson, 2005 [NPR] ) will clearly lead to continued deforestation and degradation in critical areas (historically accumulated loss of 182-199 PgC – Canadell et al., 2004 [JoC, ARC] ; expected releases in the 21st century of 40-100 PgC – Gruber et al., 2004 [NPR, ARC] ; Shvidenko et al., 2005 [NPR, ARC] ) with concomitant implications for biodiversity ( Duraiappah et al., 2005 [NPR] ) and other supporting services ( Hassan et al., 2005 [NPR] ). In most industrialised countries, forest areas are expected to increase (e.g., European forests by 2080 up to 6% for the SRES B2 scenario – Karjalainen et al., 2002 [MoS] ; Sitch et al., 2005 [MoS, ARC] ) partly due to intensified agricultural management and climate change.
Although land-use changes may dominate impacts in some areas, climate change generally exacerbates biodiversity risks, especially in biodiversity hotspots and particularly for the first half of the 21st century (montane cloud forests – ( Foster, 2001; ) Hawaii – Benning et al., 2002 [JoC, MoS] ; Costa Rica – Enquist, 2002 [MoS] ; Amazonia – Miles, 2002 [NPR] ; Australia – ( Williams et al., 2003 ) ). In tropical montane cloud forests, extinctions of amphibian species have been attributed to recent climate change ( Pounds et al., 2006 [JoC] ; see Section 4.4.7 and Table 4.1 , No. 2). In a few exceptions, climate change may increase diversity locally or regionally ( (Kienast et al., 1998 ) ) but in most cases extinction risks are projected to increase.
4.4.6 Tundra and Arctic/Antarctic ecosystems
Properties, goods and services
Tundra denotes vegetation and ecosystems north of the closed boreal forest tree line, covering an area of about 5.6 million km2, but here we also include ecosystems at circumpolar latitudes, notably the sea-ice biome in both hemispheres (e.g., ( Arrigo and Thomas, 2004; ) Section 4.4.9 ), and sub-Antarctic islands (but see also Chapter 15 ). Ecosystem services include carbon sequestration, climate regulation, biodiversity and cultural maintenance, fuel, and food and fibre production ( Chapin et al., 2005a [NPR, SRC] , p. 721-728). Climate regulation is likely to be dominated by positive feedbacks between climate and albedo changes through diminishing snow cover and, eventually, expanding forests ( Chapin et al., 2005b [JoC] ) and net emissions of greenhouse gasses, notably methane. The Arctic significantly contributes to global biodiversity ( Chapin et al., 2005a [NPR, SRC] ; Usher et al., 2005 [NPR, SRC] ). Local mixed economies of cash and subsistence depend strongly on the harvest of local resources, food preparation, storage, distribution and consumption. This forms a unique body of cultural knowledge traditionally transmitted from generation to generation ( Hassol, 2004a [NPR] ).
Key vulnerabilities
Arctic and sub-Arctic ecosystems (particularly ombrotrophic bog communities, see Glossary) above permafrost were considered likely to be most vulnerable to climatic changes, since impacts may turn Arctic regions from a net carbon sink to a net source ( Gitay et al., 2001 [NPR, ARC] ). Literature since the TAR suggests that changes in albedo and an increased release of methane from carbon stocks (e.g., Christensen et al., 2004 [JoC] ), whose magnitudes were previously substantially underestimated, will lead to positive radiative climate forcing throughout the Arctic region ( Camill, 2005 [JoC] ; Lelieveld, 2006 [JoC] ; Walter et al., 2006 [JoC] ; Zimov et al., 2006 [JoC] ). Adverse impacts, including pollution (see also Chapter 15 ), were projected for species such as marine birds, seals, polar bears, tundra birds and tundra ungulates ( Gitay et al., 2001 [NPR, ARC] ). Unique endemic biodiversity (e.g., polar bears, Box 4.3 ) as well as tundra-dependent species such as migratory birds (e.g., waterfowl, Box 4.5 , 4.4.8 , Table 4.1 ) have been confirmed to be facing increasing extinction risks, with concomitant threats to the livelihoods and food security for indigenous peoples.
Box 4.3. Polar bears – a species in peril?
There are an estimated 20,000 to 25,000 polar bears (Ursus maritimus) worldwide, mostly inhabiting the annual sea ice over the continental shelves and inter-island archipelagos of the circumpolar Arctic, where they may wander for thousands of kilometres per year. They are specialised predators on ice-breeding seals and are therefore dependent on sea ice for survival. Female bears require nourishment after emerging in spring from a 5 to 7 month fast in nursing dens ( (Ramsay and Stirling, 1988 ) ), and are thus very dependent on close proximity between land and sea ice before it breaks up. Continuous access to sea ice allows bears to hunt throughout the year, but in areas where the sea ice melts completely each summer, they are forced to spend several months in tundra fasting on stored fat reserves until freeze-up.
Polar bears face great challenges from the effects of climatic warming ( (Stirling and Derocher, 1993; ) Stirling et al., 1999 [SRC] ; Derocher et al., 2004 [SRC] ), as projected reductions in sea ice will drastically shrink marine habitat for polar bears, ice-inhabiting seals and other animals ( Hassol, 2004b [NPR] ). Break-up of the sea ice on the western Hudson Bay, Canada, already occurs about 3 weeks earlier than in the early 1970 s, resulting in polar bears in this area coming ashore earlier with reduced fat reserves (a 15% decline in body condition), fasting for longer periods of time and having reduced productivity ( Stirling et al., 1999 [SRC] ). Preliminary estimates suggest that the Western Hudson Bay population has declined from 1,200 bears in 1987 to fewer than 950 in 2004 . Although these changes are specific to one sub-population, similar impacts on other sub-populations of polar bears can be reasonably expected. In 2005, the IUCN Polar Bear Specialist Group concluded that the IUCN Red List classification of the polar bear should be upgraded from Least Concern to Vulnerable based on the likelihood of an overall decline in the size of the total population of more than 30% within the next 35 to 50 years. The U.S. Fish and Wildlife Service is also considering a petition to list the polar bear as a threatened species based in part on future risks to the species from climate change. If sea ice declines according to some projections ( Meehl et al., 2007 [NPR, ARC, 2007] , Figure 10.13; Figure 4.4 , Table 4.1 ) polar bears will face a high risk of extinction with warming of 2.8°C above pre-industrial (range 2.5-3.0°C, Table 4.1 , No. 42). Similar consequences are facing other ice-dependent species, not only in the Arctic but also in the Antarctic ( Chapter 1 Barbraud and Weimerskirch, 2001 [JoC] ; Croxall et al., 2002 [JoC] ).
Impacts
Global warming is projected to be most pronounced at high latitudes ( Phoenix and Lee, 2004 [MoS] ; Meehl et al., 2007 [NPR, ARC, 2007] ; Christensen et al., 2007 [NPR, ARC, 2007] ). Ongoing rapid climatic changes will force tundra polewards at unprecedented rates ( Velichko, 2002 [Ambiguous] ), causing lagged responses in its slow-growing plant communities ( (Camill and Clark, 2000; ) Chapin et al., 2000 [JoC] ; Callaghan et al., 2004a [SRC] , 2004c; Velichko et al., 2004 [SRC] ). Movements of some species of habitat-creating plants (edificators) require large spread rates exceeding their migrational capacity ( Callaghan et al., 2005 [NPR, SRC] ). Poleward taiga encroachment into tundra is also likely to lag these changes (see Section 4.4.5 and e.g., Callaghan et al., 2004b [SRC] ). Projections of vegetation changes in the northern Arctic suggest that by about 2080, 17.6% (range 14-23%) replacement of the current polar desert by tundra vegetation will have begun ( Callaghan et al., 2005 [NPR, SRC] ). An eventual replacement of dwarf shrub tundra by shrub tundra is projected for the Canadian Arctic by 2100 ( Kaplan et al., 2003 [MoS, SRC] ). Experimental manipulations of air temperature at eleven locations across the tundra also show that tundra plant communities change substantially through shifts in species dominance, canopy height and diversity ( Walker et al., 2006 [JoC, SRC] ), with cryptogams being particularly vulnerable ( Cornelissen et al., 2001 [SRC] ; van Wijk et al., 2004 [JoC, SRC] ). A warming of 1-3°C caused a short-term diversity decrease, but generalisations are unwarranted because of insufficiently long experimentation time ( (Graglia et al., 2001; ) ( Dormann and Woodin, 2002; ) van Wijk et al., 2004 [JoC, SRC] ; Walker et al., 2006 [JoC, SRC] ).
The thermally stable oceanic climate of the sub-Antarctic Marion Island appears to be changing, with a rise in annual mean surface air temperature of 1.2°C between 1969 and 1999 . Annual precipitation decreased more or less simultaneously, and the 1990 s was the driest in the island’s five decades with records ( Smith, 2002 [JoC] ). These changes may be linked to a shift in phase of the semi-annual oscillation in the Southern Hemisphere after about 1980 ( (Rouault et al., 2005 ) ). Climatic change will directly affect the indigenous biota of sub-Antarctic islands ( Smith, 2002 [JoC] ; Barnes et al., 2006 [MoS] ). Experimental drying of the keystone cushion plant species Azorella selago on Marion Island revealed measurable negative impacts after only a few months ( Le Roux et al., 2005 [Ambiguous] ).
While summer food availability may increase for some vertebrates ( Hinzman et al., 2005 [JoC, ARC] ), formation of ice-crust at critical winter times may reduce abundance of food below snow ( Yoccoz and Ims, 1999 [NPR] ; Aanes et al., 2002 [MoS] ; Inkley et al., 2004 [NPR, SRC] ). Tundra wetland habitat for migrant birds may dry progressively ( Hinzman et al., 2005 [JoC, ARC] ; Smith et al., 2005 [JoC] ). Many species of Arctic-breeding shorebirds and waterfowl are projected to undergo major population declines as tundra habitat shrinks ( Box 4.5 , Table 4.1 ). In contrast, northern range expansions of more southern species are expected, e.g., moose and red fox ( Callaghan et al., 2005 [NPR, SRC] ). Some colonisers might ultimately need to be considered ‘invasive’ species (e.g., North American Mink – Neuvonen, 2004 [NPR] ), such as presently-restricted populations of southern shrub species that are likely to spread in a warmer climate ( Forbes, 1995 [NPR] ) leading to possibly increased carbon sequestration ( Sturm et al., 2001 [JoC] ; Tape et al., 2006 [JoC] ; for a discussion of overall consequences for climate, see end of Section 4.4.6 ). For arctic species such as the polar bear, increasing risks of extinction are associated with the projected large decrease in the extent of the sea-ice biome and sea-ice cover ( Box 4.3 ).
Significant changes in tundra are of two main types ( Velichko et al., 2005 [SRC] ), namely in vegetation structure (and related albedo), and in below-ground processes related to a combined increase in temperature, increase in depth of the active layer (see Glossary), and moisture content. These will promote paludification (see Glossary; ( Crawford et al., 2003 ) ), thermokarst processes (see Glossary), and increase the dryness of raised areas. Moisture supply substantially influences the state of permafrost, one of the most important components of the tundra landscape ( Anisimov et al., 2002a [MoS, ARC] , 2002b ). Increasing active layer instability causes greater mixing and shifting of the soil’s mineral matrix, damaging plant roots. Generally this will favour moisture-loving species (e.g., sedges), while the peat-bog vegetation over permafrost could experience drier conditions ( Camill, 2005 [JoC] ).
Substantial recent upward revisions ( Zimov et al., 2006 [JoC] ) of carbon stocks ( Figure 4.1 ) in permafrost and yedoma (see Glossary), and measurements of methane releases from north Siberian thaw lakes ( Walter et al., 2006 [JoC] ), Scandinavian mires ( Christensen et al., 2004 [JoC] ) and Canadian permafrost ( Camill, 2005 [JoC] ) now show tundra to be a significantly larger atmospheric methane source than previously recognised. Current estimates of northern wetland methane emissions increase by 10-63% based on northern Siberian estimates alone. This methane source comprises a positive feedback to climate change, as thaw lakes ( Walter et al., 2006 [JoC] ) and mires ( Christensen et al., 2004 [JoC] ) are expanding in response to warming. While thermokarst-derived emissions are currently modest relative to anthropogenic sources, a potential stock of about 500 Pg of labile carbon in yedoma permafrost ( Figure 4.1 ) could greatly intensify the positive feedback to high-latitude warming trends that are currently projected ( Sazonova et al., 2004 [ARC] ; Mack et al., 2004 [JoC] ; Lelieveld, 2006 [JoC] ; Zimov et al., 2006 [JoC] ).
Changes in albedo associated with snow cover loss, and eventual invasion of tundra vegetation by evergreen coniferous trees, is likely to decrease regional albedo significantly and lead to a warming effect greater than the cooling projected from the increased carbon uptake by advancing trees ( Section 4.4.5 ) and shrubs ( Betts, 2000 [JoC, ARC] ; Sturm et al., 2001 [JoC] , 2005; Chapin et al., 2005b [JoC] ; McGuire and Chapin, 2006 [NPR] ; McGuire et al., 2007 [NPR, SRC, 2007] ). Remote sensing already shows that tundra has greened over the past 20 years ( Sitch et al., 2007 [MoS, 2007] ). However, the potential for CO2 sequestration varies from region to region ( Callaghan et al., 2005 [NPR, SRC] ) and model uncertainties are high ( Sitch et al., 2007 [MoS, 2007] ), since migration rates ( Section 4.4.5 ), changes in hydrology, fire, insect pest outbreaks and human impacts relevant to the carbon cycle are poorly represented (see also Sections 4.4.1 and 4.4.5 ).
4.4.7 Mountains
Properties, goods and services
Mountain regions (circa 20-24% of all land, scattered throughout the globe) exhibit many climate types corresponding to widely separated latitudinal belts within short horizontal distances. Consequently, although species richness decreases with elevation, mountain regions support many different ecosystems and have among the highest species richness globally (e.g., Väre et al., 2003 [NPR] ;( Moser et al., 2005; ) Spehn and Körner, 2005 [NPR, SRC] ). Mountain ecosystems have a significant role in biospheric carbon storage and carbon sequestration, particularly in semi-arid and arid areas (e.g., the western U.S., – Schimel et al., 2002 [Ambiguous] ; Tibetan plateau – Piao et al., 2006 [JoC] ). Mountain ecosystem services such as water purification and climate regulation extend beyond their geographical boundaries and affect all continental mainlands (e.g., ( Woodwell, 2004 ) ). Local key services allow habitability of mountain areas, e.g. through slope stabilisation and protection from natural disasters such as avalanches and rockfall. Mountains increasingly serve as refuges from direct human impacts for many endemic species. They provide many goods for subsistence livelihoods, are home to many indigenous peoples, and are attractive for recreational activities and tourism. Critically, mountains harbour a significant fraction of biospheric carbon (28% of forests are in mountains).
Key vulnerabilities
The TAR identified mountain regions as having experienced above-average warming in the 20th century, a trend likely to continue ( Beniston et al., 1997 [JoC, SRC] ; Liu and Chen, 2000 [JoC] ). Related impacts included an earlier and shortened snow-melt period, with rapid water release and downstream floods which, in combination with reduced glacier extent, could cause water shortage during the growing season. The TAR suggested that these impacts may be exacerbated by ecosystem degradation pressures such as land-use changes, over-grazing, trampling, pollution, vegetation destabilisation and soil losses, in particular in highly diverse regions such as the Caucasus and Himalayas ( Gitay et al., 2001 [NPR, ARC] ). While adaptive capacities were generally considered limited, high vulnerability was attributed to the many highly endemic alpine biota ( Pauli et al., 2003 [NPR] ). Since the TAR, the literature has confirmed a disproportionately high risk of extinction for many endemic species in various mountain ecosystems, such as tropical montane cloud forests or forests in other tropical regions on several continents ( (Williams et al., 2003; ) Pounds and Puschendorf, 2004 [JoC, MoS] ;( Andreone et al., 2005; ) Pounds et al., 2006 [JoC] ), and globally where habitat loss due to warming threatens endemic species ( Pauli et al., 2003 [NPR] ; Thuiller et al., 2005b [JoC, SRC] ).
Impacts
Because temperature decreases with altitude by 5-10°C/km, relatively short-distanced upward migration is required for persistence (e.g., MacArthur, 1972 [NPR] ; Beniston, 2000 [NPR, SRC] ; Theurillat and Guisan, 2001 [JoC, SRC] ). However, this is only possible for the warmer climatic and ecological zones below mountain peaks ( Gitay et al., 2001 [NPR, ARC] ; Peñuelas and Boada, 2003 [JoC] ). Mountain ridges, by contrast, represent considerable obstacles to dispersal for many species which tends to constrain movements to slope upward migration (e.g., ( Foster, 2001; ) Lischke et al., 2002 [MoS, SRC] ; Neilson et al., 2005 [MoS, SRC] ; Pounds et al., 2006 [JoC] ). The latter necessarily reduces a species’ geographical range (mountain tops are smaller than their bases). This is expected to reduce genetic diversity within species and to increase the risk of stochastic extinction due to ancillary stresses ( (Peters and Darling, 1985; ) Gottfried et al., 1999 [MoS] ), a hypothesis confirmed by recent genetic analysis showing gene drift effects from past climate changes (e.g., ( Alsos et al., 2005; ) ( Bonin et al., 2006 ) ). A reshuffling of species on altitude gradients is to be expected as a consequence of individualistic species responses that are mediated by varying longevities and survival rates. These in turn are the result of a high degree of evolutionary specialisation to harsh mountain climates (e.g., Theurillat et al., 1998 [NPR, SRC] ; Gottfried et al., 1999 [MoS] ; Theurillat and Guisan, 2001 [JoC, SRC] ;( Dullinger et al., 2005; ) ( Klanderud, 2005; ) Klanderud and Totland, 2005 [MoS] ;( Huelber et al., 2006 ) ), and in some cases they include effects induced by invading alien species (e.g., ( Dukes and Mooney, 1999; ) ( Mack et al., 2000 ) ). Genetic evidence for Fagus sylvatica, e.g., suggests that populations may show some capacity for an in situ adaptive response to climate change ( Jump et al., 2006 [NPR] ). However, ongoing distributional changes ( Peñuelas and Boada, 2003 [JoC] ) show that this response will not necessarily allow this species to persist throughout its range.
Upper tree lines, which represent the interface between sub-alpine forests and low-stature alpine meadows, have long been thought to be partly controlled by carbon balance ( (Stevens and Fox, 1991 ) ). This hypothesis has been challenged ( Hoch and Körner, 2003 [SRC] ; Körner, 2003a [NPR, SRC] ). Worldwide, cold tree lines appear to be characterised by seasonal mean air temperatures of circa 6°C ( Körner, 1998 [NPR, SRC] ; Körner, 2003a [NPR, SRC] ;( Grace et al., 2002; ) Körner and Paulsen, 2004 [SRC] ;( Millar et al., 2004; ) ( Lara et al., 2005; ) ( Zha et al., 2005 ) ). In many mountains, the upper tree line is located below its potential climatic position because of grazing, or disturbances such as wind or fire. In other regions such as the Himalaya, deforestation of past decades has transformed much of the environment and has led to fragmented ecosystems ( Becker and Bugmann, 2001 [NPR, SRC] ). Although temperature control may be a dominant determinant of geographical range, tree species may be unable to migrate and keep pace with changing temperature zones ( (Shiyatov, 2003; ) Dullinger et al., 2004 [MoS] ; Wilmking et al., 2004 [JoC] ).
Where warmer and drier conditions are projected, mountain vegetation is expected to be subject to increased evapotranspiration ( Ogaya et al., 2003 [Ambiguous] ; Jasper et al., 2004 [SRC] ;( Rebetez and Dobbertin, 2004; ) ( Stampfli and Zeiter, 2004; ) Jolly et al., 2005 [JoC] ; Zierl and Bugmann, 2005 [SRC] ;( Pederson et al., 2006 ) ). This leads to increased drought, which has been projected to induce forest dieback in continental climates, particularly in the interior of mountain ranges (e.g., Fischlin and Gyalistras, 1997 [SRC] ; Lischke et al., 1998 [NPR, SRC] ;( Lexer et al., 2000; ) Bugmann et al., 2005 [NPR, SRC] ), and mediterranean areas. Even in humid tropical regions, plants and animals have been shown to be sensitive to water stress on mountains (e.g., Borneo – ( Kitayama, 1996; ) Costa Rica – Still et al., 1999 [PoC, JoC, MoS, ARC] ). There is very high confidence that warming is a driver of amphibian mass extinctions at many highland localities, by creating increasingly favourable conditions for the pathogenic Batrachochytrium fungus ( Pounds et al., 2006 [JoC] ).
The duration and depth of snow cover, often correlated with mean temperature and precipitation ( Keller et al., 2005 [JoC, SRC] ; Monson et al., 2006 [JoC] ), is a key factor in many alpine ecosystems ( Körner, 2003c [JoC, SRC] ;( Daimaru and Taoda, 2004 ) ). A lack of snow cover exposes plants and animals to frost and influences water supply in spring ( Keller et al., 2005 [JoC, SRC] ). If animal movements are disrupted by changing snow patterns, as has been found in Colorado ( Inouye et al., 2000 [JoC] ), increased wildlife mortality may result. At higher altitudes, the increased winter precipitation likely to accompany warming leads to greater snowfall, so that earlier arriving altitudinal migrants are confronted with delayed snowmelt ( Inouye et al., 2000 [JoC] ).
Disturbances such as avalanches, rockfall, fire, wind and herbivore damage interact and are strongly dependent on climate (e.g., Peñuelas and Boada, 2003 [JoC] ; Whitlock et al., 2003 [MoS] ; Beniston and Stephenson, 2004 [SRC] ;( Cairns and Moen, 2004; ) Carroll et al., 2004 [NPR] ;( Hodar and Zamora, 2004; ) ( Kajimoto et al., 2004; ) Pierce et al., 2004 [JoC] ;( Schoennagel et al., 2004; ) Schumacher et al., 2004 [MoS, SRC] ). These effects may prevent recruitment and thus limit adaptive migration responses of species, and are exacerbated by human land use and other anthropogenic pressures (e.g., Lawton et al., 2001 [JoC] ;( Dirnböck et al., 2003; ) Huber et al., 2005 [NPR, SRC] ).
Ecotonal (see Glossary) sensitivity to climate change, such as upper tree lines in mountains (e.g., ( Camarero et al., 2000; ) Walther et al., 2001 [NPR] ; Diaz, 2003 [NPR, MoS] ;( Sanz-Elorza et al., 2003 ) ), has shown that populations of several mountain-restricted species are likely to decline (e.g., ( Beever et al., 2003; ) ( Florenzano, 2004 ) ). The most vulnerable ecotone species are those that are genetically poorly adapted to rapid environmental change, reproduce slowly, disperse poorly, and are isolated or highly specialised, because of their high sensitivity to environmental stresses ( McNeely, 1990 [NPR] ). Recent findings for Europe, despite a spatially coarse analysis, indicate that mountain species are disproportionately sensitive to climate change (about 60% species loss – Thuiller et al., 2005b [JoC, SRC] ). Substantial biodiversity losses are likely if human pressures on mountain biota occur in addition to climate change impacts ( Pounds et al., 1999 [JoC] , 2006; Lawton et al., 2001 [JoC] ; Pounds, 2001 [JoC] ;( Halloy and Mark, 2003; ) Peterson, 2003 [JoC] ;( Solorzano et al., 2003; ) Pounds and Puschendorf, 2004 [JoC, MoS] ).
4.4.8 Freshwater wetlands, lakes and rivers
Properties, goods and services
Inland aquatic ecosystems (covering about 10.3 Mkm2) vary greatly in characteristics and global distribution. The majority of natural freshwater lakes are located in the higher latitudes, most artificial lakes occur in mid- and lower latitudes, and many saline lakes occur at altitudes up to 5,000 m, especially in the Himalaya and Tibet. The majority of natural wetlands (peatlands) are in the boreal region but most managed wetlands (rice paddies) are in the tropics and sub-tropics (where peatlands also occur). Global estimates of the area under rivers, lakes and wetlands vary greatly depending upon definition ( Finlayson et al., 2005 [NPR] ). This chapter follows the TAR in considering ‘wetlands’ as distinct from rivers and lakes. Wetlands encompass a most heterogeneous spectrum of habitats following hydrological and nutrient gradients, and all key processes, including goods and services provided, depend on the catchment level hydrology. Inland waters are subject to many pressures from human activities. Aquatic ecosystems provide a wide range of goods and services ( Gitay et al., 2001 [NPR, ARC] ; Finlayson et al., 2005 [NPR] ). Wetlands are often biodiversity ‘hotspots’ ( Reid et al., 2005 [NPR, SRC] ), as well as functioning as filters for pollutants from both point and non-point sources, and being important for carbon sequestration and emissions ( Finlayson et al., 2005 [NPR] ). Rivers transport water and nutrients from the land to the oceans and provide crucial buffering capacity during drought spells especially if fed by mountain springs and glaciers (e.g., European summer 2003; Box 4.1 ; Chapter 12 , Section 12.6.1 ). Closed lakes serve as sediment and carbon sinks ( Cohen, 2003 [NPR] ), providing crucial repositories of information on past climate changes.
Key vulnerabilities
Gitay et al. 2001 [NPR, ARC] ) have described some inland aquatic ecosystems (Arctic, sub-Arctic ombrotrophic bog communities on permafrost, depressional wetlands with small catchments, drained or otherwise converted peatlands) as most vulnerable to climate change, and have indicated the limits to adaptations due to the dependence on water availability controlled by outside factors. More recent results show vulnerability varying by geographical region ( Van Dam et al., 2002 [NPR] ; Stern, 2007 [NPR, 2007] ). This includes significant negative impacts across 25% of Africa by 2100 (SRES B1 emissions scenario, de Wit and Stankiewicz, 2006 [JoC, MoS] ) with both water quality and ecosystem goods and services deteriorating. Since it is generally difficult and costly to control hydrological regimes, the interdependence between catchments across national borders often leaves little scope for adaptation.
Impacts
Climate change impacts on inland aquatic ecosystems will range from the direct effects of the rise in temperature and CO2 concentration to indirect effects through alterations in the hydrology resulting from the changes in the regional or global precipitation regimes and the melting of glaciers and ice cover (e.g., Chapters 1 and 3 Cubasch et al., 2001 [NPR, ARC] ; Lemke et al., 2007 [NPR, ARC, 2007] ; Meehl et al., 2007 [NPR, ARC, 2007] ).
Studies since the TAR have confirmed and strengthened the earlier conclusions that rising temperature will lower water quality in lakes through a fall in hypolimnetic (see Glossary) oxygen concentrations, release of phosphorus (P) from sediments, increased thermal stability, and altered mixing patterns ( (McKee et al., 2003; ) Verburg et al., 2003 [JoC] ; Winder and Schindler, 2004 [JoC] ;( Jankowski et al., 2006 ) ). In northern latitudes, ice cover on lakes and rivers will continue to break up earlier and the ice-free periods to increase ( Chapter 1 ( Weyhenmeyer et al., 2004; ) Duguay et al., 2006 [ARC] ). Higher temperatures will negatively affect micro-organisms and benthic invertebrates ( Kling et al., 2003 [NPR] ) and the distribution of many species of fish ( (Lake et al., 2000; ) Poff et al., 2002 [NPR] ; Kling et al., 2003 [NPR] ); invertebrates, waterfowl and tropical invasive biota are likely to shift polewards ( (Moss et al., 2003; ) ( Zalakevicius and Svazas, 2005 ) ) with some potential extinctions ( Jackson and Mandrak, 2002 [NPR] ; Chu et al., 2005 [ARC] ). Major changes will be likely to occur in the species composition, seasonality and production of planktonic communities (e.g., increases in toxic blue-green algal blooms) and their food web interactions ( (Gerten and Adrian, 2002; ) Kling et al., 2003 [NPR] ; Winder and Schindler, 2004 [JoC] ) with consequent changes in water quality ( (Weyhenmeyer, 2004 ) ). Enhanced UV-B radiation and increased summer precipitation will significantly increase dissolved organic carbon (DOC, see Glossary) concentrations, altering major biogeochemical cycles ( Zepp et al., 2003 [SRC] ; Phoenix and Lee, 2004 [MoS] ; Frey and Smith, 2005 [JoC] ). Studies along an altitudinal gradient in Sweden show that NPP can increase by an order of magnitude for a 6°C air temperature increase ( Karlsson et al., 2005 [JoC] ). However, tropical lakes may respond with a decrease in NPP and a decline in fish yields (e.g., 20% NPP and 30% fish yield reduction in Lake Tanganyika due to warming over the last century – O’Reilly et al., 2003 [JoC, ARC] ). Higher CO2 levels will generally increase NPP in many wetlands, although in bogs and paddy fields it may also stimulate methane flux, thereby negating positive effects ( Ziska et al., 1998 [JoC] ; Schrope et al., 1999 [NPR] ; Freeman et al., 2004 [JoC] ;( Megonigal et al., 2005; ) Zheng et al., 2006 [JoC] ).
Boreal peatlands will be affected most by warming (see also Sections 4.4.5 and 4.4.6 ) and increased winter precipitation as the species composition of both plant and animal communities will change significantly ( (Weltzin et al., 2000, ) 2001, 2003; Berendse et al., 2001 [JoC] ; Keller et al., 2004 [JoC] ; Sections 4.4.5 and 4.4.6 ). Numerous arctic lakes will dry out with a 2-3°C temperature rise ( Smith et al., 2005 [JoC] ; Symon et al., 2005 [NPR] ). The seasonal migration patterns and routes of many wetland species will need to change and some may be threatened with extinction ( Inkley et al., 2004 [NPR, SRC] ; Finlayson et al., 2005 [NPR] ; Reid et al., 2005 [NPR, SRC] ;( Zalakevicius and Svazas, 2005; ) Box 4.5 ).
Small increases in the variability of precipitation regimes will significantly impact wetland plants and animals at different stages of their life cycle ( Keddy, 2000 [NPR] ). In monsoonal regions, increased variability risks diminishing wetland biodiversity and prolonged dry periods promote terrestrialisation of wetlands as witnessed in Keoladeo National Park, India ( Chauhan and Gopal, 2001 [NPR, SRC] ; Gopal and Chauhan, 2001 [NPR, SRC] ). In dryland wetlands, changes in precipitation regimes may cause biodiversity loss ( Bauder, 2005 [MoS] ). Changes in climate and land use will place additional pressures on already-stressed riparian ecosystems along many rivers in the world ( Naiman et al., 2005 [NPR] ). An increase or decrease in freshwater flows will also affect coastal wetlands ( Chapter 6 ) by altering salinity, sediment inputs and nutrient loadings ( Schallenberg et al., 2001 [NPR] ;( Flöder and Burns, 2004 ) ).
4.4.9 Oceans and shallow seas
Properties, goods and services
Oceans cover over 71% of the Earth’s surface area from polar to tropical regions to a mean depth of 4,000 m, comprising about 14 billion km3, are a massive reservoir of inorganic carbon, yet contain only 698-708 Pg organic carbon, 13-23 Pg of which is in living and dead biomass ( Figure 4.1 Denman et al., 2007 [NPR, 2007] , Section 7.3.4.1). Despite low biomass, phytoplankton carries out almost half of global primary production, and is the basis of the marine food web ( Field et al., 1998 [JoC, ARC] ). Substantial biodiversity exists in both pelagic and benthic realms and along coastlines, in a diverse range of ecosystems from highly productive (e.g., upwelling regions) to those with low productivity (e.g., oceanic gyres). Ocean primary productivity depends on sunlight and nutrients supplied from deep waters ( Sarmiento et al., 2004a [JoC] ). Marine ecosystems provide goods and services such as fisheries, provision of energy, recreation and tourism, CO2 sequestration and climate regulation, decomposition of organic matter and regeneration of nutrients and coastal protection – many of which are critical to the functioning of the Earth system ( Chapter 5 Costanza et al., 1997 [JoC] ; McLean et al., 2001 [NPR, ARC] , Sections 6.3.2 , 6.3.4, 6.3.5, 6.4.5 and 6.4.6; Hassan et al., 2005 [NPR] , Table 18.2). Marine biodiversity supports ecosystem function and the services it provides ( Worm et al., 2006 [JoC] ) with over 1 billion people relying on fish as their main animal protein source, especially in developing nations ( Pauly et al., 2005 [NPR] ). Coastal zones, particularly low-lying areas, and the highly valuable local and global socioeconomic services they provide (e.g., agricultural land, human settlements and associated infrastructure and industry, aquaculture and fisheries and freshwater supply) are particularly vulnerable to climate change ( McLean et al., 2001 [NPR, ARC] , Section 6.5 Hassan et al., 2005 [NPR] , Section 19.3.2 , Table 19.2 ).
Key vulnerabilities
Since the TAR, literature has confirmed that salient vulnerable ecosystems are warm-water coral reefs ( Box 4.4 ), cold-water corals, the Southern Ocean and marginal sea-ice ecosystems. Ocean uptake of CO2, resulting from increasing atmospheric CO2 concentrations, reduces surface ocean pH and carbonate ion concentrations, an impact that was overlooked in the TAR. This is expected to affect coral reefs, cold water corals, and ecosystems (e.g., the Southern Ocean), where aragonite (used by many organisms to make their shells or skeletons) will decline or become undersaturated. These and other ecosystems where calcareous organisms (e.g., pteropods, see Glossary) play an important role will become vulnerable this century (reviewed by Raven et al., 2005 [NPR, SRC] ; Haugan et al., 2006 [NPR, SRC] ; Table 4.1 ). Synergistic impacts of higher seawater temperatures and declining carbonate make these ecosystems even more vulnerable (e.g., Raven et al., 2005 [NPR, SRC] ; Turley et al., 2006 [NPR, SRC] ; Box 4.4 ). Marginal sea-ice and surrounding ecosystems are vulnerable to warming, particularly in the Northern Hemisphere ( (Sarmiento et al., 2004b; ) Christensen et al., 2007 [NPR, ARC, 2007] ).
Box 4.4. Coral reefs: endangered by climate change?
Reefs are habitat for about a quarter of marine species and are the most diverse among marine ecosystems ( Roberts et al., 2002 [JoC] ; Buddemeier et al., 2004 [NPR] ). They underpin local shore protection, fisheries, tourism ( Chapter 6 Hoegh-Guldberg et al., 2000 [NPR, ARC] ; Cesar et al., 2003 [NPR] ;( Willig et al., 2003; ) Hoegh-Guldberg, 2004 [ARC] , 2005 ) and, though supplying only about 2-5% of the global fisheries harvest, comprise a critical subsistence protein and income source in the developing world ( Whittingham et al., 2003 [NPR] ; Pauly et al., 2005 [NPR] ;( Sadovy, 2005 ) ).
Corals are affected by warming of surface waters ( Chapter 6 , Box 6.1 Reynaud et al., 2003 [JoC] ; McNeil et al., 2004 [JoC] ; McWilliams et al., 2005 [ARC] ) leading to bleaching (loss of algal symbionts – Chapter 6 , Box 6.1 ). Many studies incontrovertibly link coral bleaching to warmer sea surface temperature (e.g., McWilliams et al., 2005 [ARC] ) and mass bleaching and coral mortality often results beyond key temperature thresholds ( Chapter 6 , Box 6.1 ). Annual or bi-annual exceedance of bleaching thresholds is projected at the majority of reefs worldwide by 2030 to 2050 ( Hoegh-Guldberg, 1999 [MoS, ARC] ; Sheppard, 2003 [JoC, MoS] ; Donner et al., 2005 [JoC, ARC] ). After bleaching, algae quickly colonise dead corals, possibly inhibiting later coral recruitment (e.g., ( McClanahan et al., 2001; ) ( Szmant, 2001; ) Gardner et al., 2003 [JoC, ARC] ;( Jompa and McCook, 2003 ) ). Modelling predicts a phase switch to algal dominance on the Great Barrier Reef and Caribbean reefs in 2030 to 2050 ( Wooldridge et al., 2005 [ARC] ).
Coral reefs will also be affected by rising atmospheric CO2 concentrations ( Orr et al., 2005 [PoC, JoC] ; Raven et al., 2005 [NPR, SRC] ; Denman et al., 2007 [NPR, 2007] , Box 7.3 ) resulting in declining calcification. Experiments at expected aragonite concentrations demonstrated a reduction in coral calcification ( Marubini et al., 2001 [NPR] ;( Langdon et al., 2003; ) ( Hallock, 2005 ) ), coral skeleton weakening ( (Marubini et al., 2003 ) ) and strong temperature dependence ( Reynaud et al., 2003 [JoC] ). Oceanic pH projections decrease at a greater rate and to a lower level than experienced over the past 20 million years ( Caldeira and Wickett, 2003 [JoC] ; Raven et al., 2005 [NPR, SRC] ; Turley et al., 2006 [NPR, SRC] ). Doubling CO2 will reduce calcification in aragonitic corals by 20%-60% ( Kleypas et al., 1999 [JoC] ; Kleypas and Langdon, 2002 [NPR] ; Reynaud et al., 2003 [JoC] ; Raven et al., 2005 [NPR, SRC] ). By 2070 many reefs could reach critical aragonite saturation states ( Feely et al., 2004 [JoC] ; Orr et al., 2005 [PoC, JoC] ), resulting in reduced coral cover and greater erosion of reef frameworks ( Kleypas et al., 2001 [MoS] ; Guinotte et al., 2003 [MoS] ).
Adaptation potential ( Hughes et al., 2003 [MoS, SRC] ) by reef organisms requires further experimental and applied study ( Coles and Brown, 2003 [ARC] ; Hughes et al., 2003 [MoS, SRC] ). Natural adaptive shifts to symbionts with +2°C resistance may delay demise of some reefs to roughly 2100 ( Sheppard, 2003 [JoC, MoS] ), rather than mid-century ( Hoegh-Guldberg, 2005 [ARC] ) although this may vary widely across the globe ( Donner et al., 2005 [JoC, ARC] ). Estimates of warm-water coral cover reduction in the last 20-25 years are 30% or higher ( Wilkinson, 2004 [NPR] ; Hoegh-Guldberg, 2005 [ARC] ) due largely to increasing higher SST frequency ( Hoegh-Guldberg, 1999 [MoS, ARC] ). In some regions, such as the Caribbean, coral losses have been estimated at 80% ( Gardner et al., 2003 [JoC, ARC] ). Coral migration to higher latitudes with more optimal SST is unlikely, due both to latitudinally decreasing aragonite concentrations and projected atmospheric CO2 increases ( Kleypas et al., 2001 [MoS] ; Guinotte et al., 2003 [MoS] ; Orr et al., 2005 [PoC, JoC] ; Raven et al., 2005 [NPR, SRC] ). Coral migration is also limited by lack of available substrate ( Chapter 6 , Section 6.4.1.5 ). Elevated SST and decreasing aragonite have a complex synergy ( Harvell et al., 2002 [JoC] ; Reynaud et al., 2003 [JoC] ; McNeil et al., 2004 [JoC] ; Kleypas et al., 2005 [JoC, ARC] ) but could produce major coral reef changes ( Guinotte et al., 2003 [MoS] ; Hoegh-Guldberg, 2005 [ARC] ). Corals could become rare on tropical and sub-tropical reefs by 2050 due to the combined effects of increasing CO2 and increasing frequency of bleaching events (at 2-3 * CO2 ( Kleypas and Langdon, 2002 [NPR] ; Hoegh-Guldberg, 2005 [ARC] ; Raven et al., 2005 [NPR, SRC] ). Other climate change factors (such as sea-level rise, storm impact and aerosols) and non-climate factors (such as over-fishing, invasion of non-native species, pollution, nutrient and sediment load (although this could also be related to climate changes through changes to precipitation and river flow; Chapter 6 , Box 6.1 ; Chapter 11 , Box 11.3 ; Chapter 16 )) add multiple impacts on coral reefs ( Chapter 16 , Box 16.2 ), increasing their vulnerability and reducing resilience to climate change ( Koop et al., 2001 [ARC] ; Kleypas and Langdon, 2002 [NPR] ; Cole, 2003 [JoC] ; Buddemeier et al., 2004 [NPR] ;( Hallock, 2005 ) ).
Impacts
Climate change can impact marine ecosystems through ocean warming ( Wang et al., 2004b [JoC] ), by increasing thermal stratification and reducing upwelling ( Cox et al., 2000 [JoC, MoS, ARC] ; Sarmiento et al., 2004a [JoC] ), sea level rise ( IPCC, 2001 [NPR] ), and through increases in wave height and frequency ( Monahan et al., 2000 [JoC] ; Wang et al., 2004b [JoC] ), loss of sea ice ( (Sarmiento et al., 2004b; ) Meehl et al., 2007 [NPR, ARC, 2007] ; Christensen et al., 2007 [NPR, ARC, 2007] ), increased risk of diseases in marine biota ( Harvell et al., 2002 [JoC] ) and decreases in the pH and carbonate ion concentration of the surface oceans ( Caldeira and Wickett, 2003 [JoC] ; Feely et al., 2004 [JoC] ; Sabine et al., 2004 [NPR, ARC] ; Raven et al., 2005 [NPR, SRC] ).
Theoretically, nutrient speciation could be influenced by the lower pH expected this century ( Zeebe and Wolf-Gladrow, 2001 [NPR] ; Raven et al., 2005 [NPR, SRC] ). Decreases in both upwelling and formation of deep water and increased stratification of the upper ocean will reduce the input of essential nutrients into the sunlit regions of oceans and reduce productivity ( Cox et al., 2000 [JoC, MoS, ARC] ; Loukos et al., 2003 [MoS] ; Lehodey et al., 2003 [MoS] ; Sarmiento et al., 2004a [JoC] ). In coastal areas and margins, increased thermal stratification may lead to oxygen deficiency, loss of habitats, biodiversity and distribution of species, and impact whole ecosystems ( (Rabalais et al., 2002 ) ). Changes to rainfall and nutrient flux from land may exacerbate these hypoxic events ( (Rabalais et al., 2002 ) ).
Projections of ocean biological response to climate warming by 2050 show contraction of the highly productive marginal sea-ice biome by 42% and 17% in Northern and Southern Hemispheres ( (Sarmiento et al., 2004b; ) see also Meehl et al., 2007 [NPR, ARC, 2007] ; Christensen et al., 2007 [NPR, ARC, 2007] ). The sea-ice biome accounts for a large proportion of primary production in polar waters and supports a substantial food web. As timing of the spring phytoplankton bloom is linked to the sea-ice edge, loss of sea ice ( Walsh and Timlin, 2003 [MoS, ARC] ) and large reductions of the total primary production in the marginal sea-ice biome in the Northern Hemisphere ( Behrenfeld and Falkowski, 1997 [MoS] ; Marra et al., 2003 [NPR] ) would have strong effects, for example, on the productivity of the Bering Sea( (Stabeno et al., 2001 ) ). Reductions in winter sea-ice will affect the reproduction, growth and development of fish, krill, and their predators, including seals and seal-dependent polar bears (e.g., ( Barber and Iacozza, 2004; ) Box 4.3 ), leading to further changes in abundance and distribution of marine species ( Chapter 15 , Section 15.4.3 ). An expansion by 4.0% (Northern Hemisphere) and 9.4% (Southern), and of the sub-polar gyre biome by 16% (Northern) and 7% (Southern), has been projected for the permanently stratified sub-tropical gyre biome with its low productivity. This effect has now been observed in the North Pacific and Atlantic ( (McClain et al., 2004; ) ( Sarmiento et al., 2004b ) ). A contraction by 11% of the seasonally stratified sub-tropical gyre is also projected in both hemispheres by 2050 due to climate warming. These changes are likely to have significant impacts on marine ecosystem productivity globally, with uncertainties in projections of NPP using six mainly IS92a-based scenarios narrowing to an increase of between 0.7% and 8.1% by mid-century (?Tglobal ~1.5-3°C).
Changes to planktonic and benthic community composition and productivity have been observed in the North Sea since 1955 ( (Clark and Frid, 2001 ) ) and since the mid- 1980 s may have reduced the survival of young cod ( Beaugrand et al., 2003 [JoC, SRC] ). Large shifts in pelagic biodiversity ( Beaugrand et al., 2002 [JoC, ARC] ) and in fish community composition have been seen ( (Genner et al., 2004; ) Perry et al., 2005 [JoC] ). Changes in seasonality or recurrence of hydrographic events or productive periods could be affected by trophic links to many marine populations, including exploited or cultured populations ( Stenseth et al., 2002 [JoC] , 2003; Platt et al., 2003 [JoC] ;( Llope et al., 2006 ) ). Elevated temperatures have increased mortality of winter flounder eggs and larvae ( (Keller and Klein-Macphee, 2000 ) ) and have led to later spawning migrations ( (Sims et al., 2004 ) ). A 2°C rise in sea surface temperature (SST) would result in removal of Antarctic bivalves and limpets from the Southern Ocean ( (Peck et al., 2004 ) ). Tuna populations may spread towards presently temperate regions, based on predicted warming of surface water and increasing primary production at mid- and high latitudes ( Loukos et al., 2003 [MoS] ).
Marine mammals, birds, cetaceans and pinnipeds (seals, sea lions and walruses), which feed mainly on plankton, fish and squid, are vulnerable to climate change-driven changes in prey distribution, abundance and community composition in response to climatic factors ( (Learmonth et al., 2006 ) ). Changing water temperature also has an effect on the reproduction of cetaceans and pinnipeds, indirectly through prey abundance, either through extending the time between individual breeding attempts, or by reducing breeding condition of the mother ( (Whitehead, 1997 ) ). Current extreme climatic events provide an indication of potential future effects. For example, the warm-water phase of ENSO is associated with large-scale changes in plankton abundance and associated impacts on food webs ( (Hays et al., 2005 ) ), and changes to behaviour ( (Lusseau et al., 2004 ) ), sex ratio ( (Vergani et al., 2004 ) ) and feeding and diet ( (Piatkowski et al., 2002 ) ) of marine mammals.
Melting Arctic ice-sheets will reduce ocean salinities ( IPCC, 2001 [NPR] ), causing species-specific shifts in the distribution and biomass of major constituents of Arctic food webs, including poleward shifts in communities and the potential loss of some polar species (such as the narwhal, Monodon monoceros). Migratory whales (e.g., grey whale, Eschrichtius robustus), that spend summer in Arctic feeding grounds, are likely to experience disruptions in their food sources ( (Learmonth et al., 2006 ) ). Nesting biology of sea turtles is strongly affected by temperature, both in timing and in the determination of the sex ratio of hatchlings ( Hays et al., 2003 [JoC] ), but implications for population size are unknown. A predicted sea-level rise of 0.5 m will eliminate up to 32% of sea-turtle nesting beaches in the Caribbean ( Fish et al., 2005 [MoS, ARC] ).
Surface ocean pH has decreased by 0.1 unit due to absorption of anthropogenic CO2 emissions (equivalent to a 30% increase in hydrogen ion concentration) and is predicted to decrease by up to a further 0.3-0.4 units by 2100 ( Caldeira and Wickett, 2003 [JoC] ). This may impact a wide range of organisms and ecosystems (e.g., coral reefs, Box 4.4 , reviewed by Raven et al., 2005 [NPR, SRC] ), including juvenile planktonic, as well as adult, forms of benthic calcifying organisms (e.g., echinoderms, gastropods and shellfish), and will affect their recruitment (reviewed by Turley et al., 2006 [NPR, SRC] ). Polar and sub-polar surface waters and the Southern Ocean will be aragonite under-saturated by 2100 ( Orr et al., 2005 [PoC, JoC] ) and Arctic waters will be similarly threatened ( Haugan et al., 2006 [NPR, SRC] ). Organisms using aragonite to make their shells (e.g., pteropods) will be at risk and this will threaten ecosystems such as the Southern and Arctic Oceans in which they play a dominant role in the food web and carbon cycling ( Orr et al., 2005 [PoC, JoC] ; Haugan et al., 2006 [NPR, SRC] ).
Cold-water coral ecosystems exist in almost all the world’s oceans and their aerial coverage could equal or exceed that of warm-water coral reefs ( Freiwald et al., 2004 [NPR] ;( Guinotte et al., 2006 ) ). They harbour a distinct and rich ecosystem, provide habitats and nursery grounds for a variety of species, including commercial fish and numerous new species previously thought to be extinct ( Raven et al., 2005 [NPR, SRC] ). These geologically ancient, long-lived, slow-growing and fragile reefs will suffer reduced calcification rates and, as the aragonite saturation horizon moves towards the ocean surface, large parts of the oceans will cease to support them by 2100 ( Feely et al., 2004 [JoC] ; Orr et al., 2005 [PoC, JoC] ; Raven et al., 2005 [NPR, SRC] ;( Guinotte et al., 2006 ) ). Since cold-water corals do not have symbiotic algae but depend on extracting food particles sinking from surface waters or carried by ocean currents, they are also vulnerable to changes to ocean currents, primary productivity and flux of food particles ( (Guinotte et al., 2006 ) ). Warm-water coral reefs are also sensitive to multiple impacts including increased SST and decreasing aragonite concentrations within this century ( Box 4.4 ).
4.4.10 Cross-biome impacts
This section highlights issues that cut across biomes, such as large-scale geographical shifts of vegetation ( Figure 4.3 ) or animal migration patterns (e.g., Box 4.3 ; Box 4.5 ), and changes in land use and aquatic systems.
Biome shifts
Boreal forest and Arctic tundra ecosystems are projected generally to show increased growth due to longer and warmer growing seasons ( Lucht et al., 2002 [JoC, SRC] ; Figure 4.3 ). Woody boreal vegetation is expected to spread into tundra at higher latitudes and higher elevations ( (Grace et al., 2002; ) Kaplan et al., 2003 [MoS, SRC] ; Gerber et al., 2004 [PoC, JoC, MoS] ). At the southern ecotone (see Glossary) with continental grasslands, a contraction of boreal forest is projected due to increased impacts of drought, insects and fires ( Bachelet et al., 2001 [SRC] ; Scholze et al., 2006 [JoC, ARC] ), together with a lower rate of sapling survival ( (Hogg and Schwarz, 1997 ) ). Drought stress could partially be counteracted by concurrent CO2-induced enhanced water-use efficiency ( Gerten et al., 2005 [JoC, SRC] ), small regional increases in precipitation, and an increased depth of permafrost thawing. It is uncertain whether peak summer heat stress on boreal tree species could cause regional transitions to grassland where the continental winter climate remains too cold for temperate forest species to succeed ( Gerber et al., 2004 [PoC, JoC, MoS] ; Lucht et al., 2006 [SRC] ). In temperate forests, milder winters may reduce winter hardening in trees, increasing their vulnerability to frost ( Hänninen et al., 2001 [NPR] ;( Hänninen, 2006 ) ).
Vegetation change in the lower to mid latitudes is uncertain because transitions between tropical desert and woody vegetation types are difficult to forecast. Climate models disagree in pattern and magnitude of projected changes in atmospheric circulation and climate variability, particularly for precipitation (e.g., with respect to the Indian and West African monsoons). For the Sahel and other semi-arid regions, increasing drought is predicted by some models ( Held et al., 2005 [JoC, MoS] ), while increased water-use efficiency is projected to cause more greening ( Figure 4.3 ), though potentially associated with more frequent fires, in others ( Bachelet et al., 2003 [MoS, SRC] ; Woodward and Lomas, 2004b [MoS] ; Ni et al., 2006 [MoS] ; Schaphoff et al., 2006 [JoC, MoS, SRC] ). In savannas, woody encroachment is projected to be a consequence of enhanced water-use efficiency and increased precipitation in some regions ( Bachelet et al., 2001 [SRC] ; Lucht et al., 2006 [SRC] ; Ni et al., 2006 [MoS] ; Schaphoff et al., 2006 [JoC, MoS, SRC] ; Section 4.4.3 ; Figure 4.3 ). The moderate drying, including desert amelioration, as projected in southern Africa, the Sahel region, central Australia, the Arabian Peninsula and parts of central Asia ( Figure 4.3 ) may be due to a positive impact of rising atmospheric CO2, as noted in eastern Namibia through sensitivity analysis ( Thuiller et al., 2006b [JoC, SRC] ).
A general increase of deciduous at the expense of evergreen vegetation is predicted at all latitudes, although the forests in both the eastern USA and eastern Asia appear to be sensitive to drought stress and decline under some scenarios ( Bachelet et al., 2001 [SRC] ; Gerten et al., 2005 [JoC, SRC] ; Lucht et al., 2006 [SRC] ; Scholze et al., 2006 [JoC, ARC] ). Tropical ecosystems are expected to change, particularly in the Amazon, where a subset of GCMs shows strong to moderate reductions in precipitation with the consequence of transitions of evergreen tropical forest to rain-green forest or grasslands ( Cox et al., 2004 [MoS, ARC] ; Cramer et al., 2004 [SRC] ; Woodward and Lomas, 2004b [MoS] ). However, representations of tropical succession remain underdeveloped in current models. The global land biosphere is projected by some models to lose carbon beyond temperature increases of 3°C ( Gerber et al., 2004 [PoC, JoC, MoS] ), mainly from temperate and boreal soils, with vegetation carbon declining beyond temperature increases above 5°C ( Gerber et al., 2004 [PoC, JoC, MoS] ). Carbon sinks persist mainly in the Arctic and in savanna grasslands ( Woodward and Lomas, 2004b [MoS] ; Schaphoff et al., 2006 [JoC, MoS, SRC] ). However, there is large variability between the projections of different vegetation ( Cramer et al., 2001 [JoC, MoS, SRC] ) and climate ( Schaphoff et al., 2006 [JoC, MoS, SRC] ) models for a given emissions scenario.
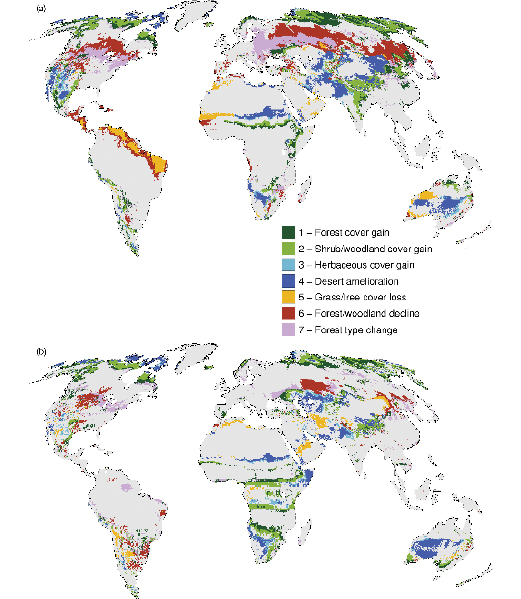
Figure 4.3. Projected appreciable changes in terrestrial ecosystems by 2100 relative to 2000 as simulated by DGVM LPJ ( Sitch et al., 2003 [JoC, MoS, SRC] ; Gerten et al., 2004 [MoS, SRC] ) for two SRES emissions scenarios (Naki´cenovi´c et al., 2000 [NotFound] ) forcing two climate models: (a) HadCM3 A2, (b) ECHAM5 B1 ( Lucht et al., 2006 [SRC] ; Schaphoff et al., 2006 [JoC, MoS, SRC] ). Changes are considered appreciable and are only shown if they exceed 20% of the area of a simulated grid cell (see Figure 4.2 for further explanations).
Migration patterns
Vagile (see Glossary) animals such as polar bears (sea-ice biome, tundra; Box 4.3 ) and in particular migratory animals (tundra, wetlands, lakes, tropical forests, savannas, etc.; Box 4.5 ) respond to impacts both within and across biomes. Many species breed in one area then move to another to spend the non-breeding season ( Robinson et al., 2005 [NPR, ARC] ). Many migratory species may be more vulnerable to climate change than resident species ( Price and Root, 2005 [NPR, SRC] ). As migratory species often move annually in response to seasonal climate changes, their behaviour, including migratory routes, is sensitive to climate. Numerous studies have found that many of these species are arriving earlier ( Chapter 1 and e.g., Root et al., 2003 [PoC, JoC, SRC] ). Changes in the timing of biological events are of particular concern because of a potential disconnect between migrants and their food resources if the phenology of each advances at different rates ( Inouye et al., 2000 [JoC] ; Root et al., 2003 [PoC, JoC, SRC] ;( Visser et al., 2004 ) ). The potential impact of climate change on migratory birds has been especially well studied ( Box 4.5 ).
Box 4.5. Crossing biomes: impacts of climate change on migratory birds
Migratory species can be affected by climate change in their breeding, wintering and/or critical stopover habitats. Models project changes in the future ranges of many species ( Peterson et al., 2002 [JoC, MoS] ; Price and Glick, 2002 [NPR, SRC] ;( Crick, 2004 ) ), some suggesting that the ranges of migrants may shift to a greater extent than non-migrants ( Price and Root, 2001 [SRC] ). In some cases this may lead to a lengthening and in others to a shortening of migration routes. Moreover, changes in wind patterns, especially in relation to seasonal migration timing, could help or hinder migration ( (Butler et al., 1997 ) ). Other expected impacts include continuing changes in phenology, behaviour, population sizes and possibly genetics (reviewed in ( Crick, 2004; ) Robinson et al., 2005 [NPR, ARC] ).
Many migratory species must cross geographical barriers (e.g., the Sahara Desert, oceans) in moving between their wintering and breeding areas. Many species must stop in the Sahel to refuel en route from their breeding to their wintering areas. Degradation of vegetation quality in the Sahel ( Box 4.2 ) could potentially lead to population declines in these species in areas quite remote from the Sahel ( Robinson et al., 2005 [NPR, ARC] ).
More than 80% of the species living within the Arctic Circle winter farther south ( Robinson et al., 2005 [NPR, ARC] ). However, climate-induced habitat change may be greatest in the Arctic ( Zöckler and Lysenko, 2000 [NPR] ; Symon et al., 2005 [NPR] ). For example, the red knot could potentially lose 15%-37% of its tundra breeding habitat by 2100 (HadCM2a1, UKMO). Additionally, at least some populations of this species could also lose critical migratory stopover habitat (Delaware Bay, USA) to sea-level rise ( Galbraith et al., 2002 [ARC] ).
The breeding areas of many Arctic breeding shorebirds and waterfowl are projected to decline by up to 45% and 50%, respectively ( Folkestad et al., 2005 [NPR] ) for global temperature increases of 2°C above pre-industrial. A temperature increase of 2.9°C above pre-industrial would cause larger declines of up to 76% for waterfowl and up to 56% for shorebirds. In North America’s Prairie Pothole region, models have projected an increase in drought with a 3°C regional temperature increase and varying changes in precipitation, leading to large losses of wetlands and to declines in the populations of waterfowl breeding there ( (Johnson et al., 2005 ) ). Many of these species also winter in coastal areas vulnerable to sea-level rise ( Inkley et al., 2004 [NPR, SRC] ). One review of 300 migrant bird species found that 84% face some threat from climate change, almost half because of changes in water regime (lowered watertables and drought), and this was equal to the summed threats due to all other anthropogenic causes ( Robinson et al., 2005 [NPR, ARC] ).
Land use
The relative importance of key drivers on ecosystem change varies across regions and biomes ( Sala et al., 2000 [JoC, SRC] ; Sala, 2005 [NPR] ). Several global studies suggest that at least until 2050 land-use change will be the dominant driver of terrestrial biodiversity loss in human-dominated regions ( Sala et al., 2000 [JoC, SRC] ; UNEP, 2002 [NPR, MoS] ;( Gaston et al., 2003; ) Jenkins, 2003 [JoC] ; Scharlemann et al., 2004 [JoC] ; Sala, 2005 [NPR] ). Conversely, climate change is likely to dominate where human interventions are limited, such as in the tundra, boreal, cool conifer forests, deserts and savanna biomes ( Sala et al., 2000 [JoC, SRC] ; Duraiappah et al., 2005 [NPR] ). Assessment of impacts on biodiversity differ if other drivers than climate change are taken into account ( Thomas et al., 2004a [JoC, SRC] ; Sala, 2005 [NPR] ; Malcolm et al., 2006 [SRC] ). Interactions among these drivers may mitigate or exacerbate the overall effects of climate change ( (Opdam and Wascher, 2004 ) ). The effects of land-use change on species through landscape fragmentation at the regional scale may further exacerbate impacts from climate change ( Holman et al., 2005a [JoC, SRC] ; del Barrio et al., 2006 [MoS] ; Harrison et al., 2006 [MoS] ; Rounsevell et al., 2006 [MoS, SRC] ).
Global land-use change studies project a significant reduction in native vegetation (mostly forest) in non-industrialised countries and arid regions due to expansion of agricultural or urban land use driven principally by population growth, especially in Africa, South America and in South Asia ( Hassan et al., 2005 [NPR] ). This reduction in native habitat will result in biodiversity loss (e.g., Duraiappah et al., 2005 [NPR] ; Section 4.4.11 ). Northern-latitude countries and high-altitude regions may become increasingly important for biodiversity and species conservation as the ranges of species distributions move poleward and upward in response to climate change ( Berry et al., 2006 [SRC] ). Northern-latitude countries and high-altitude regions are also sensitive to the effects of climate change on land use, especially agriculture, which is of particular relevance if those regions are to support adaptation strategies to mitigate the negative effects of future climate and land-use change. Biomes at the highest latitudes that have not already been converted to agriculture are likely to remain relatively unchanged in the future ( Duraiappah et al., 2005 [NPR] ).
Aquatic systems
Higher CO2 concentrations lower the nutritional quality of the terrestrial litter ( (Lindroth et al., 2001; ) Tuchman et al., 2002 [JoC] , 2003a, 2003b ) which in turn will affect the food web relationships of benthic communities in rivers. Greater amounts of DOC (dissolved organic carbon) released in peatlands at higher CO2 levels are exported to streams and finally reach coastal waters ( Freeman et al., 2004 [JoC] ).
4.4.11 Global synthesis including impacts on biodiversity
Considerable progress has been made since the TAR in key fields that allow projection of future climate change impacts on species and ecosystems. Two of these key fields, namely climate envelope modelling (also called niche-based, or bioclimatic modelling) and dynamic global vegetation modelling have provided numerous recent results. The synthesis of these results provides a picture of potential impacts and risks that is far from perfect, in some instances apparently contradictory, but overall highlights a wide array of key vulnerabilities (Figures 4.2 ; 4.4 ; 4.5, Table 4.1 ).
Climate envelope modelling has burgeoned recently due to increased availability of species distribution data, together with finer-scale climate data and new statistical methods that have allowed this correlative method to be widely applied (e.g., Guisan and Thuiller, 2005 [MoS, SRC] ;( McClean et al., 2005; ) Thuiller et al., 2005b [JoC, SRC] ). Despite several limitations ( Section 4.3 and references cited therein) these models offer the advantage of assessing climate change impacts on biodiversity quantitatively (e.g., Thomas et al., 2004a [JoC, SRC] ). Climate envelope models do not simulate dynamic population or migration processes, and results are typically constrained to the regional level, so that the implications for biodiversity at the global level are difficult to infer ( Malcolm et al., 2002a [NPR, ARC] ).
In modelling ecosystem function and plant functional type response, understanding has deepened since the TAR, though consequential uncertainties remain. The ecophysiological processes affected by climate change and the mechanisms by which climate change may impact biomes, ecosystem components such as soils, fire behaviour and vegetation structure (i.e., biomass distribution and leaf area index) are now explicitly modelled and have been bolstered by experimental results (e.g., Woodward and Lomas, 2004b [MoS] ). One emerging key message is that climate change impacts on the fundamental regulating services may previously have been underestimated (Sections 4.4.1 , 4.4.10 , Figures 4.2 ; 4.3 ; 4.4 ). Nevertheless, the globally applicable DGVMs are limited inasmuch as the few plant functional types used within the models aggregate numerous species into single entities ( Sitch et al., 2003 [JoC, MoS, SRC] ). These are assumed to be entities with very broad environmental tolerances, which are immutable and immune to extinction. Therefore, underlying changes in species richness are not accounted for, and the simultaneous free dispersal of PFTs is assumed (e.g., Neilson et al., 2005 [MoS, SRC] ; Midgley et al., 2007 [NPR, SRC, 2007] ). The strength of DGVMs is especially in their global application, realistic dynamics and simulation of ecosystem processes including essential elements of the global C-cycle (e.g., Malcolm et al., 2002b [MoS, SRC] ). Thus, it is reasonable to equate changes in DGVM-simulated vegetation (e.g., Figure 4.3 ) to changes in community and population structures in the real world.
What overall picture emerges from the results reviewed here? It appears that moderate levels of atmospheric CO2 rise and climate change relative to current conditions may be beneficial in some regions ( Nemani et al., 2003 [JoC, ARC] ), depending on latitude, on the CO2 responsiveness of plant functional types, and on the natural adaptive capacity of indigenous biota (mainly through range shifts that are now being widely observed – see Chapter 1 ). But as change continues, greater impacts are projected, while ecosystem and species response may be lagged (Sections 4.4.5 , 4.4.6 ). At key points in time ( Figure 4.4 ), ecosystem services such as carbon sequestration may cease, and even reverse ( Figure 4.2 ). While such ‘tipping points’ ( Kemp, 2005 [JoC] ) are impossible to identify without substantial uncertainties, they may lead to irreversible effects such as biodiversity loss or, at the very least, impacts that have a slow recovery (e.g., on soils and corals).
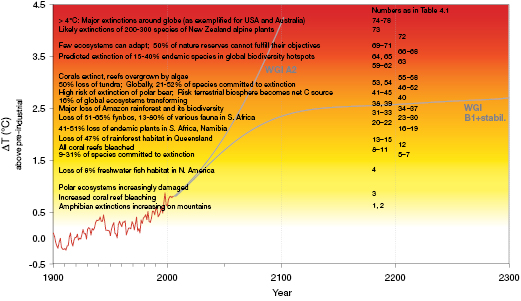
Figure 4.4. Compendium of projected risks due to critical climate change impacts on ecosystems for different levels of global mean annual temperature rise, ΔT, relative to pre-industrial climate (approach and event numbers as used in Table 4.1 and Appendix 4.1 ). It is important to note that these impacts do not take account of ancillary stresses on species due to over-harvesting, habitat destruction, landscape fragmentation, alien species invasions, fire regime change, pollution (such as nitrogen deposition), or for plants the potentially beneficial effects of rising atmospheric CO2. The red curve shows observed temperature anomalies for the period 1900 - 2005 ( Brohan et al., 2006 [MoS] , see also Trenberth et al., 2007 [NPR, PoC, 2007] , Figure 3.6). The two grey curves provide examples of the possible future evolution of temperature against time ( Meehl et al., 2007 [NPR, ARC, 2007] , Figure 10.4), providing examples of higher and lower trajectories for the future evolution of the expected value of ΔT. Shown are the simulated, multi-model mean responses to (i) the A2 emissions scenario and (ii) an extended B1 scenario, where radiative forcing beyond the year 2100 was kept constant to the 2100 value (all data from Meehl et al., 2007 [NPR, ARC, 2007] , Figure 10.4, see also Meehl et al., 2007 [NPR, ARC, 2007] , Section 10.7 ).
In the two simulations presented in Figure 4.2 (warming of 2.9°C and 5.3°C by 2100 over land relative to the 1961 - 1990 baseline), the DGVM approach reveals salient changes in a key regulating service of the world’s ecosystems: carbon sequestration. Changes in the spatial distributions of ecosystems are given in Figure 4.3 (where it must be stressed that the figure highlights only key vulnerabilities through depicting appreciable vegetation type changes, i.e., PFT change over >20% of the area of any single pixel modelled). In the B1 emissions scenario ( Figure 4.3 b) about 26% of extant ecosystems reveal appreciable changes by 2100, with some positive impacts especially in Africa and the Southern Hemisphere. However, these positive changes are likely to be due to the assumed CO2-fertilisation effect ( Section 4.4.10 , Figure 4.3 ). By contrast, in mid- to high latitudes on all continents, substantial shifts in forest structure toward more rain-green, summer-green or deciduous rather than evergreen forest, and forest and woodland decline, underlie the overall drop in global terrestrial carbon sequestration potential that occurs post- 2030, and approaches a net source by about 2070 ( Figure 4.2 ; 4.3). In the A2 emissions scenario, roughly 37% of extant ecosystems reveal appreciable changes by 2100 . Desert amelioration persists in the regions described above, but substantial decline of forest and woodland is seen at northern, tropical and sub-tropical latitudes. In both scenarios the current global sink deteriorates after 2030, and by 2070 (ΔT ~2.5°C over pre-industrial) the terrestrial biosphere becomes an increasing carbon source ( Figure 4.2 ; see also Scholze et al., 2006 [JoC, ARC] ) with the concomitant risk of positive feedback, developments that amplify climate change. Similar results were obtained by using a wide range of climate models which indicate that the biosphere becomes consistently within this century a net CO2 source with a global warming of >3°C relative to pre-industrial ( Scholze et al., 2006 [JoC, ARC] ). On the other hand, it must be noted that by about 2100 the modelled biosphere has nevertheless sequestered an additional 205-228 PgC (A2 and B1 emissions scenarios respectively) relative to the year 2000 ( Lucht et al., 2006 [SRC] ).
Climate envelope modelling suggests that climate change impacts will diminish the areal extent of some ecosystems (e.g., reduction by 2-47% alone due to 1.6°C warming above pre-industrial, Table 4.1 , No. 6) and impact many ecosystem properties and services globally. Climate impacts alone will vary regionally and across biomes and will lead to increasing levels of global biodiversity loss, as expressed through area reductions of wild habitats and declines in the abundance of wild species putting those species at risk of extinction (e.g., 3-16% of European plants with 2.2°C warming ( Table 4.1 , No. 20) or major losses of Amazon rainforest with 2.5°C warming above pre-industrial, Figure 4.4 , Table 4.1 , No. 36). Globally, biodiversity (represented by species richness and relative abundance) may decrease by 13 to 19% due to a combination of land-use change, climate change and nitrogen deposition under four scenarios by 2050 relative to species present in 1970 ( Duraiappah et al., 2005 [NPR] ). Looking at projected losses due to land-use change alone (native habitat loss), habitat reduction in tropical forests and woodland, savanna and warm mixed forest accounts for 80% of the species projected to be lost (about 30,000 species – Sala, 2005 [NPR] ). The apparent contrast between high impacts shown by projections for species (climate envelope models) relative to PFTs (DGVMs) is likely to be due to a number of reasons – most importantly, real species virtually certainly have narrower climate tolerances than PFTs, a fact more realistically represented by the climate envelope models. DGVM projections reveal some increasing success of broad-range, generalist plant species, while climate envelope model results focus on endemics. Endemics, with their smaller ranges, have been shown to have a greater vulnerability to climate change ( Thuiller et al., 2005a [MoS, SRC] ), and may furthermore be dependent on keystone species in relationships that are ignored in DGVMs. Therefore, for assessing extinction risks, climate envelope modelling currently appears to offer more realistic results.
As indicated in the TAR, climate changes are being imposed on ecosystems experiencing other substantial and largely detrimental pressures. Roughly 60% of evaluated ecosystems are currently utilised unsustainably and show increasing signs of degradation ( Reid et al., 2005 [NPR, SRC] ; Hassan et al., 2005 [NPR] ; Worm et al., 2006 [JoC] ). This alone will be likely to cause widespread biodiversity loss ( Chapin et al., 2000 [JoC] ; Jenkins, 2003 [JoC] ; Reid et al., 2005 [NPR, SRC] ), given that 15,589 species, from every major taxonomic group, are already listed as threatened ( Baillie et al., 2006 [NPR] ). The likely synergistic impacts of climate change and land-use change on endemic species have been widely confirmed ( Hannah et al., 2002a [SRC] ; Hughes, 2003 [MoS, SRC] ; Leemans and Eickhout, 2004 [JoC, SRC] ; Thomas et al., 2004a [JoC, SRC] ; Lovejoy and Hannah, 2005 [NPR] ; Hare, 2006 [NPR] ; Malcolm et al., 2006 [SRC] ; Warren, 2006 [NPR, SRC] ), as has over-exploitation of marine systems ( Worm et al., 2006 [JoC] ; Chapters 5 and 6 ).
Overall, climate change has been estimated to be a major driver of biodiversity loss in cool conifer forests, savannas, mediterranean-climate systems, tropical forests, in the Arctic tundra, and in coral reefs ( Thomas et al., 2004a [JoC, SRC] ; Carpenter et al., 2005 [NPR, ARC] ; Malcolm et al., 2006 [SRC] ). In other ecosystems, land-use change may be a stronger driver of biodiversity loss at least in the near term. In an analysis of the SRES scenarios to 2100 ( Strengers et al., 2004 [MoS, SRC] ), deforestation is reported to cease in all scenarios except A2, suggesting that beyond 2050 climate change is very likely to be the major driver for biodiversity loss globally. Due to climate change alone it has been estimated that by 2100 between 1% and 43% of endemic species (average 11.6%) will be committed to extinction (DGVM-based study – Malcolm et al., 2006 [SRC] ), whereas following another approach (also using climate envelope modelling-based studies – Thomas et al., 2004a [JoC, SRC] ) it has been estimated that on average 15% to 37% of species (combination of most optimistic assumptions 9%, most pessimistic 52%) will be committed to extinction by 2050 (i.e., their range sizes will have begun shrinking and fragmenting in a way that guarantees their accelerated extinction). Climate-change-induced extinction rates in tropical biodiversity hotspots are likely to exceed the predicted extinctions from deforestation during this century ( Malcolm et al., 2006 [SRC] ). In the mediterranean-climate region of South Africa, climate change may have at least as significant an impact on endemic Protea species’ extinction risk as land-use change does by 2020 ( Bomhard et al., 2005 [JoC, MoS, SRC] ). Based on all above findings and our compilation ( Figure 4.4 , Table 4.1 ) we estimate that on average 20% to 30% of species assessed are likely to be at increasingly high risk of extinction from climate change impacts possibly within this century as global mean temperatures exceed 2°C to 3°C relative to pre-industrial levels (this chapter). The uncertainties remain large, however, since for about 2°C temperature increase the percentage may be as low as 10% or for about 3°C as high as 40% and, depending on biota, the range is between 1% and 80% ( Table 4.1 Thomas et al., 2004a [JoC, SRC] ; Malcolm et al., 2006 [SRC] ). As global average temperature exceeds 4°C above pre-industrial levels, model projections suggest significant extinctions (40-70% species assessed) around the globe ( Table 4.1 ).
Losses of biodiversity will probably lead to decreases in the provision of ecosystem goods and services with trade-offs between ecosystem services likely to intensify (National Research Council, 1999; Carpenter et al., 2005 [NPR, ARC] ; Duraiappah et al., 2005 [NPR] ). Gains in provisioning services (e.g., food supply, water use) are projected to occur, in part, at the expense of other regulating and supporting services including genetic resources, habitat provision, climate and runoff regulation. Projected changes may also increase the likelihood of ecological surprises that are detrimental for human well-being ( Burkett et al., 2005 [SRC] ; Duraiappah et al., 2005 [NPR] ). Ecological surprises include rapid and abrupt changes in temperature and precipitation, leading to an increase in extreme events such as floods, fires and landslides, increases in eutrophication, invasion by alien species, or rapid and sudden increases in disease ( Carpenter et al., 2005 [NPR, ARC] ). This could also entail sudden shifts of ecosystems to less desired states ( Scheffer et al., 2001 [JoC] ;( Folke et al., 2004; ) e.g., Chapin et al., 2004 [SRC] ) through, for example, the exeedance of critical temperature thresholds, possibly resulting in the irreversible loss of ecosystem services, which were dependent on the previous state ( Reid et al., 2005 [NPR, SRC] ).
Table 4.1. Projected impacts of climate change on ecosystems and population systems as reported in the literature for different levels of global mean annual temperature rise, ΔTg, relative to pre-industrial climate – mean and range (event numbers as used in Figure 4.4 and Appendix 4.1 ). The global temperature change values are used as an indicator of the other associated climate changes that match particular amounts of ΔTg, e.g., precipitation change and, where considered, change in the concentration of greenhouse gases in the atmosphere. Projections from the literature were harmonised into a common framework by down/upscaling (where necessary) from local to global temperature rise using multiple GCMs, and by using a common global mean temperature reference point for the year 1990 (after Warren, 2006 [NPR, SRC] ). Whilst some of the literature relates impacts directly to global mean temperature rises or particular GCM scenarios, many studies give only local temperature rises, ΔTreg, and hence require upscaling. The thirteen GCM output data sets used are taken from the IPCC DDC at http://www.ipcc-data.org/.
| No.i | ΔTg above pre-ind ii | ΔTg above pre-ind iii (range) | ΔTreg above 1990 (range) | Impacts to unique or widespread ecosystems or population systems | Region | Ref. no. |
|---|---|---|---|---|---|---|
| 1 | 0.6 | Increased coral bleaching | Caribbean, Indian Ocean, Great Barrier Reef | 2 | ||
| 2 | 0.6 | Amphibian extinctions/extinction risks on mountains due to climate-change-induced disease outbreaks | Costa Rica, Spain, Australia | 52, 54 | ||
| 3 | <1.0 | Marine ecosystems affected by continued reductions in krill possibly impacting Adelie penguin populations; Arctic ecosystems increasingly damaged | Antarctica, Arctic | 42, 11, 14 | ||
| 4 | 1.3 | 1.1-1.6 | 1 | 8% loss freshwater fish habitat, 15% loss in Rocky Mountains, 9% loss of salmon | N. America | 13 |
| 5 | 1.6 | 1.2-2.0 | 0.7-1.5 | 9-31% (mean 18%) of species committed to extinction | Globe iv | 1 |
| 6 | 1.6 | Bioclimatic envelopes eventually exceeded, leading to 10% transformation of global ecosystems; loss of 47% wooded tundra, 23% cool conifer forest, 21% scrubland, 15% grassland/steppe, 14% savanna, 13% tundra and 12% temperate deciduous forest. Ecosystems variously lose 2-47% areal extent. | Globe | 6 | ||
| 7 | 1.6 | 1.1-2.1 | 1 | Suitable climates for 25% of eucalypts exceeded | Australia | 12 |
| 8 | 1.7 | 1-2.3 | 1°C SST | All coral reefs bleached | Great Barrier Reef, S.E. Asia, Caribbean | 2 |
| 9 | 1.7 | 1.2-2.6 | 38-45% of the plants in the Cerrado committed to extinction | Brazil | 1, 44 | |
| 10 | 1.7 | 1.3-3 | 2-18% of the mammals, 2-8% of the birds and 1-11% of the butterflies committed to extinction | Mexico | 1, 26 | |
| 11 | 1.7 | 1.3-2.4 | 2 | 16% freshwater fish habitat loss, 28% loss in Rocky Mountains, 18% loss of salmon | N. America | 13 |
| 12 | <1.9 | <1.6-2.4 | <1 | Range loss begins for golden bowerbird | Australia | 4 |
| No. | ΔTg above pre-ind | ΔTg above pre-ind (range) | ΔTreg above 1990 (range) | Impacts to unique or widespread ecosystems or population systems | Region | Ref. no. |
|---|---|---|---|---|---|---|
| 13 | 1.9 | 1.6-2.4 | 1 | 7-14% of reptiles, 8-18% of frogs, 7-10% of birds and 10-15% of mammals committed to extinction as 47% of appropriate habitat in Queensland lost | Australia | 1, 7 |
| 14 | 1.9 | 1.6-2.4 | 1 | Range loss of 40-60% for golden bowerbird | Australia | 4 |
| 15 | 1.9 | 1.0-2.8 | Most areas experience 8-20% increase in number ³7day periods with Forest Fire Weather Index >45: increased fire frequency converts forest and maquis to scrub, leads to more pest outbreaks | Mediterranean | 34 | |
| 16 | 2.1 | 41-51% loss in plant endemic species richness | S. Africa, Namibia | 39 | ||
| 17 | 2.1 | 1.0-3.2 | 1-2 | Alpine systems in Alps can tolerate local temperature rise of 1-2°C, tolerance likely to be negated by land-use change | Europe | 8 |
| 18 | 2.1 | 1.4-2.6 | 13-23% of butterflies committed to extinction | Australia | 1, 30 | |
| 19 | 2.1 | 1.4-2.6 | Bioclimatic envelopes of 2-10% plants exceeded, leading to endangerment or extinction; mean species turnover of 48% (spatial range 17-75%); mean species loss of 27% (spatial range 1- 68%) | Europe | 22 | |
| 20 | 2.2 | 3-16% of plants committed to extinction | Europe | 1 | ||
| 21 | 2.2 | 2.1-2.3 | 1.6-1.8 | 15-37% (mean 24%) of species committed to extinction | Globe iv | 1 |
| 22 | 2.2 | 1.7-3.2 | 8-12% of 277 medium/large mammals in 141 national parks critically endangered or extinct; 22-25% endangered | Africa | 23 | |
| 23 | 2.3 | 1.5-2.7 | 2°C SST | Loss of Antarctic bivalves and limpets | Southern Ocean | 51 |
| 24 | 2.3 | 2.0-2.5 | Fish populations decline, wetland ecosystems dry and disappear | Malawi, African Great Lakes | 20 | |
| 25 | 2.3 | 1.5-2.7 | 2.5-3.0 | Extinctions (100% potential range loss) of 10% endemics; 51-65% loss of Fynbos; including 21-40% of Proteaceae committed to extinction; Succulent Karoo area reduced by 80%, threatening 2,800 plant species with extinction; 5 parks lose >40% of plant species | S. Africa | 1, 5, 24, 25 |
| 26 | 2.3 | 2.3-4.0 | 2.5-3.0 | 24-59% of mammals, 28-40% of birds, 13-70% of butterflies, 18-80% of other invertebrates, 21-45% of reptiles committed to extinction; 66% of animal species potentially lost from Kruger National Park | S. Africa | 1, 27 |
| 27 | 2.3 | 2.2-4.0 | 2-20% of mammals, 3-8% of birds and 3-15% of butterflies committed to extinction | Mexico | 1, 26 | |
| 28 | 2.3 | 1.6-3.2 | 48-57% of Cerrado plants committed to extinction | Brazil | 1 | |
| 29 | 2.3 | Changes in ecosystem composition, 32% of plants move from 44% of area with potential extinction of endemics | Europe | 16 | ||
| 30 | 2.3 | 1.6-3.2 | 3 | 24% loss freshwater fish habitat, 40% loss in Rocky Mountains, 27% loss of salmon. | N. America | 13 |
| 31 | 2.4 | 63 of 165 rivers studied lose >10% of their fish species | Globe | 19 | ||
| 32 | 2.4 | Bioclimatic range of 25-57% (full dispersal) or 34-76% (no dispersal) of 5,197 plant species exceeded | Sub-Saharan Africa | 3 | ||
| 33 | >2.5 | Sink service of terrestrial biosphere saturates and begins turning into a net carbon source | Globe | 55, 56 | ||
| 34 | 2.5 | 2°C SST | Extinction of coral reef ecosystems (overgrown by algae) | Indian Ocean | 9 | |
| 35 | 2.5 | 1.9-4.3 | 42% of UK land area with bioclimate unlike any currently found there; in Hampshire, declines in curlew and hawfinch and gain in yellow-necked mouse numbers; loss of montane habitat in Scotland; potential bracken invasion of Snowdonia montane areas | 57 | ||
| 36 | 2.5 | 2.0-3.0 | Major loss of Amazon rainforest with large losses of biodiversity | S. America, Globe | 21, 46 | |
| 37 | 2.5 | 20-70% loss (mean 44%) of coastal bird habitat at 4 sites | USA | 29 | ||
| 38 | 2.6 | 1.6-3.5 | Most areas experience 20-34% increase in number ³7day periods with Forest Fire Weather Index >45: increased fire frequency converts forest and maquis to scrub, causing more pest outbreaks | Mediterranean | 34 | |
| 39 | 2.6 | 4-21% of plants committed to extinction | Europe | 1 | ||
| 40 | 2.7 | Bioclimatic envelopes exceeded leading to eventual transformation of 16% of global ecosystems: loss of 58% wooded tundra, 31% cool conifer forest, 25% scrubland, 20% grassland/steppe, 21% tundra, 21% temperate deciduous forest, 19% savanna. Ecosystems variously lose 5-66% of their areal extent. | Globe | 6 |
| No. | ΔTg above pre-ind | ΔTg above pre-ind (range) | ΔTreg above 1990 (range) | Impacts to unique or widespread ecosystems or population systems | Region | Ref. no. |
|---|---|---|---|---|---|---|
| 41 | 2.8 | 1.2-4.5 | 1-3 | Extensive loss/conversion of habitat in Kakadu wetland due to sea-level rise and saltwater intrusion | Australia | 10 |
| 42 | 2.8 | 2.5-3.0 | Multi-model mean 62% (range 40-100%) loss of Arctic summer ice extent, high risk of extinction of polar bears, walrus, seals; Arctic ecosystem stressed | Arctic | 11,53 | |
| 43 | 2.8 | 2.3-4.6 | 2.1-2.5 | Cloud-forest regions lose hundreds of metres of elevational extent, potential extinctions ΔTreg 2.1°C for C. America and ΔTreg 2.5°C for Africa | C. America, Tropical Africa, Indonesia | 17 |
| 44 | 2.8 | 2.1-3.1 | 3 | Eventual loss of 9-62% of the mammal species from Great Basin montane areas | USA | 32 |
| 45 | 2.8 | 1.9-3.8 | 3 | 38-54% loss of waterfowl habitat in Prairie Pothole region | USA | 37, 38 |
| 46 | 2.9 | 3.2-6.6 | 50% loss existing tundra offset by only 5% eventual gain; millions of Arctic-nesting shorebird species variously lose up to 5-57% of breeding area; high-Arctic species most at risk; geese species variously lose 5-56% of breeding area | Arctic | 14 | |
| 47 | 2.9 | Latitude of northern forest limits shifts N. by 0.5° latitude in W. Europe, 1.5° in Alaska, 2.5° in Chukotka and 4° in Greenland | Arctic | 40 | ||
| 48 | 2.9 | 1.6-4.1 | Threat of marine ecosystem disruption through loss of aragonitic pteropods | Southern Ocean | 49 | |
| 49 | 2.9 | 1.6-4.1 | 70% reduction in deep-sea cold-water aragonitic corals | Ocean basins | 48 | |
| 50 | 2.9 | 2.1-3.9 | 21-36% of butterflies committed to extinction; >50% range loss for 83% of 24 latitudinally-restricted species | Australia | 1,30 | |
| 51 | 2.9 | 2.6-3.3 | 2.1-2.8 | 21-52% (mean 35%) of species committed to extinction | Globe iv | 1 |
| 52 | 2.9 | Substantial loss of boreal forest | China | 15 | ||
| 53 | 3.0 | 66 of 165 rivers studied lose >10% of their fish species | Globe | 19 | ||
| 54 | 3.0 | 1.9-3.5 | 20% loss of coastal migratory bird habitat | Delaware, USA | 36 | |
| 55 | 3.1 | 2.3-3.7 | 2°C SST | Extinction of remaining coral reef ecosystems (overgrown by algae) | Globe | 2 |
| 56 | 3.1 | 1.9-4.1 | 3-4 | Alpine systems in Alps degraded | Europe | 8 |
| 57 | 3.1 | 2.5-4.0 | 2 | High risk of extinction of golden bowerbird as habitat reduced by 90% | Australia | 4 |
| 58 | 3.1 | 1.8-4.2 | 3-4 | Risk of extinction of alpine species | Europe | 41 |
| 59 | 3.3 | 2.0-4.5 | Reduced growth in warm-water aragonitic corals by 20%-60%; 5% decrease in global phytoplankton productivity | Globe | 2, 47, 48 | |
| 60 | 3.3 | 2.3-3.9 | 2.6-2.9 | Substantial loss of alpine zone, and its associated flora and fauna (e.g., alpine sky lily and mountain pygmy possum) | Australia | 45 |
| 61 | 3.3 | 2.8-3.8 | 2 | Risk of extinction of Hawaiian honeycreepers as suitable habitat reduced by 62-89% | Hawaii | 18 |
| 62 | 3.3 | 3.7 | 4-38% of birds committed to extinction | Europe | 1 | |
| 63 | 3.4 | 6-22% loss of coastal wetlands; large loss migratory bird habitat particularly in USA, Baltic and Mediterranean | Globe | 35, 36 | ||
| 64 | 3.5 | 2.0-5.5 | Predicted extinction of 15-40% endemic species in global biodiversity hotspots (case “narrow biome specificity”) | Globe | 50 | |
| 65 | 3.5 | 2.3-4.1 | 2.5 – 3.5 | Loss of temperate forest wintering habitat of monarch butterfly | Mexico | 28 |
| 66 | 3.6 | 2.6-4.3 | 3 | Bioclimatic limits of 50% of eucalypts exceeded | Australia | 12 |
| 67 | 3.6 | 2.6-3.7 | 30-40% of 277 mammals in 141 parks critically endangered/extinct; 15-20% endangered | Africa | 23 | |
| 68 | 3.6 | 3.0-3.9 | Parts of the USA lose 30-57% neotropical migratory bird species richness | USA | 43 | |
| 69 | 3.7 | Few ecosystems can adapt | Globe | 6 | ||
| 70 | 3.7 | 50% all nature reserves cannot fulfil conservation objectives | Globe | 6 | ||
| 71 | 3.7 | Bioclimatic envelopes exceeded leading to eventual transformation of 22% of global ecosystems; loss of 68% wooded tundra, 44% cool conifer forest, 34% scrubland, 28% grassland/steppe, 27% savanna, 38% tundra and 26% temperate deciduous forest. Ecosystems variously lose 7-74% areal extent. | Globe | 6 | ||
| 72 | 3.9 | 4-24% plants critically endangered/extinct; mean species turnover of 63% (spatial range 22-90%); mean species loss of 42% (spatial range 2.5-86%) | Europe | 22 |
| No. | ΔTg above pre-ind | ΔTg above pre-ind (range) | ΔTreg above 1990 (range) | Impacts to unique or widespread ecosystems or population systems | Region | Ref. no. |
|---|---|---|---|---|---|---|
| 73 | 4.0 | 3.0-5.1 | 3 | Likely extinctions of 200-300 species (32-63%) of alpine flora | New Zealand | 33 |
| 74 | >4.0 | 3.5 | 38-67% of frogs, 48-80% of mammals, 43-64% of reptiles and 49-72% of birds committed to extinction in Queensland as 85-90% of suitable habitat lost | Australia | 1, 7 | |
| 75 | >>4.0 | 5 | Bioclimatic limits of 73% of eucalypts exceeded | Australia | 12 | |
| 76 | >>4.0 | 5 | 57 endemic frogs/mammal species eventually extinct, 8 endangered | Australia | 7 | |
| 77 | >>4.0 | 7 | Eventual total extinction of all endemic species of Queensland rainforest | Australia | 7 | |
| 78 | 5.2 | 62-100% loss of bird habitat at 4 major coastal sites | USA | 29 |
4.5 Costs and valuation of ecosystem goods and services
There is growing interest in developing techniques for environmental accounting. To that end, definitions of ecosystem goods and services are currently fluid. For example, ecosystem services accrue to society in return for investing in or conserving natural capital ( Heal, 2007 [2007] ), or ecosystem services are ultimately the end products of nature, the aspects of nature that people make choices about ( (Boyd, 2006 ) ). Definitions aside, all humans clearly rely on ecosystem services ( Reid et al., 2005 [NPR, SRC] ). While many efforts have been made to use standard economic techniques to estimate the economic value of ecosystem goods and services ( Costanza et al., 1997 [JoC] , 2000; ( Costanza, 2000, ) 2001; Daily et al., 2000 [JoC] ; Giles, 2005 [JoC] ; Reid et al., 2005 [NPR, SRC] ), others argue that such efforts are not only largely futile and flawed ( (Pearce, 1998; ) ( Toman, 1998b; ) ( Bockstael et al., 2000; ) Pagiola et al., 2004 [NPR] ), but may actually provide society a disservice ( (Ludwig, 2000; ) ( Kremen, 2005 ) ). The estimates from these techniques range from unknown (incomparability cf. Chang, 1997 [NPR] ), or invaluable, or infinite ( (Toman, 1998b ) ) because of lack of human substitutes, to about 38*1012 US$ per annum (updated to 2000 levels – Costanza et al., 1997 [JoC] ; Balmford et al., 2002 [JoC] ; Hassan et al., 2005 [NPR] ), which is larger but of similar magnitude than the global gross national product (GNP) of 31*1012 US$ per annum ( 2000 levels). These monetary estimates are usually targeted at policy-makers to assist assessments of the economic benefits of the natural environment ( (Farber et al., 2006 ) ) in response to cost-benefit paradigms. Some argue ( Balmford et al., 2002 [JoC] , 2005; Reid et al., 2005 [NPR, SRC] ) that unless ecosystem values are recognised in economic terms, ecosystems will continue their decline, placing the planet’s ecological health at stake (Millennium Ecosystem Assessment, 2005 [MoS, ARC] ). Others argue that ecosystems provide goods and services which are invaluable and need to be conserved on more fundamental principles, i.e., the precautionary principle of not jeopardising the conditions for a decent, healthy and secure human existence on this planet (e.g., ( Costanza et al., 2000; ) ( van den Bergh, 2004 ) ), or a moral and ethical responsibility to natural systems not to destroy them.
What is sometimes lost in the arguments is that natural capital (including ecosystem goods and services) is part of society’s capital assets ( Arrow et al., 2004 [PoC, JoC, ARC] ). The question then may be considered as whether one should maximise present value or try to achieve a measure of sustainability. In either case, it is the change in quantities of the capital stock that must be considered (including ecosystem services). One approach in considering valuation of ecosystem services is to calculate how much of one type of capital asset would be needed to compensate for the loss of one unit of another type of capital asset ( Arrow et al., 2004 [PoC, JoC, ARC] ). What is not disputed is that factoring in the full value of ecosystem goods and services, whether in monetary or non-monetary terms, distorts measures of economic wealth such that a country may be judged to be growing in wealth according to conventional indicators, while it actually becomes poorer due to the loss of natural resources ( Balmford et al., 2002 [JoC] ; Millennium Ecosystem Assessment, 2005 [MoS, ARC] ; Mock, 2005 [NPR] p. 33-53.). Ignoring such aspects almost guarantees opportunity costs. For instance Balmford et al. 2002 [JoC] ) estimated a benefit-cost ratio of at least 100:1 for an effective global conservation programme setting aside 15% of the current Earth’s surface if all aspects conventionally ignored are factored in. Additionally, many sectors and industries depend directly or indirectly on ecosystems and their services. The impacts of climate change could hold enormous costs for forests and coastal marine systems, as well as for managed agricultural systems ( Epstein and Mills, 2005 [NPR, MoS, ARC] ; Stern, 2007 [NPR, 2007] ). Multiple industries, such as timber, fisheries, travel, tourism and agriculture, are threatened by disturbances caused by climate change. Impacts on these sectors will influence financial markets, insurance companies and large multinational investors ( Mills, 2005 [JoC, ARC] ).
The United Nations has recognised the need to develop integrated environmental and economic accounting. However, many difficulties remain, especially as ecosystems may be the most difficult of all environmental assets to quantify ( (Boyd, 2006 ) ). There is a growing recognition that national accounting procedures need to be modified to include values for ecosystem goods and services ( Heal, 2007 [2007] ). Outside of the techniques mentioned above (often using contingent valuation) others have argued for developing a Green GDP to describe the state of nature and its worth, or an Ecosystem Services Index to account for all of nature’s contributions to the welfare of human society ( Banzhaf and Boyd, 2005 [NPR, MoS] ;( Boyd, 2006 ) ). Ultimately, it may be developing economies that are the most sensitive to the direct impacts of climate change, because they are more dependent on ecosystems and agriculture ( Stern, 2007 [NPR, 2007] ). As such, it is the poor that depend most directly on ecosystem services. Thus the degradation of these systems and their services will ultimately exacerbate poverty, hunger and disease, and obstruct sustainable development (e.g., Millennium Ecosystem Assessment, 2005 [MoS, ARC] ; Mock, 2005 [NPR] ; Mooney et al., 2005 [JoC] ; Stern, 2007 [NPR, 2007] ).
4.6 Acclimation and adaptation: practices, options and constraints
Although climate change is a global issue, local efforts can help maintain and enhance resilience and limit some of the longer-term damages from climate change (e.g., Hughes et al., 2003 [MoS, SRC] ;( Singh, 2003; ) ( Opdam and Wascher, 2004 ) ). This section discusses adaptation options with respect to natural ecosystems. Adaptation of these ecosystems involves only reactive, autonomous responses to ongoing climate change, including changes in weather variability and extremes. However, ecosystem managers can proactively alter the context in which ecosystems develop. In this way they can improve the resilience, i.e., the coping capacity, of ecosystems (see Glossary). Such ecosystem management involves anticipatory adaptation options. Identifying adaptation responses and adaptation options is a rapidly developing field, so the discussion below is not exhaustive. However, one should realise that beyond certain levels of climate change ( Hansen et al., 2003 [NPR] ; Table 4.1 , Figure 4.4 ) impacts on ecosystems are severe and largely irreversible.
4.6.1 Adaptation options
As climatic changes occur, natural resource management techniques can be applied to increase the resilience of ecosystems. Increasing resilience is consistent also with the ‘ecosystem approach’ developed by the Convention on Biological Diversity (CBD) which is a “strategy for management of land, water and living resources that promotes conservation and sustainable use in an equitable way” ( Smith and Malthby, 2003 [NPR] ). There are many opportunities to increase resilience ( (Cropp and Gabrica, 2002; ) Tompkins and Adger, 2003 [NPR] ); however, they may only be effective for lower levels of climate change (²2-3°C, Executive Summary, Figure 4.4 , Table 4.1 ).
Effective responses depend on an understanding of likely regional climatic and ecological changes. Monitoring environmental change, including climate, and associated ecosystem responses is vital to allow for adjustments in management strategies (e.g., Adger et al., 2003 [ARC] ; Moldan et al., 2005 [NPR, ARC] ). Although many adaptation options are available to wildlife managers, uncertainty about the magnitude and timing of climate change and delayed ecosystem responses (e.g., Section 4.4.5 ) may discourage their application. Nevertheless, ‘no regrets’ decisions based on the ‘precautionary principle’ appear preferable. Actions to reduce the impact of other threats, such as habitat fragmentation or destruction, pollution and introduction of alien species, are very likely to enhance resilience to climate change (e.g., ( Goklany, 1998; ) Inkley et al., 2004 [NPR, SRC] ;( Opdam and Wascher, 2004 ) ). Such proactive approaches would encourage conservation planning that is relevant both today and in the future. Techniques that allow the management of conservation resources in response to climate variability may ultimately prove to be the most beneficial way of preparing for possible abrupt climate change by increasing ecosystem resilience ( Bharwani et al., 2005 [MoS, ARC] ).
A few key options to adapt at least to lower levels of climate change in intensively managed ecosystems ( Chapter 5 ) have been suggested (e.g., Hannah et al., 2002a [SRC] , 2002b; ( Hannah and Lovejoy, 2003; ) Hansen et al., 2003 [NPR] ). Expansion of reserve systems can potentially reduce the vulnerability of ecosystems to climate change ( McNeely and Schutyser, 2003 [NPR, MoS] ). Reserve systems may be designed with some consideration of long-term shifts in plant and animal distributions, natural disturbance regimes and the overall integrity of the protected species and ecosystems (e.g., Williams et al., 2005 [SRC] ). Ultimately, adaptation possibilities are determined by the conservation priorities of each reserve and by the magnitude and nature of the change in climate. Strategies to cope with climate change are beginning to be considered in conservation ( (Cowling et al., 1999; ) Chopra et al., 2005 [NPR, SRC] ; Scott and Lemieux, 2005 [ARC] ), and highlight the importance of planning guided by future climate scenarios.
A primary adaptation strategy to climate change and even current climate variability is to reduce and manage the other stresses on species and ecosystems, such as habitat fragmentation and destruction, over-exploitation, eutrophication, desertification and acidification ( Inkley et al., 2004 [NPR, SRC] ; Duraiappah et al., 2005 [NPR] ; Robinson et al., 2005 [NPR, ARC] ; Worm et al., 2006 [JoC] Robinson et al. 2005 [NPR, ARC] ) suggest that this may be the only practical large-scale adaptation policy available for marine ecosystems. In addition to removing other stressors it is necessary to maintain viable, connected and genetically diverse populations ( Inkley et al., 2004 [NPR, SRC] ; Robinson et al., 2005 [NPR, ARC] ). Small, isolated populations are often more prone to local extirpations than larger, more widespread populations (e.g., Gitay et al., 2002 [NPR, ARC] ;( Davis et al., 2005; ) Lovejoy and Hannah, 2005 [NPR] ). Although connectivity, genetic diversity and population size are important current conservation goals, climate change increases their importance. The reduction and fragmentation of habitats may also be facilitated through increases in agricultural productivity (e.g., Goklany and Trewavas, 2003 [JoC] ) reducing pressures on natural ecosystems. However, increasing demand for some types of biofuels may negate this potential benefit (e.g., Busch, 2006 [MoS] ).
Reducing stress on ecosystems is difficult, especially in densely populated regions. Recent studies in southern Africa have signalled the need for policy to focus on managing areas outside protected areas (e.g., subsistence rangelands – von Maltitz et al., 2006 [NPR] ). This can, in part, be achieved through the devolution of resource ownership and management to communities, securing community tenure rights and incentives for resource utilisation. This argument is based on the observation that greater species diversity occurs outside protected areas that are more extensive ( Scholes et al., 2004 [NPR, SRC] ). Species migration between protected areas in response to shifting climatic conditions is likely to be impeded, unless assisted by often costly interventions geared towards landscapes with greater ecological connectivity. Strategic national policies could co-ordinate with communal or private land-use systems, especially when many small reserves are involved and would be particularly cost-effective if they address climate change proactively. Finally, migration strategies are very likely to become substantially more effective when they are implemented over larger regions and across national borders (e.g., Hansen et al., 2003 [NPR] ).
Controlled burning and other techniques may be useful to reduce fuel load and the potential for catastrophic wildfires. It may also be possible to minimise the effect of severe weather events by, for example, securing water rights to maintain water levels through a drought, or by having infrastructure capable of surviving floods. Maintaining viable and widely dispersed populations of individual species also minimises the probability that localised catastrophic events will cause significant negative effects (e.g., hurricane, typhoon, flood).
Climate change is likely to increase opportunities for invasive alien species because of their adaptability to disturbance ( Stachowicz et al., 2002 [JoC] ;( Lake and Leishman, 2004 ) ). Captive breeding for reintroduction and translocation or the use of provenance trials in forestry are expensive and likely to be less successful if climate change is more rapid. Such change could result in large-scale modifications of environmental conditions, including the loss or significant alteration of existing habitat over some or all of a species’ range. Captive breeding and translocation should therefore not be perceived as panaceas for the loss of biological diversity that might accompany large changes in the climate. Populations of many species are already perilously small, and further loss of habitat and stress associated with severe climate change may push many taxa to extinction.
A costly adaptation option would be the restoration of habitats currently under serious threat, or creation of new habitats in areas where natural colonisation is unlikely to occur (Anonymous, 2000 ). In many cases the knowledge of ecosystem interactions and species requirements may be lacking. Engineering habitats to facilitate species movements may call for an entirely new field of study. Engineering interactions to defend coastlines, for example, that change the connectivity of coastal ecosystems, facilitate the spread of non-native species ( (Bulleri, 2005 ) ) as well as warm-temperate species advancing polewards ( Helmuth et al., 2006 [MoS] ;( Mieszkowska et al., 2006 ) ).
Ultimately, managers may need to enhance or replace diminished or lost ecosystem services. This could mean manual seed dispersal or reintroducing pollinators. In the case of pest outbreaks, the use of pesticides may be necessary. Enhancing or replacing other services, such as contributions to nutrient cycling, ecosystem stability and ecosystem biodiversity may be much more difficult. The loss or reduced capacity of ecosystem services is likely to be a major source of ‘surprises’ from climate change.
4.6.2 Assessing the effectiveness and costs of adaptation options
There are few factual studies that have established the effectiveness and costs of adaptation options in ecosystems. Unfortunately, this makes a comprehensive assessment of the avoided damages (i.e., benefits) and costs impossible (see also Section 4.5 ). But the costs involved in monitoring, increasing the resilience of conservation networks and adaptive management are certainly large. For example, the money spent annually on nature conservation in the Netherlands was recently estimated to be ¤1 billion (Milieu en Natuurplanbureau, 2005 ). Of this amount, ¤285 million was used to manage national parks and reserves and ¤280 million was used for new reserve network areas and habitat improvement; the main objective being to reduce fragmentation between threatened populations and to respond to other threats. The reserve network planned for the Netherlands (to be established by 2020 ) will increase the resilience of species, populations and ecosystems to climate change, but at a high cost. Although not defined explicitly in this way, a significant proportion of these costs can be interpreted as climate adaptation costs.
The UN Convention on Biological Diversity (CBD, http://www.biodiv.org) aims to conserve biodiversity, to sustainably use biodiversity and its components and to fairly and equitably share benefits arising from the utilisation of biodiversity. This goes much further than most national biodiversity policies. The CBD explicitly recognises the use of biodiversity, ecosystems and their services and frames this as a developmental issue. As such, it extends beyond UNFCCC’s objective of “avoiding dangerous human interference with the climate system at levels where ecosystems cannot adapt naturally”. The main tool proposed by the CBD is the ecosystem approach ( Smith and Malthby, 2003 [NPR] ) based on integrated response options that intentionally and actively address ecosystem services (including biodiversity) and human well-being simultaneously, and involve all stakeholders at different institutional levels. The ecosystem approach resembles sustainable forest management projects ( FAO, 2001 [NPR] ). In theory, the ecosystem approach helps the conservation and sustainable use of biodiversity, but applications of the approach have had limited success ( Brown et al., 2005a [NPR] ). Integrated responses include, however, learning by doing; a proactive approach that should increase the resilience of ecosystems and biodiversity.
4.6.3 Implications for biodiversity
Many studies and assessments stress the adverse impacts of climate change on biodiversity (e.g., Gitay et al., 2002 [NPR, ARC] ;( Hannah and Lovejoy, 2003; ) Thomas et al., 2004a [JoC, SRC] ; Lovejoy and Hannah, 2005 [NPR] ; Schröter et al., 2005 [JoC, SRC] ; Thuiller et al., 2005b [JoC, SRC] ; van Vliet and Leemans, 2006 [NPR, SRC] ), but comprehensive appraisals of adaptation options to deal with declining biodiversity are rare.
4.6.4 Interactions with other policies and policy implications
Formulating integrated policies that cut across multiple UN conventions, such as the UNFCCC, CBD and Convention to Combat Desertification (CCD), could produce win-win situations in addressing climate change, increasing resilience and dealing with other policy issues ( Nnadozie, 1998 [NPR] ). Strategies aimed at combating desertification, for example, contribute towards increased soil carbon and moisture levels. Mitigation strategies focused on afforestation, including projects under the Clean Development Mechanism (CDM, see Glossary), could help ecosystem adaptation through improved ecological connectivity. The ecosystem approach can fulfil objectives specified by different conventions ( Reid et al., 2005 [NPR, SRC] ) and, in assessing adaptation strategies, such synergies could be identified and promoted.
4.7 Implications for sustainable development
Over the past 50 years, humans have converted and modified natural ecosystems more rapidly and over larger areas than in any comparable period of human history (e.g., Steffen et al., 2004 [NPR] ). These changes have been driven by the rapidly growing demands for food, fish, freshwater, timber, fibre and fuel (e.g., Vitousek et al., 1997 [JoC] ) and have contributed to substantial net gains in human well-being and economic development, while resulting in a substantial and largely irreversible loss of biodiversity and degradation in ecosystems and their services ( Reid et al., 2005 [NPR, SRC] ).
The consequences of policies to address the vulnerability of ecosystems to climate change at both the national and international level are not yet fully understood. There is growing evidence that significant impacts on the environment may result from perverse or unintended effects of policies from other sectors, which directly or indirectly have adverse consequences on ecosystems and other environmental processes ( Chopra et al., 2005 [NPR, SRC] ). Land re-distribution policies, for example, while designed to increase food self-sufficiency also contribute to reducing carbon sequestration and loss of biodiversity through extensive clear-cutting.
Effective mechanisms to analyse cross-sectoral impacts and to feed new scientific knowledge into policy-making are necessary ( Schneider, 2004 [PoC, JoC, ARC] ). There is substantial evidence to suggest that developing and implementing policies and strategies to reduce the vulnerability of ecosystems to climate change is closely linked to the availability of capacity to address current needs (e.g., Chanda, 2001 [NPR] ). Thus, prospects for successful adaptation to climate change will remain limited as long as factors (e.g., population growth, poverty and globalisation) that contribute to chronic vulnerability to, for example, drought and floods, are not resolved ( Kates, 2000 [JoC, ARC] ; Reid et al., 2005 [NPR, SRC] ).
4.7.1 Ecosystems services and sustainable development
Large differences in natural and socio-economic conditions among regions mitigate against simple solutions to the problem of ecosystem degradation and loss of services. Many interactions, lags and feedbacks, including those that operate across a range of spatial, temporal and organisational scales generate complex patterns which are not fully understood. Past actions to slow or reverse the degradation of ecosystems have yielded significant results, but these improvements have generally not kept pace with growing pressures ( Reid et al., 2005 [NPR, SRC] ). However, sound management of ecosystem services provides several cost-effective opportunities for addressing multiple development goals in a synergistic manner ( Reid et al., 2005 [NPR, SRC] ).
Progress achieved in addressing the Millennium Development Goals (MDGs) is unlikely to be sustained if ecosystem services continue to be degraded ( (Goklany, 2005 ) ). The role of ecosystems in sustainable development and in achieving the MDGs involves an array of stakeholders ( (Jain, 2003; ) Adeel et al., 2005 [NPR, ARC] ). Evidence from different parts of the world shows that in most cases it is far from clear who is ‘in charge’ of the long-term sustainability of an ecosystem, let alone of the situation under future climates. Responding and adapting to the impacts of climate change on ecosystems calls for a clear and structured system of decision making at all levels ( (Kennett, 2002 ) ). Impacts of climate change on ecosystems also show strong interrelationships with ecosystem processes and human activities at various scales over time. Addressing these impacts requires a co-ordinated, integrated, cross-sectoral policy framework with a long-term focus; a strategy that so far has not been easy to implement ( (Brown, 2003 ) ).
4.7.2 Subsistence livelihoods and indigenous peoples
The impacts of climate change on ecosystems and their services will not be distributed equally around the world. Dryland, mountain and mediterranean regions are likely to be more vulnerable than others ( Gitay et al., 2001 [NPR, ARC] ) and ecosystem degradation is largest in these regions ( Hassan et al., 2005 [NPR] ). Climate change is likely to cause additional inequities, as its impacts are unevenly distributed over space and time and disproportionately affect the poor ( Tol, 2001 [ARC] ; Stern, 2007 [NPR, 2007] ). The term ‘double exposure’ has been used for regions, sectors, ecosystems and social groups that are confronted both by the impacts of climate change and by the consequences of economic globalisation ( O’Brien and Leichenko, 2000 [JoC, ARC] ). Thus special attention needs to be given to indigenous peoples with subsistence livelihoods and groups with limited access to information and few means of adaptation. As a result climate change and sustainable development need to incorporate issues of equity ( Kates, 2000 [JoC, ARC] ;( Jain, 2003; ) Richards, 2003 [NPR] ).
4.8 Key uncertainties and research priorities
Key uncertainties listed here are those that limit our ability to project climate change impacts on ecosystems, but only if they have implications at sub-continental and higher spatial scales, are relevant for many species, populations and communities, or significantly weaken a modelling result. In terms of climate uncertainty, it is important to highlight that projections for precipitation carry a significantly higher uncertainty than temperature, yet play a major role for many projections obtained from modelling approaches. In relation to projecting climate change impacts on ecosystems, we find key sources of uncertainty to include:
- inadequate representation of the interactive coupling between ecosystems and the climate system and, furthermore, of the multiple interacting drivers of global change. This prevents a fully integrated assessment of climate change impacts on ecosystem services;
- major biotic feedbacks to the climate system, especially through trace gases from soils in all ecosystems, and methane from labile carbon stocks such as wetlands, peatlands, permafrost and yedoma;
- how aggregation within current DGVMs with respect to the functional role of individual species and the assumption of their instantaneous migration biases impact estimates;
- the net result of changing disturbance regimes (especially through fire, insects and land-use change) on biotic feedbacks to the atmosphere, ecosystem structure, function, biodiversity and ecosystem services;
- the magnitude of the CO2-fertilisation effect in the terrestrial biosphere and its components over time;
- the limitations of climate envelope models used to project responses of individual species to climate changes, and for deriving estimations of species extinction risks;
- the synergistic role of invasive alien species in both biodiversity and ecosystem functioning;
- the effect of increasing surface ocean CO2 and declining pH on marine productivity, biodiversity, biogeochemistry and ecosystem functioning;
- the impacts of interactions between climate change and changes in human use and management of ecosystems as well as other drivers of global environmental change.
Guided by the above, the following research needs can be identified as priorities for reducing uncertainties.
- Identify key vulnerabilities in permafrost–soil–vegetation interactions at high latitudes, and their potential feedback to the biosphere trace-gas composition. Recent estimates suggest that terrestrial permafrost contains more than 1,000 PgC, which is increasingly emitting CO2 and more importantly, methane (e.g., Walter et al., 2006; Zimov et al., 2006). The implications of this for abrupt and significant climate forcing are significant (e.g., Schellnhuber, 2002; iLEAPS, 2005; Symon et al., 2005, p. 1015; Lelieveld, 2006; Zimov et al., 2006).
- More robust modelling of interactions between biota and their geophysical environment using several independently developed DGVMs and Earth-system models. Validation (Price et al., 2001) beyond model intercomparisons is required, especially also with respect to the methane cycle. The goal should be to narrow uncertainties relating to the vulnerability of the carbon sequestration potential of ecosystems including more realistic estimates of lagged and threshold responses (e.g., Scheffer et al., 2001; iLEAPS, 2005).
- More emphasis on precipitation projections (e.g., Handel and Risbey, 1992) and resulting water regime effects. These should emphasise interactions between vegetation and atmosphere, including CO2-fertilisation effects, in mature forests in the Northern Hemisphere, seasonal tropical forests, and arid or semi-arid grassland and savannas (e.g., Jasienski et al., 1998; Karnosky, 2003).
- Improved understanding of the role of disturbance regimes, i.e., frequency and intensity of episodic events (drought, fire, insect outbreaks, diseases, floods and wind-storms) and that of alien species invasions, as they interact with ecosystem responses to climate change itself and pollution (e.g., Osmond et al., 2004; Opdam and Wascher, 2004).
- Development of integrated large spatial-scale remote sensing with long-term field studies (May, 1999b; Kräuchi et al., 2000; Morgan et al., 2001b; Osmond et al., 2004; Opdam and Wascher, 2004; Symon et al., 2005, p. 1019) to better address scale mismatches between the climate system and ecosystems (Root and Schneider, 1995).
- Studies on impacts of rising atmospheric CO2 on ocean acidification, and warming on coral reefs and other marine systems (Coles and Brown, 2003; Anonymous, 2004), and widening the range of terrestrial ecosystems for which CO2-fertilisation responses have been quantified (e.g., Bond et al., 2003).
- Validating species-specific climate envelope models by testing model projections against the plethora of range shifts observed in nature (e.g., Walther et al., 2001; Chapter 1 ).
- Advances in understanding the relationship between biodiversity and the resilience of ecosystem services at a scale relevant to human well-being, to quote Sir Robert May (1999a): “The relatively rudimentary state of ecological science prevents us from making reliable predictions about how much biological diversity we can lose before natural systems collapse and deprive us of services upon which we depend.”
- Improve identification of environmental key factors influencing ecosystem structures that determine functionality and provisioning services of ecosystems together with quantitative information on economic impacts (including implications for adaptation costs – Toman, 1998a; Winnett, 1998; Kremen, 2005; Symon et al., 2005, e.g., p. 1019).
- Integrative vulnerability studies on adaptive management responses to preserve biodiversity (including conservation and reservation management) and ecosystem services in relation to pressures from land-use change and climate change (Kappelle et al., 1999; Lorenzoni et al., 2005; Stenseth and Hurrell, 2005; Symon et al., 2005).
References
Aanes, R., B.E. Saether, F.M. Smith, E.J. Cooper, P.A. Wookey and N.A. Oritsland, 2002: The Arctic Oscillation predicts effects of climate change in two trophic levels in a high-arctic ecosystem. Ecol. Lett., 5, 445-453. [MoS]
Aber, J., R.P. Neilson, S. McNulty, J.M. Lenihan, D. Bachelet and R.J. Drapek, 2001: Forest processes and global environmental change: predicting the effects of individual and multiple stressors. BioScience, 51, 735-751. [MoS, SRC]
Adeel, Z., U. Safriel, D. Niemeijer, R. White, G. de Kalbermatten, M. Glantz, B. Salem, R. Scholes, M. Niamir-Fuller, S. Ehui and V. Yapi-Gnaore, Eds., 2005: Ecosystems and Human Well-being: Desertification Synthesis. Island Press, Washington, District of Columbia, 36 pp. [NPR, ARC]
Adger, W.N., S. Huq, K. Brown, D. Conway and M. Hulme, 2003: Adaptation to climate change in the developing world. Prog. Dev. Stud., 3, 179-195. [ARC]
Ainsworth, E.A. and S.P. Long, 2005: What have we learned from 15 years of free-air CO2 enrichment (FACE)? A meta-analytic review of the responses of photosynthesis, canopy properties and plant production to rising CO2. New Phytol., 165, 351-371. Clean
Alcamo, J., K. Kok, G. Busch, J. Priess, B. Eickhout, M. Rounsevell, D. Rothman and M. Heistermann, 2006: Searching for the future of land: scenarios from the local to global scale. Land-use and Land-cover Change: Local Processes, Global Impacts, E.F. Lambin and H. Geist, Eds., Springer Verlag, Berlin, 137-156. [NPR, SRC]
Alencar, A., D. Nepstad and M.D.V. Diaz, 2006: Forest understorey fire in the Brazilian Amazon in ENSO and non-ENSO years: area burned and committed carbon emissions. Earth Interactions, 10, 1-17. Clean
Allen Consulting Group, 2005: Climate Change Risk and Vulnerability: Promoting an Efficient Adaptation Response in Australia. Australian Greenhouse Office, Department of the Environment and Heritage, Canberra, 159 pp. [NPR]
Alsos, I.G., T. Engelskjon, L. Gielly, P. Taberlet and C. Brochmann, 2005: Impact of ice ages on circumpolar molecular diversity: insights from an ecological key species. Mol. Ecol., 14, 2739-2753. Clean
Andreae, M.O., D. Rosenfeld, P. Artaxo, A.A. Costa, G.P. Frank, K.M. Longo and M.A.F. Silva-Dias, 2004: Smoking rain clouds over the Amazon. Science, 303, 1337-1342. [JoC]
Andreone, F., J.E. Cadle, N. Cox, F. Glaw, R.A. Nussbaum, C.J. Raxworthy, S.N. Stuart, D. Vallan and M. Vences, 2005: Species review of amphibian extinction risks in Madagascar: conclusions from the global amphibian assessment. Conserv. Biol., 19, 1790-1802. Clean
Andrew, N.R. and L. Hughes, 2005: Herbivore damage along a latitudinal gradient: relative impacts of different feeding guilds. Oikos, 108, 176-182. [SRC]
Angert, A., S. Biraud, C. Bonfils, C.C. Henning, W. Buermann, J. Pinzon, C.J. Tucker and I. Fung, 2005: Drier summers cancel out the CO2 uptake enhancement induced by warmer springs. P. Natl. Acad. Sci. USA, 102, 10823-10827. [JoC]
Anisimov, O.A., N.I. Shiklomanov and F.E. Nelson, 2002a: Variability of seasonal thaw depth in permafrost regions: a stochastic modeling approach. Ecol. Model., 153, 217-227. [MoS, ARC]
Anisimov, O.A., A.A. Velichko, P.F. Demchenko, A.V. Eliseev, I.I. Mokhov and V.P. Nechaev, 2002b: Effect of climate change on permafrost in the past, present, and future. Izv. Atmos. Ocean. Phys., 38, S25-S39. [MoS, SRC]
Anonymous, 2000: Potential UK adaptation strategies for climate change. DETR Summary Report 99DPL013, Department of the Environment, Transport and the Regions, Wetherby, West Yorkshire, UK, 19 pp. [NPR]
Anonymous, 2001: Lothar: Der Orkan 1999. Eidg. Forschungsanstalt WSL and Bundesamt für Umwelt, Wald und Landschaft BUWAL, Birmensdorf, Bern, 365 pp. [NPR]
Anonymous, 2004: Priorities for research on the ocean in a high-CO2 world. Symposium on The Ocean in a High-CO2 World, Scientific Committee on Oceanic Research (SCOR) and the Intergovernmental Oceanographic Commission (IOC) of UNESCO, UNESCO Headquarters, Paris, 12 pp. [NPR]
Anyamba, A. and C.J. Tucker, 2005: Analysis of Sahelian vegetation dynamics using NOAA-AVHRR NDVI data from 1981-2003. J. Arid Environ., 63, 596-614. Clean
Araújo, M.B., R.J. Whittaker, R.J. Ladle and M. Erhard, 2005: Reducing uncertainty in projections of extinction risk from climate change. Global Ecol. Biogeogr., 14, 529-538. [MoS, ARC]
Arnell, N.W., M.G.R. Cannell, M. Hulme, R.S. Kovats, J.F.B. Mitchell, R.J. Nicholls, M.L. Parry, M.T.J. Livermore and A. White, 2002: The consequences of CO2 stabilisation for the impacts of climate change. Climatic Change, 53, 413-446. [JoC, ARC]
Arnell, N.W., M.J.L. Livermore, S. Kovats, P.E. Levy, R. Nicholls, M.L. Parry and S.R. Gaffin, 2004: Climate and socio-economic scenarios for global-scale climate change impacts assessments: characterising the SRES storylines. Global Environ. Chang., 14, 3-20. [JoC, ARC]
Arrigo, K.R. and D.N. Thomas, 2004: Large scale importance of sea ice biology in the Southern Ocean. Antarct. Sci., 16, 471-486. Clean
Arrow, K., P. Dasgupta, L. Goulder, G. Daily, P. Ehrlich, G. Heal, S. Levin, K.G. Maler, S. Schneider, D. Starett and B. Walker, 2004: Are we consuming too much? J. Econ. Perspect., 18, 147-172. [PoC, JoC, ARC]
Arzel, O., T. Fichefet and H. Goose, 2006: Sea ice evolution over the 20th and 21st centuries as simulated by current AOGCMs. Ocean Model., 12, 401-415. [MoS]
Asner, G.P., D.E. Knapp, E.N. Broadbent, P.J.C. Oliveira, M. Keller and J.N. Silva, 2005: Selective logging in the Brazilian Amazon. Science, 310, 480-482. [JoC]
Augustin, L., C. Barbante, P.R.F. Barnes, J.M. Barnola, M. Bigler, E. Castellano, O. Cattani, J. Chappellaz, D. Dahljensen, B. Delmonte, G. Dreyfus, G. Durand, S. Falourd, H. Fischer, J. Fluckiger, M.E. Hansson, P. Huybrechts, R. Jugie, S.J. Johnsen, J. Jouzel, P. Kaufmann, J. Kipfstuhl, F. Lambert, V.Y. Lipenkov, G.V.C. Littot, A. Longinelli, R. Lorrain, V. Maggi, V. Masson-Delmotte, H. Miller, R. Mulvaney, J. Oerlemans, H. Oerter, G. Orombelli, F. Parrenin, D.A. Peel, J.R. Petit, D. Raynaud, C. Ritz, U. Ruth, J. Schwander, U. Siegenthaler, R. Souchez, B. Stauffer, J.P. Steffensen, B. Stenni, T.F. Stocker, I.E. Tabacco, R. Udisti, R.S.W. van de Wal, M. van den Broeke, J. Weiss, F. Wilhelms, J.G. Winther, E.W. Wolff and M. Zucchelli, 2004: Eight glacial cycles from an Antarctic ice core. Nature, 429, 623-628. [JoC]
Bachelet, D., R.P. Neilson, J.M. Lenihan and R.J. Drapek, 2001: Climate change effects on vegetation distribution and carbon budget in the United States. Ecosystems, 4, 164-185. [SRC]
Bachelet, D., R.P. Neilson, T. Hickler, R.J. Drapek, J.M. Lenihan, M.T. Sykes, B. Smith, S. Sitch and K. Thonicke, 2003: Simulating past and future dynamics of natural ecosystems in the United States. Global Biogeochem. Cy., 17, 1045, doi:10.1029/2001GB001508. [MoS, SRC]
Badeck, F.W., A. Bondeau, K. Bottcher, D. Doktor, W. Lucht, J. Schaber and S. Sitch, 2004: Responses of spring phenology to climate change. New Phytol., 162, 295-309. [SRC]
Baillie, J.E.M., C. Hilton-Taylor and S.N. Stuart, Eds., 2006: 2004 IUCN Red List of Threatened Species: A Global Species Assessment. IUCN: The World Conservation Union, Gland, and Cambridge, 217 pp. [NPR]
Baker, T.R., O.L. Phillips, Y. Malhi, S. Almeida, L. Arroyo, A. Di Fiore, T. Erwin, N. Higuchi, T.J. Killeen, S.G. Laurance, W.F. Laurance, S.L. Lewis, A. Monteagudo, D.A. Neill, P.N. Vargas, N.C.A. Pitman, J.N.M. Silva and R.V. Martinez, 2004: Increasing biomass in Amazonian forest plots. Philos. T. Roy. Soc. Lond. B, 359, 353-365. Clean
Bakkenes, M., J.R.M. Alkemade, F. Ihle, R. Leemans and J.B. Latour, 2002: Assessing effects of forecasted climate change on the diversity and distribution of European higher plants for 2050. Global Change Biol., 8, 390-407. [JoC, MoS, SRC]
Balmford, A., A. Brunner, P. Cooper, R. Costanza, S. Farber, R.E. Green, M. Jenkins, P. Jefferiss, V. Jessamy, J. Madden, K. Munro, N. Myers, S. Naeem, J. Paavola, M. Rayment, S. Rosendo, J. Roughgarden, K. Trumper and R.K. Turner, 2002: Economic reasons for conserving wild nature. Science, 297, 950-953. [JoC]
Balmford, A., P. Crane, A. Dobson, R.E. Green and G.M. Mace, 2005: The 2010 challenge: data availability, information needs and extraterrestrial insights. Philos. T. Roy. Soc. Lond. B, 360, 221-228. Clean
Balzter, H., F.F. Gerard, C.T. George, C.S. Rowland, T.E. Jupp, I. McCallum, A. Shvidenko, S. Nilsson, A. Sukhinin, A. Onuchin and C. Schmullis, 2005: Impact of the Arctic Oscillation pattern on interannual forest fire variability in Central Siberia. Geophys. Res. Lett., 32, L14709, doi:10.1029/2005GL022526. [JoC, ARC]
Banzhaf, S. and J. Boyd, 2005: The architecture and measurement of an ecosystem services index. Discussion paper RFF DP 05-22, Resources for the Future, Washington, District of Columbia, 57 pp. [NPR, MoS]
Barber, D.G. and J. Iacozza, 2004: Historical analysis of sea ice conditions in M’Clintock channel and the Gulf of Boothia, Nunavut: implications for ringed seal and polar bear habitat. Arctic, 57, 1-14. Clean
Barbosa, P., G. Libertà and G. Schmuck, 2003: The European Forest Fires Information System (EFFIS) results on the 2003 fire season in Portugal by the 20th of August. Report 20030820, European Commission, Directorate General Joint Research Centre, Institute for Environment and Sustainability, Land Management Unit, Ispra, 10 pp. [NPR]
Barbraud, C. and H. Weimerskirch, 2001: Emperor penguins and climate change. Nature, 411, 183-186. [JoC]
Barnes, D.K.A., D.A. Hodgson, P. Convey, C.S. Allen and A. Clarke, 2006: Incursion and excursion of Antarctic biota: past, present and future. Global Ecol. Biogeogr., 15, 121-142. [MoS]
Bauder, E.T., 2005: The effects of an unpredictable precipitation regime on vernal pool hydrology. Freshwater Biol., 50, 2129-2135. [MoS]
Beaugrand, G., P.C. Reid, F. Ibanez, J.A. Lindley and M. Edwards, 2002: Reorganization of North Atlantic marine copepod biodiversity and climate. Science, 296, 1692-1694. [JoC, ARC]
Beaugrand, G., K.M. Brander, J.A. Lindley, S. Souissi and P.C. Reid, 2003: Plankton effect on cod recruitment in the North Sea. Nature, 426, 661-664. [JoC, SRC]
Beaumont, L.J. and L. Hughes, 2002: Potential changes in the distributions of latitudinally restricted Australian butterfly species in response to climate change. Global Change Biol., 8, 954-971. [JoC, SRC]
Becker, A. and H. Bugmann, 2001: Global change and mountain regions. IGBP Report, IGBP Secretariat, Royal Swedish Academy of Sciences, Stockholm, 88 pp. [NPR, SRC]
Beever, E.A., P.F. Brussard and J. Berger, 2003: Patterns of apparent extirpation among isolated populations of pikas (Ochotona princeps) in the Great Basin. J. Mammal., 84, 37-54. Clean
Behrenfeld, M.J. and P.G. Falkowski, 1997: A consumer’s guide to phytoplankton primary production models. Limnol. Oceanogr., 42, 1479-1491. [MoS]
Bellamy, P.H., P.J. Loveland, R.I. Bradley, R.M. Lark and G.J.D. Kirk, 2005: Carbon losses from all soils across England and Wales 1978-2003. Nature, 437, 245. [JoC]
Beniston, M., 2000: Environmental Change in Mountains and Uplands. Arnold, London, 172 pp. [NPR, SRC]
Beniston, M., 2003: Climatic change in mountain regions: a review of possible impacts. Climatic Change, 59, 5-31. [JoC, SRC]
Beniston, M., 2004: The 2003 heat wave in Europe: a shape of things to come? An analysis based on Swiss climatological data and model simulations. Geophys. Res. Lett., 31, L02202, doi:10.1029/2003GL018857. [JoC, MoS, SRC]
Beniston, M. and P. Jungo, 2002: Shifts in the distributions of pressure, temperature and moisture and changes in the typical weather patterns in the Alpine region in response to the behavior of the North Atlantic Oscillation. Theor. Appl. Climatol., 71, 29-42. [SRC]
Beniston, M. and D.B. Stephenson, 2004: Extreme climatic events and their evolution under changing climatic conditions. Global Planet. Change, 44, 1-9. [SRC]
Beniston, M., H.F. Diaz and R.S. Bradley, 1997: Climatic change at high elevation sites: an overview. Climatic Change, 36, 233-251. [JoC, SRC]
Benning, T.L., D. Lapointe, C.T. Atkinson and P.M. Vitousek, 2002: Interactions of climate change with biological invasions and land use in the Hawaiian Islands: modeling the fate of endemic birds using a geographic information system. P. Natl. Acad. Sci. USA, 99, 14246-14249. [JoC, MoS]
Berendse, F., N. van Breemen, H. Kanrydin, A. Buttler, M. Heijmans, M.R. Hoosbeek, J. Lee, E. Mitchell, T. Saarinen, H. Vassander and B. Wallen, 2001: Raised atmospheric CO2 levels and increased N deposition cause shifts in plant species composition and production in Sphagnum bogs. Global Change Biol., 7, 591-598. [JoC]
Bergengren, J.C., S.L. Thompson, D. Pollard and R.M. Deconto, 2001: Modeling global climate-vegetation interactions in a doubled CO2 world. Climatic Change, 50, 31-75. [JoC, MoS]
Bergeron, Y., S. Gauthier, V. Kafka, P. Lefort and D. Lesieur, 2001: Natural fire frequency for the eastern Canadian boreal forest: consequences for sustainable forestry. Can. J. Forest Res., 31, 384-391. Clean
Bergeron, Y., M. Flannigan, S. Gauthier, A. Leduc and P. Lefort, 2004: Past, current and future fire frequency in the Canadian boreal forest: implications for sustainable forest management. Ambio, 33, 356-360. [MoS]
Berry, P.M., P.A. Harrison, T.P. Dawson and C.A. Walmsley, 2005: Modelling natural resource responses to climate change (MONARCH): a local approach. UKCIP Technical Report, UK Climate Impacts Programme, Oxford, 24 pp. [NPR, MoS]
Berry, P.M., M.D.A. Rounsevell, P.A. Harrison and E. Audsley, 2006: Assessing the vulnerability of agricultural land use and species to climate change and the role of policy in facilitating adaptation. Environ. Sci. Policy, 9, 189-204. [SRC]
Berthelot, M., P. Friedlingstein, P. Ciais, P. Monfray, J.L. Dufresne, H. Le Treut and L. Fairhead, 2002: Global response of the terrestrial biosphere to CO2 and climate change using a coupled climate-carbon cycle model. Global Biogeochem. Cy., 16, 1084, doi:10.1029/2001GB001827. [MoS]
Bertness, M.D. and P.J. Ewanchuk, 2002: Latitudinal and climate-driven variation in the strength and nature of biological interactions in New England salt marshes. Oecologia, 132, 392-401. Clean
Betts, R.A., 2000: Offset of the potential carbon sink from boreal forestation by decreases in surface albedo. Nature, 408, 187-190. [JoC, ARC]
Betts, R.A. and H.H. Shugart, 2005: Dynamic ecosystem and Earth system models. Climate Change and Biodiversity, T.E. Lovejoy and L. Hannah, Eds., Yale University Press, New Haven, Connecticut, 232-251. [NPR, ARC]
Betts, R.A., P.M. Cox and F.I. Woodward, 2000: Simulated responses of potential vegetation to doubled-CO2 climate change and feedbacks on near-surface temperature. Global Ecol. Biogeogr., 9, 171-180. [MoS, ARC]
Betts, R.A., P.M. Cox, M. Collins, P.P. Harris, C. Huntingford and C.D. Jones, 2004: The role of ecosystem-atmosphere interactions in simulated Amazonian precipitation decrease and forest dieback under global climate warming. Theor. Appl. Climatol., 78, 157-175. [MoS, ARC]
Bharwani, S., M. Bithell, T.E. Downing, M. New, R. Washington and G. Ziervogel, 2005: Multi-agent modelling of climate outlooks and food security on a community garden scheme in Limpopo, South Africa. Philos. T. Roy. Soc. Lond. B, 360, 2183-2194. [MoS, ARC]
Billings, S.A., S.M. Schaeffer, S. Zitzer, T. Charlet, S.D. Smith and R.D. Evans, 2002: Alterations of nitrogen dynamics under elevated carbon dioxide in an intact Mojave Desert ecosystem: evidence from nitrogen-15 natural abundance. Oecologia, 131, 463-467. Clean
Billings, S.A., S.M. Schaeffer and R.D. Evans, 2003: Nitrogen fixation by biological soil crusts and heterotrophic bacteria in an intact Mojave Desert ecosystem with elevated CO2 and added soil carbon. Soil Biol. Biochem., 35, 643-649. Clean
Blaschke, L., M. Schulte, A. Raschi, N. Slee, H. Rennenberg and A. Polle, 2001: Photosynthesis, soluble and structural carbon compounds in two Mediterranean oak species (Quercus pubescens and Q. ilex) after lifetime growth at naturally elevated CO2 concentrations. Plant Biol., 3, 288-297. Clean
Blodau, C., 2002: Carbon cycling in peatlands: a review of processes and controls. Environ. Rev., 10, 111-134. Clean
Bockstael, N.E., A.M. Freeman, R.J. Kopp, P.R. Portney and V.K. Smith, 2000: On measuring economic values for nature. Environ. Sci. Technol., 34, 1384-1389. Clean
Boisvenue, C. and S.W. Running, 2006: Impacts of climate change on natural forest productivity: evidence since the middle of the 20th century. Global Change Biol., 12, 862-882. [JoC, ARC]
Bomhard, B., D.M. Richardson, J.S. Donaldson, G.O. Hughes, G.F. Midgley, D.C. Raimondo, A.G. Rebelo, M. Rouget and W. Thuiller, 2005: Potential impacts of future land use and climate change on the Red List status of the Proteaceae in the Cape Floristic Region, South Africa. Global Change Biol., 11, 1452-1468. [JoC, MoS, SRC]
Bonan, G.B., 2002: Ecological Climatology: Concepts and Applications. Cambridge University Press, Cambridge, 678 pp. [NPR]
Bond, W.J. and G.F. Midgley, 2000: A proposed CO2-controlled mechanism of woody plant invasion in grasslands and savannas. Global Change Biol., 6, 865-869. [JoC, SRC]
Bond, W.J. and J.J. Midgley, 2003: The evolutionary ecology of sprouting in woody plants. Int. J. Plant Sci., 164, S103-S114. [SRC]
Bond, W.J. and J.E. Keeley, 2005: Fire as a global ‘herbivore’: the ecology and evolution of flammable ecosystems. Trends Ecol. Evol., 20, 387-394. [SRC]
Bond, W.J., G.F. Midgley and F.I. Woodward, 2003: The importance of low atmospheric CO2 and fire in promoting the spread of grasslands and savannas. Global Change Biol., 9, 973-982. [JoC, SRC]
Bond, W.J., F.I. Woodward and G.F. Midgley, 2005: The global distribution of ecosystems in a world without fire. New Phytol., 165, 525-537. [SRC]
Bonin, A., P. Taberlet, C. Miaud and F. Pompanon, 2006: Explorative genome scan to detect candidate loci for adaptation along a gradient of altitude in the common frog (Rana temporaria). Mol. Biol. Evol., 23, 773-783. Clean
Bopp, L., O. Aumont, S. Belviso and P. Monfray, 2003: Potential impact of climate change on marine dimethyl sulfide emissions. Tellus B, 55, 11-22. Clean
Bopp, L., O. Boucher, O. Aumont, S. Belviso, J.L. Dufresne, M. Pham and P. Monfray, 2004: Will marine dimethylsulfide emissions amplify or alleviate global warming? A model study. Can. J. Fish. Aquat. Sci., 61, 826-835. [MoS]
Borghetti, M., S. Cinnirella, F. Magnani and A. Saracino, 1998: Impact of long-term drought on xylem embolism and growth in Pinus halepensis Mill. Trees–Struct. Funct., 12, 187-195. Clean
Bosch, J., L.M. Carrascal, L. Durain, S. Walker and M.C. Fisher, 2006: Climate change and outbreaks of amphibian chytriomycosis in a montane area of central Spain: is there a link? P. Roy. Soc. Lond. B, 274, 253-260. Clean
Bowman, D.M.J.S., A. Walsh and D.J. Milne, 2001: Forest expansion and grassland contraction within a Eucalyptus savanna matrix between 1941 and 1994 at Litchfield National Park in the Australian monsoon tropics. Global Ecol. Biogeogr., 10, 535-548. Clean
Boyd, J., 2006: The nonmarket benefits of nature: what should be counted in green GDP? Ecol. Econ., 61, 716-723. Clean
Breshears, D.D. and C.D. Allen, 2002: The importance of rapid, disturbance-induced losses in carbon management and sequestration. Global Ecol. Biogeogr., 11, 1-5. Clean
Breshears, D.D., N.S. Cobb, P.M. Rich, K.P. Price, C.D. Allen, R.G. Balice, W.H. Romme, J.H. Kastens, M.L. Floyd, J. Belnap, J.J. Anderson, O.B. Myers and C.W. Meyer, 2005: Regional vegetation die-off in response to global-change-type drought. P. Natl. Acad. Sci. USA, 102, 15144-15148. [JoC]
Broennimann, O., W. Thuiller, G. Hughes, G.F. Midgley, J.M.R. Alkemade and A. Guisan, 2006: Do geographic distribution, niche property and life form explain plants’ vulnerability to global change? Global Change Biol., 12, 1079-1093. [JoC, SRC]
Brohan, P., J.J. Kennedy, I. Haris, S.F.B. Tett and P.D. Jones, 2006: Uncertainty estimates in regional and global observed temperature changes: a new dataset from 1850. J. Geophys. Res. D, 111, D12106, doi:10.1029/2005JD006548. [MoS]
Brooks, M.L., C.M. D’Antonio, D.M. Richardson, J.B. Grace, J.E. Keeley, J.M. Ditomaso, R.J. Hobbs, M. Pellant and D. Pyke, 2004: Effects of invasive alien plants on fire regimes. BioScience, 54, 677-688. Clean
Brown, K., 2003: Integrating conservation and development: a case of institutional misfit. Front. Ecol. Environ., 1, 479-487. Clean
Brown, K., J. Mackensen, S. Rosendo, K. Viswanathan, L. Cimarrusti, K. Fernando, C. Morsello, I.M. Siason, S. Singh, I. Susilowati, M. Soccoro Manguiat, G. Bialluch and W.F. Perrin, 2005a: Integrated responses. Ecosystems and Human Well-being: Volume 3: Policy Responses, K. Chopra, R. Leemans, P. Kumar and H. Simons, Eds., Island Press, Washington, District of Columbia, 425-464. [NPR]
Brown, K.J., J.S. Clark, E.C. Grimm, J.J. Donovan, P.G. Mueller, B.C.S. Hansen and I. Stefanova, 2005b: Fire cycles in North American interior grasslands and their relation to prairie drought. P. Natl. Acad. Sci. USA, 102, 8865-8870. [JoC]
Brown, T.J. and B.L. Hall, 2001: Climate analysis of the 2000 fire season. CE-FA Report 01-02, Program for Climate Ecosystem and Fire Applications, Desert Research Institute, Division of Atmospheric Sciences, Reno, Nevada, 40 pp. [NPR]
Brown, T.J., B.L. Hall and A.L. Westerling, 2004: The impact of twenty-first century climate change on wildland fire danger in the western United States: an applications perspective. Climatic Change, 62, 365-388. [JoC]
Buchmann, N., 2002: Plant ecophysiology and forest response to global change. Tree Physiol., 22, 1177-1184. Clean
Buddemeier, R.W., J.A. Kleypas and R.B. Aronson, 2004: Coral Reefs and Global Climate Change: Potential Contributions of Climate Change to Stresses on Coral Reef Ecosystems. Pew Center on Global Climatic Change, Arlington, Virginia, 44 pp. [NPR]
Bugmann, H. and C. Pfister, 2000: Impacts of interannual climate variability on past and future forest composition. Reg. Environ. Change, 1, 112-125. [MoS, SRC]
Bugmann, H., B. Zierl and S. Schumacher, 2005: Projecting the impacts of climate change on mountain forests and landscapes. Global Change and Mountain Regions: An Overview of Current Knowledge, U.M. Huber, H.K.M. Bugmann and M.A. Reasoner, Eds., Springer, Berlin, 477-488. [NPR, SRC]
Bulleri, F., 2005: Role of recruitment in causing differences between intertidal assemblages on seawalls and rocky shores. Mar. Ecol.–Prog. Ser., 287, 53-64. Clean
Burgan, R.E., P.L. Andrews, L.S. Bradshaw, C.H. Chase, R.A. Hartford and D.J. Latham, 1997: WFAS: wildland fire assessment system. Fire Management Notes; 57, 14-17. Clean
Burke, E.J., S.J. Brown and N. Christidis, 2006: Modelling the recent evolution of global drought and projections for the 21st century with the Hadley Centre climate model. J. Hydrometeorol., 7, 113-1125. [JoC, MoS]
Burkett, V.R., D.A. Wilcox, R. Stottlemyer, W. Barrow, D. Fagre, J. Baron, J. Price, J.L. Nielsen, C.D. Allen, D.L. Peterson, G. Ruggerone and T. Doyle, 2005: Nonlinear dynamics in ecosystem response to climatic change: case studies and policy implications. Ecol. Complex., 2, 357-394. [SRC]
Busch, G., 2006: Future European agricultural landscapes: what can we learn from existing quantitative land use scenario studies? Agr. Ecosyst. Environ., 114, 121-140. [MoS]
Bush, M.B., M.R. Silman and D.H. Urrego, 2004: 48,000 years of climate and forest change in a biodiversity hot spot. Science, 303, 827-829. [JoC]
Butler, R.W., T.D. Williams, N. Warnock and M. Bishop, 1997: Wind assistance: a requirement for shorebird migration. Auk, 114, 456-466. Clean
Cairns, D.M. and J. Moen, 2004: Herbivory influences tree lines. J. Ecol., 92, 1019-1024. Clean
Caldeira, K. and M.E. Wickett, 2003: Anthropogenic carbon and ocean pH. Nature, 425, 365-365. [JoC]
Callaghan, T.V., L.O. Bjorn, Y. Chernov, T. Chapin, T.R. Christensen, B. Huntley, R.A. Ims, M. Johansson, D. Jolly, S. Jonasson, N. Matveyeva, N. Panikov, W. Oechel and G. Shaver, 2004a: Effects on the function of arctic ecosystems in the short- and long-term perspectives. Ambio, 33, 448-458. [SRC]
Callaghan, T.V., L.O. Bjorn, Y. Chernov, T. Chapin, T.R. Christensen, B. Huntley, R.A. Ims, M. Johansson, D. Jolly, S. Jonasson, N. Matveyeva, N. Panikov, W. Oechel, G. Shaver and H. Henttonen, 2004b: Effects on the structure of arctic ecosystems in the short- and long-term perspectives. Ambio, 33, 436-447. [SRC]
Callaghan, T.V., L.O. Bjorn, Y. Chernov, T. Chapin, T.R. Christensen, B. Huntley, R.A. Ims, M. Johansson, D. Jolly, S. Jonasson, N. Matveyeva, N. Panikov, W. Oechel, G. Shaver, S. Schaphoff and S. Sitch, 2004c: Effects of changes in climate on landscape and regional processes, and feedbacks to the climate system. Ambio, 33, 459-468. [SRC]
Callaghan, T.V., L.O. Björn, F.S. Chapin III, Y. Chernov, T.R. Christensen, B. Huntley, R. Ims, M. Johansson, D.J. Riedlinger, S. Jonasson, N. Matveyeva, W. Oechel, N. Panikov and G. Shaver, 2005: Arctic tundra and polar desert ecosystems. Arctic Climate Impact Assessment (ACIA): Scientific Report, C. Symon, L. Arris and B. Heal, Eds., Cambridge University Press, Cambridge, 243-352. [NPR, SRC]
Camarero, J.J., E. Gutierrez and M.J. Fortin, 2000: Boundary detection in altitudinal treeline ecotones in the Spanish central Pyrenees. Arct. Antarct. Alp. Res., 32, 117-126. Clean
Camill, P., 2005: Permafrost thaw accelerates in boreal peatlands during late-20th century climate warming. Climatic Change, 68, 135-152. [JoC]
Camill, P. and J.S. Clark, 2000: Long-term perspectives on lagged ecosystem responses to climate change: permafrost in boreal peatlands and the grassland/woodland boundary. Ecosystems, 3, 534-544. Clean
Canadell, J.G., P. Ciais, P. Cox and M. Heimann, 2004: Quantifying, understanding and managing the carbon cycle in the next decades. Climatic Change, 67, 147-160. [JoC, ARC]
Carpenter, S., P. Pingali, E. Bennett and M. Zurek, Eds., 2005: Ecosystems and Human Well-being: Volume 2: Scenarios. Island Press, Washington, District of Columbia, 560 pp. [NPR, ARC]
Carroll, A.L., S.W. Taylor, J. Régniêre and L. Safranyik, 2004: Effects of climate change on range expansion by the mountain pine beetle in British Columbia. Mountain Pine Beetle Symposium: Challenges and Solutions, T.L. Shore, J.E. Brooks and J.E. Stone, Eds., Natural Resources Canada, Canadian Forest Service, Pacific Forestry Centre, Victoria, British Columbia, 223-232. [NPR]
Cech, P., S. Pepin and C. Körner, 2003: Elevated CO2 reduces sap flux in mature deciduous forest trees. Oecologia, 137, 258-268. [SRC]
Cesar, H., L. Burke and L. Pet-Soede, 2003: The Economics of Worldwide Coral Reef Degradation. Cesar Environmental Economics Consulting (CEEC), Arnhem, 23 pp. [NPR]
Chanda, R., 2001: Safe-guarding the globe or basic well-being? Global environmental change issues and Southern Africa. Globalization, Democracy and Development in Africa: Challenges and Prospects, T. Assefa, S.M. Rugumamu and A.G.M. Ahmed, Eds., Organisation of Social Science Research in Eastern and Southern Africa (OSSREA), Addis Ababa, 246-262. [NPR]
Chang, R., Ed., 1997: Incommensurability, Incomparability, and Practical Reason. Harvard University Press, Cambridge, Massachusetts, 303 pp. [NPR]
Chapin, F.S. and A.M. Starfield, 1997: Time lags and novel ecosystems in response to transient climatic change in arctic Alaska. Climatic Change, 35, 449-461. [JoC]
Chapin, F.S., E.S. Zavaleta, V.T. Eviner, R.L. Naylor, P.M. Vitousek, H.L. Reynolds, D.U. Hooper, S. Lavorel, O.E. Sala, S.E. Hobbie, M.C. Mack and S. Diaz, 2000: Consequences of changing biodiversity. Nature, 405, 234-242. [JoC]
Chapin, F.S., P.A. Matson and H.A. Mooney, 2002: Principles of Terrestrial Ecosystem Ecology. Springer, New York, 436 pp. [NPR]
Chapin, F.S., T.V. Callaghan, Y. Bergeron, M. Fukuda, J.F. Johnstone, G. Juday and S.A. Zimov, 2004: Global change and the boreal forest: thresholds, shifting states or gradual change? Ambio, 33, 361-365. [SRC]
Chapin, F.S., III, M. Berman, T.V. Callaghan, P. Convey, A.S. Crépin, K. Danell, H. Ducklow, B. Forbes, G. Kofinas, A.D. McGuire, M. Nuttall, R. Virginia, O. Young and S.A. Zimov, 2005a: Polar systems. Ecosystems and Human Well-being: Volume 1: Current State and Trends, R. Hassan, R. Scholes and N. Ash, Eds., Island Press, Washington, District of Columbia, 717-746. [NPR, SRC]
Chapin, F.S., M. Sturm, M.C. Serreze, J.P. McFadden, J.R. Key, A.H. Lloyd, A.D. McGuire, T.S. Rupp, A.H. Lynch, J.P. Schimel, J. Beringer, W.L. Chapman, H.E. Epstein, E.S. Euskirchen, L.D. Hinzman, G. Jia, C.L. Ping, K.D. Tape, C.D.C. Thompson, D.A. Walker and J.M. Welker, 2005b: Role of land-surface changes in arctic summer warming. Science, 310, 657-660. [JoC]
Chapuis, J.L., Y. Frenot and M. Lebouvier, 2004: Recovery of native plant communities after eradication of rabbits from the subantarctic Kerguelen Islands, and influence of climate change. Biol. Conserv., 117, 167-179. Clean
Chauhan, M. and B. Gopal, 2001: Biodiversity and management of Keoladeo National Park (India): a wetland of international importance. Biodiversity in Wetlands: Assessment, Function and Conservation: Volume 2, B. Gopal, W.J. Junk and J.A. Davies, Eds., Backhuys Publishers, Leiden, 217-256. [NPR, SRC]
Cheddadi, R., J. Guiot and D. Jolly, 2001: The Mediterranean vegetation: what if the atmospheric CO2 increased? Landscape Ecol., 16, 667-675. Clean
Chidumayo, E.N., 2001: Climate and phenology of savanna vegetation in southern Africa. J. Veg. Sci., 12, 347-354. Clean
Chopra, K., R. Leemans, P. Kumar and H. Simons, Eds., 2005: Ecosystems and Human Well-being: Volume 3: Policy Responses. Island Press, Washington, District of Columbia, 621 pp. [NPR, SRC]
Chornesky, E.A. and J.M. Randall, 2003: The threat of invasive alien species to biological diversity: setting a future course. Ann. Mo. Bot. Gard., 90, 67-76. [MoS]
Christensen, J., T. Carter and F. Giorgi, 2002: PRUDENCE employs new methods to assess European climate change. EOS Transactions, 83, 147. [ARC]
Christensen, T.R., T.R. Johansson, H.J. Akerman, M. Mastepanov, N. Malmer, T. Friborg, P. Crill and B.H. Svensson, 2004: Thawing sub-arctic permafrost: effects on vegetation and methane emissions. Geophys. Res. Lett., 31, L04501, doi:10.1029/2003GL018680. [JoC]
Christensen, J.H., B. Hewitson, A. Busuioc, A. Chen, X. Gao, I. Held, R. Jones, W.-T. Kwon, R. Laprise, V.M. Rueda, L. Mearns, C.G. Menéndez, J. Räisänen, A. Rinke, R.K. Kolli, A. Sarr and P. Whetton, 2007: Regional climate projections. Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, S. Solomon, D. Qin, M. Manning, Z. Chen, M. Marquis, K.B. Averyt, M. Tignor and H.L. Miller, Eds., Cambridge University Press, Cambridge, 847-940. [NPR, ARC, 2007]
Chu, C., N.E. Mandrak and C.K. Minns, 2005: Potential impacts of climate change on the distributions of several common and rare freshwater fishes in Canada. Divers. Distrib., 11, 299-310. [ARC]
Chytry, M., P. Pysek, L. Tichy, I. Knollová and J. Danihelka, 2005: Invasions by alien plants in the Czech Republic: a quantitative assessment across habitats. Preslia, 77, 339-354. Clean
Ciais, P., M. Reichstein, N. Viovy, A. Granier, J. Ogee, V. Allard, M. Aubinet, N. Buchmann, Chr. Bernhofer, A. Carrara, F. Chevallier, N. De Noblet, A.D. Friend, P. Friedlingstein, T. Grünwald, B. Heinesch, P. Keronen, A. Knohl, G. Krinner, D. Loustau, G. Manca, G. Matteucci, F. Miglietta, J.M. Ourcival, D. Papale, K. Pilegaard, S. Rambal, G. Seufert, J.F. Soussana, M.J. Sanz, E.D. Schulze, T. Vesala and R. Valentini, 2005: Europe-wide reduction in primary productivity caused by the heat and drought in 2003. Nature, 437, 529-533. [JoC, ARC]
Clark, J.S., 1998: Why trees migrate so fast: confronting theory with dispersal biology and the paleorecord. Am. Nat., 152, 204-224. Clean
Clark, J.S., M. Lewis and L. Horvath, 2001: Invasion by extremes: population spread with variation in dispersal and reproduction. Am. Nat., 157, 537-554. Clean
Clark, J.S., M. Lewis, J.S. McLachlan and J. Hillerislambers, 2003: Estimating population spread: what can we forecast and how well? Ecology, 84, 1979-1988. [MoS]
Clark, R.A. and C.L.J. Frid, 2001: Long-term changes in the North Sea ecosystem. Environ. Rev., 9, 131-187. Clean
Cochrane, M.A., 2003: Fire science for rainforests. Nature, 421, 913-919. [JoC]
Cochrane, M.A. and W.F. Laurance, 2002: Fire as a large-scale edge effect in Amazonian forests. J. Trop. Ecol., 18, 311-325. Clean
Cohen, A.S., 2003: Paleolimnology: The History and Evolution of Lake Systems. Oxford University Press, Oxford, 528 pp. [NPR]
Cole, J., 2003: Global change: dishing the dirt on coral reefs. Nature, 421, 705-706. [JoC]
Coles, S.L. and B.E. Brown, 2003: Coral bleaching: capacity for acclimatization and adaptation. Adv. Mar. Biol., 46, 183-223. [ARC]
Collingham, Y.C. and B. Huntley, 2000: Impacts of habitat fragmentation and patch size upon migration rates. Ecol. Appl., 10, 131-144. Clean
Cornelissen, J.H.C., T.V. Callaghan, J.M. Alatalo, A. Michelsen, E. Graglia, A.E. Hartley, D.S. Hik, S.E. Hobbie, M.C. Press, C.H. Robinson, G.H.R. Henry, G.R. Shaver, G.K. Phoenix, D.G. Jones, S. Jonasson, F.S. Chapin, U. Molau, C. Neill, J.A. Lee, J.M. Melillo, B. Sveinbjornsson and R. Aerts, 2001: Global change and arctic ecosystems: is lichen decline a function of increases in vascular plant biomass? J. Ecol., 89, 984-994. [SRC]
Costanza, R., 2000: Social goals and the valuation of ecosystem services. Ecosystems, 3, 4-10. Clean
Costanza, R., 2001: Visions, values, valuation, and the need for an ecological economics. BioScience, 51, 459-468. Clean
Costanza, R., R. D’Arge, R. de Groot, S. Farber, M. Grasso, B. Hannon, K. Limburg, S. Naeem, R.V. O’Neill, J. Paruelo, R.G. Raskin, P. Sutton and M. van den Belt, 1997: The value of the world’s ecosystem services and natural capital. Nature, 387, 253-260. [JoC]
Costanza, R., M. Daly, C. Folke, P. Hawken, C.S. Holling, A.J. McMichael, D. Pimentel and D. Rapport, 2000: Managing our environmental portfolio. BioScience, 50, 149-155. [ARC]
Cowling, R.M., P.W. Rundel, B.B. Lamont, M.K. Arroyo and M. Arianoutsou, 1996: Plant diversity in Mediterranean-climate regions. Trends Ecol. Evol., 11, 362-366. Clean
Cowling, R.M., R.L. Pressey, A.T. Lombard, P.G. Desmet and A.G. Ellis, 1999: From representation to persistence: requirements for a sustainable system of conservation areas in the species-rich Mediterranean-climate desert of southern Africa. Divers. Distrib., 5, 51-71. Clean
Cowling, R.M., F. Ojeda, B. Lamont, P.W. Rundel and R. Lechmere-Oertel, 2005: Rainfall reliability, a neglected factor in explaining convergence and divergence of plant traits in fire-prone Mediterranean-climate ecosystems. Global Ecol. Biogeogr., 14, 509-519. Clean
Cox, P.M., R.A. Betts, C.D. Jones, S.A. Spall and I.J. Totterdell, 2000: Acceleration of global warming due to carbon-cycle feedbacks in a coupled climate model. Nature, 408, 184-187. [JoC, MoS, ARC]
Cox, P.M., R.A. Betts, M. Collins, P.P. Harris, C. Huntingford and C.D. Jones, 2004: Amazonian forest dieback under climate-carbon cycle projections for the 21st century. Theor. Appl. Climatol., 78, 137-156. [MoS, ARC]
Cox, P.M., C. Huntingford and C.D. Jones, 2006: Conditions for sink-to-source transitions and runaway feedbacks from the land carbon cycle. Avoiding Dangerous Climate Change, H.J. Schellnhuber, W. Cramer, N. Naki?enovi?, T.M.L. Wigley and G. Yohe, Eds., Cambridge University Press, Cambridge, 155-161. [NPR]
Craig, P., P. Trail and T.E. Morrell, 1994: The decline of fruit bats in American-Samoa due to hurricanes and overhunting. Biol. Conserv., 69, 261-266. [ARC]
Cramer, W., A. Bondeau, F.I. Woodward, I.C. Prentice, R.A. Betts, V. Brovkin, P.M. Cox, V. Fisher, J.A. Foley, A.D. Friend, C. Kucharik, M.R. Lomas, N. Ramankutty, S. Sitch, B. Smith, A. White and C. Young-Molling, 2001: Global response of terrestrial ecosystem structure and function to CO2 and climate change: results from six dynamic global vegetation models. Global Change Biol., 7, 357-373. [JoC, MoS, SRC]
Cramer, W., A. Bondeau, S. Schaphoff, W. Lucht, B. Smith and S. Sitch, 2004: Tropical forests and the global carbon cycle: impacts of atmospheric carbon dioxide, climate change and rate of deforestation. Philos. T. Roy. Soc. Lond. B, 359, 331-343. [SRC]
Crawford, R.M.M., C.E. Jeffree and W.G. Rees, 2003: Paludification and forest retreat in northern oceanic environments. Ann. Bot., 91, 213-226. Clean
Crawley, M.J., 1989: Chance and timing in biological invasions. Biological Invasions: A Global Perspective, J. Drake, F. Dicastri, R. Groves, F. Kruger, H. Mooney, M. Rejmanek and M. Williamson, Eds., John Wiley, Chichester, 407-423. [NPR]
Crick, H.Q.P., 2004: The impact of climate change on birds. Ibis, 146, 48-56. Clean
Cropp, R. and A. Gabrica, 2002: Ecosystem adaptation: do ecosystems maximize resilience? Ecology, 83, 2019-2026. Clean
Croxall, J.P., P.N. Trathan and E.J. Murphy, 2002: Environmental change and Antarctic seabird populations. Science, 297, 1510-1514. [JoC]
Cubasch, U., G.A. Meehl, G.J. Boer, R.J. Stouffer, M. Dix, A. Noda, C.A. Senior, S. Raper and K.S. Yap, 2001: Projections of future climate change. Climate Change 2001: The Scientific Basis. Contribution of Working Group I to the Third Assessment Report of the Intergovernmental Panel on Climate Change, J.T. Houghton, Y. Ding, D.J. Griggs, M. Noguer, P.J. van der Linden, X. Dai, K. Maskell and C.A. Johnson, Eds., Cambridge University Press, Cambridge, 525-582. [NPR, ARC]
Currie, D.J., 2001: Projected effects of climate change on patterns of vertebrate and tree species richness in the conterminous United States. Ecosystems, 4, 216-225. Clean
Daily, G.C., T. Söderqvist, S. Aniyar, K. Arrow, P. Dasgupta, P.R. Ehrlich, C. Folke, A. Jansson, B. Jansson, N. Kautsky, S. Levin, J. Lubchenco, K. Mäler, D. Simpson, D. Starrett, D. Tilman and B. Walker, 2000: The value of nature and the nature of value. Science, 289, 395-396. [JoC]
Daimaru, H. and H. Taoda, 2004: Effects of snow pressure on the distribution of subalpine Abies mariesii forests in northern Honshu Island, Japan. Journal of Agricultural Meteorology, 60, 253-261. Clean
Dale, V.H., L.A. Joyce, S. McNulty and R.P. Neilson, 2000: The interplay between climate change, forests, and disturbances. Sci. Total Environ., 262, 201-204. [SRC]
Davidson, E.A. and I.A. Janssens, 2006: Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature, 440, 165-173. [JoC]
Davis, M.B., 1989: Lags in vegetation response to greenhouse warming. Climatic Change, 15, 75-82. [JoC]
Davis, M.B. and R.G. Shaw, 2001: Range shifts and adaptive responses to Quaternary climate change. Science, 292, 673-679. [JoC]
Davis, M.B., R.G. Shaw and J.R. Etterson, 2005: Evolutionary responses to changing climate. Ecology, 86, 1704-1714. Clean
De Bono, A., P. Peduzzi, G. Giuliani and S. Kluser, 2004: Impacts of Summer 2003 Heat Wave in Europe. Early Warning on Emerging Environmental Threats 2, UNEP: United Nations Environment Programme, Nairobi, 4 pp. [NPR]
de Graaff, M.A., K.J. van Groenigen, J. Six, B. Hungate and C. van Kessel, 2006: Interactions between plant growth and soil nutrient cycling under elevated CO2: a meta-analysis. Global Change Biol., 12, 2077-2091. [JoC]
de Groot, W.J., P.M. Bothwell, D.H. Carlsson and K.A. Logan, 2003: Simulating the effects of future fire regimes on western Canadian boreal forests. J. Veg. Sci., 14, 355-364. [MoS]
de Koning, F., R. Olschewski, E. Veldkamp, P. Benitez, M. Lopez-Ulloa, T. Schlichter and M. de Urquiza, 2005: The ecological and economic potential of carbon sequestration in forests: examples from South America. Ambio, 34, 224-229. Clean
de Wit, M. and J. Stankiewicz, 2006: Changes in surface water supply across Africa with predicted climate change. Science, 311, 1917-1921. [JoC, MoS]
Del Barrio, G., P.A. Harrison, P.M. Berry, N. Butt, M. Sanjuan, R.G. Pearson and T.P. Dawson, 2006: Integrating multiple modelling approaches to predict the potential impacts of climate change on species’ distributions in contrasting regions: comparison and implications for policy. Environ. Sci. Policy, 9, 129-147. [MoS]
Denman, K.L., G. Brasseur, A. Chidthaisong, P. Ciais, P. Cox, R.E. Dickinson, D. Hauglustaine, C. Heinze, E. Holland, D. Jacob, U. Lohmann, S. Ramachandran, P.L. da Silva Dias, S.C. Wofsy and X. Zhang, 2007: Couplings between changes in the climate system and biogeochemistry. Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, S. Solomon, D. Qin, M. Manning, Z. Chen, M. Marquis, K.B. Averyt, M. Tignor and H.L. Miller, Eds., Cambridge University Press, Cambridge, 499-587. [NPR, 2007]
Derocher, A.E., N.J. Lunn and I. Stirling, 2004: Polar bears in a warming climate. Integr. Comp. Biol., 44, 163-176. [SRC]
Deutschewitz, K., A. Lausch, I. Kuhn and S. Klotz, 2003: Native and alien plant species richness in relation to spatial heterogeneity on a regional scale in Germany. Global Ecol. Biogeogr., 12, 299-311. Clean
Diaz, H.F., Ed., 2003: Climate Variability and Change in High Elevation Regions: Past, Present and Future. Kluwer Academic, Dordrecht, 282 pp. [NPR, MoS]
Diaz, H.F. and V. Markgraf, Eds., 1992: El Niño Historical and Paleoclimatic Aspects of the Southern Oscillation. Cambridge University Press, Cambridge, 476 pp. [NPR]
Diaz, S., J. Fargione, F.S. Chapin and D. Tilman, 2006: Biodiversity loss threatens human well-being. PLoS Biol., 4, 1300-1305. Clean
Dirnböck, T., S. Dullinger and G. Grabherr, 2003: A regional impact assessment of climate and land-use change on alpine vegetation. J. Biogeogr., 30, 401-417. Clean
Donner, S.D., W.J. Skirving, C.M. Little, M. Oppenheimer and O. Hoegh-Guldberg, 2005: Global assessment of coral bleaching and required rates of adaptation under climate change. Global Change Biol., 11, 2251-2265. [JoC, ARC]
Dormann, C.F. and S.J. Woodin, 2002: Climate change in the Arctic: using plant functional types in a meta-analysis of field experiments. Funct. Ecol., 16, 4-17. Clean
Dube, O.P. and G. Pickup, 2001: Effects of rainfall variability and communal and semi-commercial grazing on land cover in southern African rangelands. Climate Res., 17, 195-208. Clean
Dufresne, J.L., P. Friedlingstein, M. Berthelot, L. Bopp, P. Ciais, L. Fairhead, H. Le Treut and P. Monfray, 2002: On the magnitude of positive feedback between future climate change and the carbon cycle. Geophys. Res. Lett., 29, 1405, doi:10.1029/2001GL013777. [JoC, MoS]
Duguay, C.R., T.D. Prowse, B.R. Bonsal, R.D. Brown, M.P. Lacroix and P. Menard, 2006: Recent trends in Canadian lake ice cover. Hydrol. Process., 20, 781-801. [ARC]
Dukes, J.S. and H.A. Mooney, 1999: Does global change increase the success of biological invaders? Trends Ecol. Evol., 14, 135-139. Clean
Dukes, J.S., N.R. Chiariello, E.E. Cleland, L.A. Moore, M.R. Shaw, S. Thayer, T. Tobeck, H.A. Mooney and C.B. Field, 2005: Responses of grassland production to single and multiple global environmental changes. PLoS Biol., 3, 1829-1837. [ARC]
Dullinger, S., T. Dirnbock and G. Grabherr, 2004: Modelling climate change-driven treeline shifts: relative effects of temperature increase, dispersal and invasibility. J. Ecol., 92, 241-252. [MoS]
Dullinger, S., T. Dirnbock, R. Kock, E. Hochbichler, T. Englisch, N. Sauberer and G. Grabherr, 2005: Interactions among tree-line conifers: differential effects of pine on spruce and larch. J. Ecol., 93, 948-957. Clean
Duraiappah, A., S. Naeem, T. Agardi, N. Ash, D. Cooper, S. Díaz, D.P. Faith, G. Mace, J.A. McNeilly, H.A. Mooney, A.A. Oteng-Yeboah, H.M. Pereira, S. Polasky, C. Prip, W.V. Reid, C. Samper, P.J. Schei, R. Scholes, F. Schutyser and A. van Jaarsveld, Eds., 2005: Ecosystems and Human Well-being: Biodiversity Synthesis. Island Press, Washington, District of Columbia, 100 pp. [NPR]
Durieux, L., L.A.T. Machado and H. Laurent, 2003: The impact of deforestation on cloud cover over the Amazon arc of deforestation. Remote Sens. Environ., 86, 132-140. Clean
Eastman, J.L., M.B. Coughenour and R.A. Pielke, 2001: The regional effects of CO2 and landscape change using a coupled plant and meteorological model. Global Change Biol., 7, 797-815. [JoC, MoS]
ECF, Ed., 2004: What is Dangerous Climate Change? Initial Results of a Symposium on Key Vulnerable Regions Climate Change and Article 2 of the UNFCCC. International Symposium, Beijing, 27-30 October. European Climate Forum, Buenos Aires, 39 pp. [NPR]
Edwards, E.J., D.G. Benham, L.A. Marland and A.H. Fitter, 2004: Root production is determined by radiation flux in a temperate grassland community. Global Change Biol., 10, 209-227. [JoC]
Edwards, M. and A.J. Richardson, 2004: Impact of climate change on marine pelagic phenology and trophic mismatch. Nature, 430, 881-884. [JoC, ARC]
Edwards, M., G. Beaugrand, P.C. Reid, A.A. Rowden and M.B. Jones, 2002: Ocean climate anomalies and the ecology of the North Sea. Mar. Ecol.–Prog. Ser., 239, 1-10. [ARC]
Edwards, M., D.G. Johns, S.C. Leterme, E. Svendsen and A.J. Richardson, 2006: Regional climate change and harmful algal blooms in the northeast Atlantic. Limnol. Oceanogr., 51, 820-829. [ARC]
Eliasson, P.E., R.E. McMurtrie, D.A. Pepper, M. Stromgren, S. Linder and G.I. Agren, 2005: The response of heterotrophic CO2 flux to soil warming. Global Change Biol., 11, 167-181. [JoC]
Eliot, I., C.M. Finlayson and P. Waterman, 1999: Predicted climate change, sea-level rise and wetland management in the Australian wet-dry tropics. Wetlands Ecology and Management, 7, 63-81. [MoS]
Enhalt, D. and M. Prather, 2001: Atmospheric chemistry and greenhouse gases. Climate Change 2001: The Scientific Basis. Contribution of Working Group I to the Third Assessment Report of the Intergovernmental Panel on Climate Change, J.T. Houghton, Y. Ding, D.J. Griggs, M. Noguer, P.J. van der Linden, X. Dai, K. Maskell and C.A. Johnson, Eds., Cambridge University Press, Cambridge, 239-287. [NPR]
Enquist, C.A.F., 2002: Predicted regional impacts of climate change on the geographical distribution and diversity of tropical forests in Costa Rica. J. Biogeogr., 29, 519-534. [MoS]
Epstein, H.E., R.A. Gill, J.M. Paruelo, W.K. Lauenroth, G.J. Jia and I.C. Burke, 2002: The relative abundance of three plant functional types in temperate grasslands and shrublands of North and South America: effects of projected climate change. J. Biogeogr., 29, 875-888. Clean
Epstein, P.R. and E. Mills, 2005: Climate Change Futures: Health, Ecological and Economic Dimensions. Center for Health and the Global Environment, Harvard Medical School, Boston, Massachusetts, 142 pp. [NPR, MoS, ARC]
Erasmus, B.F.N., A.S. Van Jaarsveld, S.L. Chown, M. Kshatriya and K.J. Wessels, 2002: Vulnerability of South African animal taxa to climate change. Global Change Biol., 8, 679-693. [JoC]
Evans, R.D. and J. Belnap, 1999: Long-term consequences of disturbance on nitrogen dynamics in an arid ecosystem. Ecology, 80, 150-160. Clean
Facon, B., B.J. Genton, J. Shykoff, P. Jarne, A. Estoup and P. David, 2006: A general eco-evolutionary framework for understanding bioinvasions. Trends Ecol. Evol., 21, 130-135. Clean
Fahrig, L., 2003: Effects of habitat fragmentation on biodiversity. Annu. Rev. Ecol. Evol. Syst., 34, 487-515. Clean
FAO, 2001: Global forest resource assessment 2000: main report. FAO Forestry Paper 140, Food and Agriculture Organization of the United Nations, Rome, Italy, 479 pp. [NPR]
Farber, S., R. Costanza, D.L. Childers, J. Erickson, K. Gross, M. Grove, C.S. Hopkinson, J. Kahn, S. Pincetl, A. Troy, P. Warren and M. Wilson, 2006: Linking ecology and economics for ecosystem management. BioScience, 56, 121-133. Clean
Fay, P.A., J.D. Carlisle, A.K. Knapp, J.M. Blair and S.L. Collins, 2000: Altering rainfall timing and quantity in a mesic grassland ecosystem: design and performance of rainfall manipulation shelters. Ecosystems, 3, 308-319. Clean
Fay, P.A., J.D. Carlisle, B.T. Danner, M.S. Lett, J.K. McCarron, C. Stewart, A.K. Knapp, J.M. Blair and S.L. Collins, 2002: Altered rainfall patterns, gas exchange, and growth in grasses and forbs. Int. J. Plant Sci., 163, 549-557. Clean
Fay, P.A., J.D. Carlisle, A.K. Knapp, J.M. Blair and S.L. Collins, 2003: Productivity responses to altered rainfall patterns in a C-4-dominated grassland. Oecologia, 137, 245-251. Clean
Feddema, J.J., K.W. Oleson, G.B. Bonan, L.O. Mearns, L.E. Buja, G.A. Meehl and W.M. Washington, 2005: The importance of land-cover change in simulating future climates. Science, 310, 1674-1678. [JoC, MoS, ARC]
Feely, R.A., C.L. Sabine, K. Lee, W. Berelson, J. Kleypas, V.J. Fabry and F.J. Millero, 2004: Impact of anthropogenic CO2 on the CaCO3 system in the oceans. Science, 305, 362-366. [JoC]
Field, C.B., M.J. Behrenfeld, J.T. Randerson and P. Falkowski, 1998: Primary production of the biosphere: integrating terrestrial and oceanic components. Science, 281, 237-240. [JoC, ARC]
Fink, A.H., T. Brücher, A. Krüger, G.C. Leckebusch, J.G. Pinto and U. Ulbrich, 2004: The 2003 European summer heatwaves and drought: synoptic diagnosis and impacts. Weather, 59, 209-216. Clean
Finlayson, C.M., R. D’Cruz, N. Davidson, J. Alder, S. Cork, R. de Groot, C. Lévêque, G.R. Milton, G. Peterson, D. Pritchard, B.D. Ratner, W.V. Reid, C. Revenga, M. Rivera, F. Schutyser, M. Siebentritt, M. Stuip, R. Tharme, S. Butchart, E. Dieme-Amting, H. Gitay, S. Raaymakers and D. Taylor, Eds., 2005: Ecosystems and Human Well-being: Wetlands and Water Synthesis. Island Press, Washington, District of Columbia, 80 pp. [NPR]
Fischer, R., Ed., 2005: The condition of forests in Europe: 2005 executive report. United Nations Economic Commission for Europe (UN-ECE), Geneva, 36 pp. [NPR]
Fischlin, A. and D. Gyalistras, 1997: Assessing impacts of climatic change on forests in the Alps. Global Ecol. Biogeogr. Lett., 6, 19-37. [SRC]
Fish, M.R., I.M. Cote, J.A. Gill, A.P. Jones, S. Renshoff and A.R. Watkinson, 2005: Predicting the impact of sea-level rise on Caribbean sea turtle nesting habitat. Conserv. Biol., 19, 482-491. [MoS, ARC]
Flanagan, L.B., L.A. Wever and P.J. Carlson, 2002: Seasonal and interannual variation in carbon dioxide exchange and carbon balance in a northern temperate grassland. Global Change Biol., 8, 599-615. [JoC]
Flannigan, M.D., B.J. Stocks and B.M. Wotton, 2000: Climate change and forest fires. Sci. Total Environ., 262, 221-229. Clean
Fleming, R.A., J.N. Candau and R.S. McAlpine, 2002: Landscape-scale analysis of interactions between insect defoliation and forest fire in Central Canada. Climatic Change, 55, 251-272. [JoC]
Flöder, S. and C.W. Burns, 2004: Phytoplankton diversity of shallow tidal lakes: influence of periodic salinity changes on diversity and species number of a natural assemblage. J. Phycol., 40, 54-61. Clean
Florenzano, G.T., 2004: Birds as indicators of recent environmental changes in the Apennines (Foreste Casentinesi National Park, central Italy). Ital. J. Zool., 71, 317-324. Clean
Foley, J.A., M.T. Coe, M. Scheffer and G.L. Wang, 2003: Regime shifts in the Sahara and Sahel: interactions between ecological and climatic systems in northern Africa. Ecosystems, 6, 524-539. Clean
Foley, J.A., R. Defries, G.P. Asner, C. Barford, G. Bonan, S.R. Carpenter, F.S. Chapin, M.T. Coe, G.C. Daily, H.K. Gibbs, J.H. Helkowski, T. Holloway, E.A. Howard, C.J. Kucharik, C. Monfreda, J.A. Patz, I.C. Prentice, N. Ramankutty and P.K. Snyder, 2005: Global consequences of land use. Science, 309, 570-574. [JoC, ARC]
Folke, C., S. Carpenter, B. Walker, M. Scheffer, T. Elmqvist, L. Gunderson and C.S. Holling, 2004: Regime shifts, resilience, and biodiversity in ecosystem management. Annu. Rev. Ecol. Evol. Syst., 35, 557-581. Clean
Folkestad, T., M. New, J.O. Kaplan, J.C. Comiso, S. Watt-Cloutier, T. Fenge, P. Crowley and L.D. Rosentrater, 2005: Evidence and implications of dangerous climate change in the Arctic. Avoiding Dangerous Climate Change, H.J. Schellnhuber, W. Cramer, N. Naki?enovi?, T.M.L. Wigley and G. Yohe, Eds., Cambridge University Press, Cambridge, 215-218. [NPR]
Forbes, B.C., 1995: Effects of surface disturbance on the movement of native and exotic plants under a changing climate. Global Change and Arctic Terrestrial Ecosystems, T.V. Callaghan, Ed., European Commission, Directorate-General, Science, Research and Development, Brussels, 209-219. [NPR]
Forcada, J., P.N. Trathan, K. Reid, E.J. Murphy and J.P. Croxall, 2006: Contrasting population changes in sympatric penguin species in association with climate warming. Global Change Biol., 12, 411-423. [JoC]
Forster, P., V. Ramaswamy, P. Araxo, T. Berntsen, R.A. Betts, D.W. Fahey, J. Haywood, J. Lean, D.C. Lowe, G. Myhre, J. Nganga, R. Prinn, G. Raga, M. Schulze and R. Van Dorland, 2007: Changes in atmospheric constituents and in radiative forcing. Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, S. Solomon, D. Qin, M. Manning, Z. Chen, M. Marquis, K.B. Averyt, M. Tignor and H.L. Miller, Eds., Cambridge University Press, Cambridge, 130-234. [NPR, ARC, 2007]
Foster, P., 2001: The potential negative impacts of global climate change on tropical montane cloud forests. Earth Sci. Rev., 55, 73-106. Clean
Freeman, C., N. Fenner, N.J. Ostle, H. Kang, D.J. Dowrick, B. Reynolds, M.A. Lock, D. Sleep, S. Hughes and J. Hudson, 2004: Export of dissolved organic carbon from peatlands under elevated carbon dioxide levels. Nature, 430, 195-198. [JoC]
Freiwald, A., J.H. Fossa, A. Grehan, T. Koslow and J.M. Roberts, 2004: Cold-water coral reefs: out of sight no longer out of mind. UNEP-WCMC No. 22, UNEP World Conservation Monitoring Centre, Cambridge, 86 pp. [NPR]
Frey, K.E. and L.C. Smith, 2005: Amplified carbon release from vast West Siberian peatlands by 2100. Geophys. Res. Lett., 32, L09401, doi:10.1029/2004GL02202. [JoC]
Fried, J.S., M.S. Torn and E. Mills, 2004: The impact of climate change on wildfire severity: a regional forecast for northern California. Climatic Change, 64, 169-191. [JoC, MoS, ARC]
Friedlingstein, P., J.L. Dufresne, P.M. Cox and P. Rayner, 2003: How positive is the feedback between climate change and the carbon cycle? Tellus B, 55, 692-700. Clean
Friedlingstein, P., P. Cox, R. Betts, L. Bopp, W. Von Bloh, V. Brovkin, P. Cadule, S. Doney, M. Eby, I. Fung, G. Bala, J. John, C. Jones, F. Joos, T. Kato, M. Kawamiya, W. Knorr, K. Lindsay, H.D. Matthews, T. Raddatz, P. Rayner, C. Reick, E. Roeckner, K.-G. Schnitzler, R. Schnur, K. Strassmann, A.J. Weaver, C. Yoshikawa and N. Zeng, 2006: Climate-carbon cycle feedback analysis: results from the (CMIP)-M-4 model intercomparison. J. Climate, 19, 3337-3353. [PoC, JoC, MoS, ARC]
Fuhlendorf, S.D., D.D. Briske and F.E. Smeins, 2001: Herbaceous vegetation change in variable rangeland environments: The relative contribution of grazing and climatic variability. Appl. Veg. Sci, 4, 177-188. Clean
Fuhrer, J., M. Beniston, A. Fischlin, C. Frei, S. Goyette, K. Jasper and C. Pfister, 2006: Climate risks and their impact on agriculture and forests in Switzerland. Climatic Change, 79, 79-102. [JoC, SRC]
Galbraith, H., R. Jones, R. Park, J. Clough, S. Herrod-Julius, B. Harrington and G. Page, 2002: Global climate change and sea level rise: potential losses of intertidal habitat for shorebirds. Waterbirds, 25, 173-183. [ARC]
Gamache, I. and S. Payette, 2005: Latitudinal response of subarctic tree lines to recent climate change in eastern Canada. J. Biogeogr., 32, 849-862. Clean
Gan, J.B., 2004: Risk and damage of southern pine beetle outbreaks under global climate change. Forest Ecol. Manag., 191, 61-71. Clean
Gardner, T.A., I.M. Cote, J.A. Gill, A. Grant and A.R. Watkinson, 2003: Long-term region-wide declines in Caribbean corals. Science, 301, 958-960. [JoC, ARC]
Gaston, K.J., T.M. Blackburn and K.K. Goldewijk, 2003: Habitat conversion and global avian biodiversity loss. P. Roy. Soc. Lond. B, 270, 1293-1300. Clean
Gedney, N., P.M. Cox, R.A. Betts, O. Boucher, C. Huntingford and P.A. Stott, 2006: Detection of a direct carbon dioxide effect in continental river runoff records. Nature, 439, 835-838. [JoC, ARC]
Geist, H.J. and E.F. Lambin, 2004: Dynamic causal patterns of desertification. BioScience, 54, 817-829. Clean
Genner, M.J., D.W. Sims, V.J. Wearmouth, E.J. Southall, A.J. Southward, P.A. Henderson and S.J. Hawkins, 2004: Regional climatic warming drives long-term community changes of British marine fish. P. Roy. Soc. Lond. B, 271, 655-661. Clean
Gerber, S., F. Joos and I.C. Prentice, 2004: Sensitivity of a dynamic global vegetation model to climate and atmospheric CO2. Global Change Biol., 10, 1223-1239. [PoC, JoC, MoS]
Gerten, D. and R. Adrian, 2002: Species-specific changes in the phenology and peak abundance of freshwater copepods in response to warm summers. Freshwater Biol., 47, 2163-2173. Clean
Gerten, D., S. Schaphoff, U. Haberlandt, W. Lucht and S. Sitch, 2004: Terrestrial vegetation and water balance: hydrological evaluation of a dynamic global vegetation model. J. Hydrol., 286, 249-270. [MoS, SRC]
Gerten, D., W. Lucht, S. Schaphoff, W. Cramer, T. Hickler and W. Wagner, 2005: Hydrologic resilience of the terrestrial biosphere. Geophys. Res. Lett., 32, L21408, doi:10.1029/2005GL024247. [JoC, SRC]
Gielen, B., H.J. De Boeck, C.M.H.M. Lemmens, R. Valcke, I. Nijs and R. Ceulemans, 2005: Grassland species will not necessarily benefit from future elevated air temperatures: a chlorophyll fluorescence approach to study autumn physiology. Physiol. Plant., 125, 52-63. [MoS, ARC]
Giles, J., 2005: Millennium group nails down the financial value of ecosystems. Nature, 434, 547. [JoC]
Gillett, N.P., A.J. Weaver, F.W. Zwiers and M.D. Flannigan, 2004: Detecting the effect of climate change on Canadian forest fires. Geophys. Res. Lett., 31, L18211, doi:10.1029/2004GL020876. [JoC]
Gitay, H., S. Brown, W. Easterling and B. Jallow, 2001: Ecosystems and their goods and services. Climate Change 2001: Impacts, Adaptation, and Vulnerability. Contribution of Working Group II to the Third Assessment Report of the Intergovernmental Panel on Climate Change, J.J. McCarthy, O.F. Canziani, N.A. Leary, D.J. Dokken and K.S. White, Eds., Cambridge University Press, Cambridge, 237-342. [NPR, ARC]
Gitay, H., A. Suárez and R.T. Watson, 2002: Climate Change and Biodiversity. IPCC Technical Paper, Intergovernmental Panel on Climate Change, Geneva, 77 pp. [NPR, ARC]
Gitz, V. and P. Ciais, 2003: Amplifying effects of land-use change on future atmospheric CO2 levels. Global Biogeochem. Cy., 17, 1024-1038, doi:10.1029/2002GB001963. [MoS]
Gobron, N., B. Pinty, F. Melin, M. Taberner, M.M. Verstraete, A. Belward, T. Lavergne and J.L. Widlowski, 2005: The state of vegetation in Europe following the 2003 drought. Int. J. Remote Sens., 26, 2013-2020. Clean
Goklany, I.M., 1998: Saving habitat and conserving biodiversity on a crowded planet. BioScience, 48, 941-953. Clean
Goklany, I.M., 2005: A climate policy for the short and medium term: stabilization or adaptation? Energ. Environ., 16, 667-680. Clean
Goklany, I.M. and A.J. Trewavas, 2003: How technology can reduce our impact on the Earth. Nature, 423, 115-115. [JoC]
Goldstein, A.H., N.E. Hultman, J.M. Fracheboud, M.R. Bauer, J.A. Panek, M. Xu, Y. Qi, A.B. Guenther and W. Baugh, 2000: Effects of climate variability on the carbon dioxide, water and sensible heat fluxes above a ponderosa pine plantation in the Sierra Nevada (CA). Agr. Forest Meteorol., 101, 113-129. Clean
Gonzalez, P., 2001: Desertification and a shift of forest species in the West African Sahel. Climate Res., 17, 217-228. Clean
Gopal, B. and M. Chauhan, 2001: South Asian wetlands and their biodiversity: the role of monsoons. Biodiversity in Wetlands: Assessment, Function and Conservation: Volume 2, B. Gopal, W.J. Junk and J.A. Davis, Eds., Backhuys Publishers, Leiden, 257-275. [NPR, SRC]
Gottfried, M., H. Pauli, K. Reiter and G. Grabherr, 1999: A fine-scaled predictive model for changes in species distribution patterns of high mountain plants induced by climate warming. Divers. Distrib., 5, 241-251. [MoS]
Grace, J., F. Berninger and L. Nagy, 2002: Impacts of climate change on the tree line. Ann. Bot., 90, 537-544. Clean
Graglia, E., R. Julkunen-Tiitto, G.R. Shaver, I.K. Schmidt, S. Jonasson and A. Michelsen, 2001: Environmental control and intersite variations of phenolics in Betula nana in tundra ecosystems. New Phytol., 151, 227-236. Clean
Gruber, N., P. Friedlingstein, C.B. Field, R. Valentini, M. Heimann, J.F. Richey, P. Romero, E.D. Schulze and A. Chen, 2004: The vulnerability of the carbon cycle in the 21st century: an assessment of carbon-climate-human interactions. Global Carbon Cycle: Integrating Humans, Climate, and the Natural World, C.B. Field and M.R. Raupach, Eds., Island Press, Washington, District of Columbia, 45-76. [NPR, ARC]
Guinotte, J.M., R.W. Buddemeier and J.A. Kleypas, 2003: Future coral reef habitat marginality: temporal and spatial effects of climate change in the Pacific basin. Coral Reefs, 22, 551-558. [MoS]
Guinotte, J.M., J. Orr, S. Cairns, A. Freiwald, L. Morgan and R. George, 2006: Will human-induced changes in seawater chemistry alter the distribution of deep-sea scleractinian corals? Front. Ecol. Environ., 4, 141-146. Clean
Guisan, A. and N.E. Zimmermann, 2000: Predictive habitat distribution models in ecology. Ecol. Model., 135, 147-186. [MoS, SRC]
Guisan, A. and W. Thuiller, 2005: Predicting species distribution: offering more than simple habitat models. Ecol. Lett., 8, 993-1009. [MoS, SRC]
Gyalistras, D. and A. Fischlin, 1999: Towards a general method to construct regional climatic scenarios for model-based impacts assessments. Petermann. Geogr. Mitt., 143, 251-264. [MoS, SRC]
Gyalistras, D., H. von Storch, A. Fischlin and M. Beniston, 1994: Linking GCM-simulated climatic changes to ecosystem models: case studies of statistical downscaling in the Alps. Climate Res., 4, 167-189. [MoS, SRC]
Hallock, P., 2005: Global change and modern coral reefs: new opportunities to understand shallow-water carbonate depositional processes. Sediment. Geol., 175, 19-33. Clean
Halloy, S.R.P. and A.F. Mark, 2003: Climate-change effects on alpine plant biodiversity: a New Zealand perspective on quantifying the threat. Arct. Antarct. Alp. Res., 35, 248-254. Clean
Hamerlynck, E.P., T.E. Huxman, T.N. Charlet and S.D. Smith, 2002: Effects of elevated CO2 (FACE) on the functional ecology of the drought-deciduous Mojave Desert shrub, Lycium andersonii. Environ. Exp. Bot., 48, 93-106. [NPR]
Handel, M.D. and J.S. Risbey, 1992: Reflections on more than a century of climate change research. Climatic Change, 21, 91-96. [JoC]
Hanley, M.E., S. Trofimov and G. Taylor, 2004: Species-level effects more important than functional group-level responses to elevated CO2: evidence from simulated turves. Funct. Ecol., 18, 304-313. [MoS]
Hannah, L. and T.E. Lovejoy, 2003: Climate change and biodiversity: synergistic impacts. Advances in Applied Biodiversity Science, 4, 1-123. Clean
Hannah, L., G.F. Midgley, T. Lovejoy, W.J. Bond, M. Bush, J.C. Lovett, D. Scott and F.I. Woodward, 2002a: Conservation of biodiversity in a changing climate. Conserv. Biol., 16, 264-268. [SRC]
Hannah, L., G.F. Midgley and D. Millar, 2002b: Climate change-integrated conservation strategies. Global Ecol. Biogeogr., 11, 485-495. [SRC]
Hänninen, H., 2006: Climate warming and the risk of frost damage to boreal forest trees: identification of critical ecophysiological traits. Tree Physiol., 26, 889-898. Clean
Hänninen, H., E. Beuker, Ø.L.I. Johnsen, M. Murray, L. Sheppard and T. Skrøppa, 2001: Impacts of climate change on cold hardiness of conifers. Conifer Cold Hardiness, F.J. Bigras, and S.J. Colombo, Eds., Kluwer Academic, Dordrecht, 305-333. [NPR]
Hansen, A.J., R.P. Neilson, V.H. Dale, C.H. Flather, L.R. Iverson, D.J. Currie, S. Shafer, R. Cook and P. J. Bartlein, 2001: Global change in forests: Responses of species, communities, and biomes. BioScience, 51, 765-779. [SRC]
Hansen, L.J., J.L. Biringer and J.R. Hoffmann, 2003: Buying Time: A User’s Manual for Building Resistance and Resilience to Climate Change in Natural Systems. WWF Climate Change Program, Berlin, 246 pp. [NPR]
Hanson, P.J. and J.F. Weltzin, 2000: Drought disturbance from climate change: response of United States forests. Sci. Total Environ., 262, 205-220. Clean
Harden, J.W., S.E. Trumbore, B.J. Stocks, A. Hirsch, S.T. Gower, K.P. O’Neill and E.S. Kasischke, 2000: The role of fire in the boreal carbon budget. Global Change Biol., 6, 174-184. [JoC]
Harding, R., P. Kuhry, T.R. Christensen, M.T. Sykes, R. Dankers and S. van der Linden, 2002: Climate feedbacks at the tundra-taiga interface. Ambio Special Report, 12, 47-55. Clean
Hardy, J.T., 2003: Climate Change Causes, Effects and Solutions. Wiley, Chichester, 247 pp. [NPR]
Hare, W., 2006: Relationship between increases in global mean temperature and impacts on ecosystems, food production, water and socio-economic systems. Avoiding Dangerous Climate Change, H.J. Schellnhuber, W. Cramer, N. Naki?enovi?, T.M.L. Wigley and G. Yohe, Eds., Cambridge University Press, Cambridge, 77-185. [NPR]
Harmens, H., P.D. Williams, S.L. Peters, M.T. Bambrick, A. Hopkins and T.W. Ashenden, 2004: Impacts of elevated atmospheric CO2 and temperature on plant community structure of a temperate grassland are modulated by cutting frequency. Grass Forage Sci., 59, 144-156. Clean
Harper, C.W., J.M. Blair, P.A. Fay, A.K. Knapp and J.D. Carlisle, 2005: Increased rainfall variability and reduced rainfall amount decreases soil CO2 flux in a grassland ecosystem. Global Change Biol., 11, 322-334. [JoC]
Harrison, P.A., P.M. Berry, N. Butt and M. New, 2006: Modelling climate change impacts on species’ distributions at the European scale: implications for conservation policy. Environ. Sci. Policy, 9, 116-128. [MoS]
Harrison, S.P. and A.I. Prentice, 2003: Climate and CO2 controls on global vegetation distribution at the last glacial maximum: analysis based on palaeovegetation data, biome modelling and palaeoclimate simulations. Global Change Biol., 9, 983-1004. [JoC, MoS]
Harvell, C.D., C.E. Mitchell, J.R. Ward, S. Altizer, A.P. Dobson, R.S. Ostfeld and M.D. Samuel, 2002: Climate warming and disease risks for terrestrial and marine biota. Science, 296, 2158-2162. [JoC]
Hassan, R., R. Scholes and N. Ash, Eds., 2005: Ecosystems and Human Well-being: Volume 1: Current State and Trends. Island Press, Washington, District of Columbia, 917 pp. [NPR]
Hassol, S.J., Ed., 2004a: Impacts of a Warming Arctic: Arctic Climate Impact Assessment – Highlights. Cambridge University Press, Cambridge, 20 pp. [NPR]
Hassol, S.J., Ed., 2004b: Impacts of a Warming Arctic: Arctic Climate Impact Assessment – Overview Report. Cambridge University Press, Cambridge, 146 pp. [NPR]
Haugaasen, T., J. Barlow and C.A. Peres, 2003: Surface wildfires in central Amazonia: short-term impact on forest structure and carbon loss. Forest Ecol. Manag., 179, 321-331. Clean
Haugan, P.M., C. Turley and H.O. Poertner, 2006: Effects on the marine environment of ocean acidification resulting from elevated levels of CO2 in the atmosphere. Biodiversity Series 285/2006 DN-utredning 2006-1, OSPAR Commission Convention for the Protection of the Marine Environment of the North-East Atlantic (the ‘OSPAR Convention’), London, 33 pp. [NPR, SRC]
Hayhoe, K., D. Cayan, C.B. Field, P.C. Frumhoff, E.P. Maurer, N.L. Miller, S.C. Moser, S.H. Schneider, K.N. Cahill, E.E. Cleland, L. Dale, R. Drapek, R.M. Hanemann, L.S. Kalkstein, J. Lenihan, C.K. Lunch, R.P. Neilson, S.C. Sheridan and J.H. Verville, 2004: Emissions pathways, climate change, and impacts on California. P. Natl. Acad. Sci. USA, 101, 12422-12427. [PoC, JoC, SRC]
Hays, G.C., A.C. Broderick, F. Glen and B.J. Godley, 2003: Climate change and sea turtles: a 150-year reconstruction of incubation temperatures at a major marine turtle rookery. Global Change Biol., 9, 642-646. [JoC]
Hays, G.C., A.J. Richardson and C. Robinson, 2005: Climate change and marine plankton. Trends Ecol. Evol., 20, 337-344. Clean
Heal, G., 2007: Environmental accounting for ecosystems. Ecol. Econ., 61, 693-694. [2007]
Heath, J., E. Ayres, M. Possell, R.D. Bardgett, H.I.J. Black, H. Grant, P. Ineson and G. Kerstiens, 2005: Rising atmospheric CO2 reduces sequestration of root-derived soil carbon. Science, 309, 1711-1713. [JoC]
Hein, L. and N. Ridder, 2006: Desertification in the Sahel: a reinterpretation. Global Change Biol., 12, 751-758. [JoC]
Held, I.M., T.L. Delworth, J. Lu, K.L. Findell and T.R. Knutson, 2005: Simulation of Sahel drought in the 20th and 21st centuries. P. Natl. Acad. Sci. USA, 102, 17891-17896. [JoC, MoS]
Helmuth, B., N. Mieszkowska, P. Moore and S.J. Hawkins, 2006: Living on the edge of two changing worlds: forecasting the responses of rocky intertidal ecosystems to climate change. Annu. Rev. Ecol. Evol. Syst., 37, 337-404. [MoS]
Herrmann, S.M. and C.F. Hutchinson, 2005: The changing contexts of the desertification debate. J. Arid Environ., 63, 538-555. Clean
Herrmann, S.M., A. Anyamba and C.J. Tucker, 2005: Recent trends in vegetation dynamics in the African Sahel and their relationship to climate. Global Environ. Chang., 15, 394-404. [JoC]
Hess, J.C., C.A. Scott, G.L. Hufford and M.D. Fleming, 2001: El Niño and its impact on fire weather conditions in Alaska. Int. J. Wildland Fire; 10, 1-13. Clean
Hetherington, A.M. and F.I. Woodward, 2003: The role of stomata in sensing and driving environmental change. Nature, 424, 901-908. [JoC]
Heywood, V.H. and R.T. Watson, Eds., 1995: Global Biodiversity Assessment. Cambridge University Press, Cambridge, 1140 pp. [NPR]
Hicke, J.A., G.P. Asner, E.S. Kasischke, N.H.F. French, J.T. Randerson, G.J. Collatz, B.J. Stocks, C.J. Tucker, S.O. Los and C.B. Feild, 2003: Postfire response of North American boreal forest net primary productivity analyzed with satellite observations. Global Change Biol., 9, 1145-1157. [JoC]
Hickler, T., L. Eklundh, J.W. Seaquist, B. Smith, J. Ardo, L. Olsson, M.T. Sykes and M. Sjostrom, 2005: Precipitation controls Sahel greening trend. Geophys. Res. Lett., 32, L21415, doi:10.1029/2005GL024370. [JoC]
Hiernaux, P. and M.D. Turner, 2002: The influence of farmer and pastoral management practices on desertification processes in the Sahel. Global Desertification. Do Humans Cause Deserts? J.F. Reynolds and D.M. Stafford-Smith, Eds., Dahlem University Press, Berlin, 135-148. [NPR]
Higgins, P.A.T. and J. Harte, 2006: Biophysical and biogeochemical responses to climate change depend on dispersal and migration. BioScience, 56, 407-417. Clean
Higgins, S.I., J.S. Clark, R. Nathan, T. Hovestadt, F. Schurr, J.M.V. Fragoso, M.R. Aguiar, E. Ribbens and S. Lavorel, 2003: Forecasting plant migration rates: managing uncertainty for risk assessment. J. Ecol., 91, 341-347. [MoS]
Hilbert, D.W., M. Bradford, T. Parker and D.A. Westcott, 2004: Golden bowerbird (Prionodura newtonia) habitat in past, present and future climates: predicted extinction of a vertebrate in tropical highlands due to global warming. Biol. Conserv., 116, 367-377. [MoS]
Hinzman, L., N. Bettez, W. Bolton, F. Chapin, M. Dyurgerov, C. Fastie, B. Griffith, R. Hollister, A. Hope, H. Huntington, A. Jensen, G. Jia, T. Jorgenson, D. Kane, D. Klein, G. Kofinas, A. Lynch, A. Lloyd, A. McGuire, F. Nelson, W. Oechel, T. Osterkamp, C. Racine, V. Romanovsky, R. Stone, D. Stow, M. Sturm, C. Tweedie, G. Vourlitis, M. Walker, D. Walker, P. Webber, J. Welker, K. Winker and K. Yoshikawa, 2005: Evidence and implications of recent climate change in northern Alaska and other Arctic regions. Climatic Change, 72, 251-298. [JoC, ARC]
Hoch, G. and C. Körner, 2003: The carbon charging of pines at the climatic treeline: a global comparison. Oecologia, 135, 10-21. [SRC]
Hoch, G. and C. Körner, 2003: The carbon charging of pines at the climatic treeline: a global comparison. Oecologia, 135, 10-21. [SRC]
Hodar, J.A. and R. Zamora, 2004: Herbivory and climatic warming: a Mediterranean outbreaking caterpillar attacks a relict, boreal pine species. Biodivers. Conserv., 13, 493-500. Clean
Hoegh-Guldberg, O., 1999: Climate change, coral bleaching and the future of the world’s coral reefs. Mar. Freshwater Res., 50, 839-866. [MoS, ARC]
Hoegh-Guldberg, O., 2004: Coral reefs in a century of rapid environmental change. Symbiosis, 37, 1-31. [ARC]
Hoegh-Guldberg, O., 2005: Low coral cover in a high-CO2 world. J. Geophys. Res. C, 110, C09S06, doi:10.1029/2004JC002528. [ARC]
Hoegh-Guldberg, O., H. Hoegh-Guldberg, D.K. Stout, H. Cesar and A. Timmerman, 2000: Pacific in Peril: Biological, Economic and Social Impacts of Climate Change on Pacific Coral Reefs. Greenpeace, Sidney, 72 pp. [NPR, ARC]
Hoffmann, W.A. and R.B. Jackson, 2000: Vegetation–climate feedbacks in the conversion of tropical savanna to grassland. J. Climate, 13, 1593-1602. [JoC]
Hogg, E.H. and A.G. Schwarz, 1997: Regeneration of planted conifers across climatic moisture gradients on the Canadian prairies: implications for distribution and climate change. J. Biogeogr., 24, 527-534. Clean
Hogg, E.H. and R.W. Wein, 2005: Impacts of drought on forest growth and regeneration following fire in southwestern Yukon, Canada. Can. J. Forest Res., 35, 2141-2150. Clean
Holligan, P.M., E. Fernandez, J. Aiken, W.M. Balch, P. Boyd, P.H. Burkill, M. Finch, S.B. Groom, G. Malin, K. Muller, D.A. Purdie, C. Robinson, C.C. Trees, S.M. Turner and P. Vanerwal, 1993: A biogeochemical study of the coccolithophore, Emiliania huxleyi, in the North Atlantic. Global Biogeochem. Cy., 7, 879-900. Clean
Holman, I.P., R.J. Nicholls, P.M. Berry, P.A. Harrison, E. Audsley, S. Shackley and M.D.A. Rounsevell, 2005a: A regional, multi-sectoral and integrated assessment of the impacts of climate and socio-economic change in the UK. Part II. Results. Climatic Change, 71, 43-73. [JoC, SRC]
Holman, I.P., M.D.A. Rounsevell, S. Shackley, P.A. Harrison, R.J. Nicholls, P.M. Berry and E. Audsley, 2005b: A regional, multi-sectoral and integrated assessment of the impacts of climate and socio-economic change in the UK. Part I. Methodology. Climatic Change, 71, 9-41. [JoC, SRC]
Holmgren, M. and M. Scheffer, 2001: El Niño as a window of opportunity for the restoration of degraded arid ecosystems. Ecosystems, 4, 151-159. Clean
Holmgren, M., M. Scheffer, E. Ezcurra, J.R. Gutierrez and G.M.J. Mohren, 2001: El Niño effects on the dynamics of terrestrial ecosystems. Trends Ecol. Evol., 16, 89-94. Clean
Hooper, D.U., F.S. Chapin, J.J. Ewel, A. Hector, P. Inchausti, S. Lavorel, J.H. Lawton, D.M. Lodge, M. Loreau, S. Naeem, B. Schmid, H. Setala, A.J. Symstad, J. Vandermeer and D.A. Wardle, 2005: Effects of biodiversity on ecosystem functioning: a consensus of current knowledge. Ecol. Monogr., 75, 3-35. Clean
Houghton, R.A., 2003a: Revised estimates of the annual net flux of carbon to the atmosphere from changes in land use and land management 1850-2000. Tellus B, 55, 378-390. [MoS]
Houghton, R.A., 2003b: Why are estimates of the terrestrial carbon balance so different? Global Change Biol., 9, 500-509. [JoC, MoS]
House, J.I., I.C. Prentice and C. Le Quéré, 2002: Maximum impacts of future reforestation or deforestation on atmospheric CO2. Global Change Biol., 8, 1047-1052. [JoC, MoS]
Houweling, S., T. Rockmann, I. Aben, F. Keppler, M. Krol, J.F. Meirink, E.J. Dlugokencky and C. Frankenberg, 2006: Atmospheric constraints on global emissions of methane from plants. Geophys. Res. Lett., 33, L15821, doi:10.1029/2006GL026162. [JoC]
Huber, U.M., H.K.M. Bugmann and M.A. Reasoner, Eds., 2005: Global Change and Mountain Regions: An Overview of Current Knowledge. Springer, Berlin, 650 pp. [NPR, SRC]
Huelber, K., M. Gottfried, H. Pauli, K. Reiter, M. Winkler and G. Grabherr, 2006: Phenological responses of snowbed species to snow removal dates in the Central Alps: implications for climate warming. Arct. Antarct. Alp. Res., 38, 99-103. Clean
Hughes, L., 2003: Climate change and Australia: trends, projections and impacts. Austral Ecol., 28, 423-443. [MoS, SRC]
Hughes, L., E.M. Cawsey and M. Westoby, 1996: Climatic range sizes of Eucalyptus species in relation to future climate change. Global Ecol. Biogeogr. Lett., 5, 23-29. [MoS, SRC]
Hughes, T.P., A.H. Baird, D.R. Bellwood, M. Card, S.R. Connolly, C. Folke, R. Grosberg, O. Hoegh-Guldberg, J.B.C. Jackson, J. Kleypas, J.M. Lough, P. Marshall, M. Nystrom, S.R. Palumbi, J.M. Pandolfi, B. Rosen and J. Roughgarden, 2003: Climate change, human impacts, and the resilience of coral reefs. Science, 301, 929-933. [JoC, ARC]
Hulme, M., 2001: Climatic perspectives on Sahelian desiccation: 1973-1998. Global Environ. Chang., 11, 19-29. [JoC]
Hulme, M., J. Mitchell, W. Ingram, J. Lowe, T. Johns, M. New and D. Viner, 1999: Climate change scenarios for global impacts studies. Global Environ. Chang., 9, S3-S19. [JoC, ARC]
Hungate, B.A., C.H. Jaeger III, G. Gamara, F.S. Chapin III and C.B. Field, 2000: Soil microbiota in two annual grasslands: responses to elevated atmospheric CO2. Oecologia, 124, 589-598. Clean
Hungate, B.A., J.S. Dukes, M.R. Shaw, Y.Q. Luo and C.B. Field, 2003: Nitrogen and climate change. Science, 302, 1512-1513. [JoC, ARC]
Hungate, B.A., D.W. Johnson, P. Dukstra, G. Hymus, P. Stiling, J.P. Megonigal, A.L. Pagel, J.L. Moan, F. Day, J. Li, C.R. Hinkle and B.G. Drake, 2006: Nitrogen cycling during seven years of atmospheric CO2 enrichment in a scrub oak woodland. Ecology, 87, 26-40. Clean
Hurrell, J.W. and H. van Loon, 1997: Decadal variations in climate associated with the North Atlantic oscillation. Climatic Change, 36, 301-326. [JoC]
Hutchinson, C.F., S.M. Herrmann, T. Maukonen and J. Weber, 2005: Introduction: the “greening” of the Sahel. J. Arid Environ., 63, 535-537. Clean
iLEAPS, Ed., 2005: Integrated land ecosystem-atmosphere processes study science plan and implementation strategy. IGBP report No. 54, International Geosphere and Biosphere Program Secretariat, Stockholm, 52 pp. [NPR]
Inkley, D.B., M.G. Anderson, A.R. Blaustein, V.R. Burkett, B. Felzer, B. Griffith, J. Price and T.L. Root, 2004: Global Climate Change and Wildlife in North America. The Wildlife Society, Bethesda, Maryland, 34 pp. [NPR, SRC]
Inouye, D.W., B. Barr, K.B. Armitage and B.D. Inouye, 2000: Climate change is affecting altitudinal migrants and hibernating species. P. Natl. Acad. Sci. USA, 97, 1630-1633. [JoC]
IPCC, 1990: IPCC First Assessment Report (1990): Scientific Assessment of Climate Change. Contribution of Working Group I to the Intergovernmental Panel on Climate Change, J.T. Houghton, G.J. Jenkins and J.J. Ephraums, Eds., Cambridge University Press, Cambridge, 365 pp. [NPR]
IPCC, 1996: Climate Change 1995: The Science of Climate Change. Contribution of Working Group I to the Second Assessment Report of the Intergovernmental Panel on Climate Change, J.T. Houghton, L.G.M. Filho, B.A. Callender, N. Harris, A. Kattenberg and K. Maskell, Eds., Cambridge University Press, Cambridge, 572 pp. [NPR]
IPCC, 2001: Climate Change 2001: The Scientific Basis. Contribution of Working Group I to the Third Assessment Report of the Intergovernmental Panel on Climate Change, J.T. Houghton, Y. Ding, D.J. Griggs, M. Noguer, P.J. van der Linden, X. Dai, K. Maskell and C.A. Johnson, Eds., Cambridge University Press, Cambridge, 881 pp. [NPR]
IPCC, 2007: Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, S. Solomon, D. Qin, M. Manning, Z. Chen, M. Marquis, K.B. Averyt, M. Tignor and H.L. Miller, Eds., Cambridge University Press, Cambridge, 996 pp. [NPR, 2007]
IPCC-TGCIA, 1999: Guidelines on the Use of Scenario Data for Climate Impact and Adaptation Assessment. Prepared by T.R. Carter, M. Hulme and M. Lal, Intergovernmental Panel on Climate Change (IPCC): Task Group on Scenarios for Climate Impact Assessment (TGCIA), Norwich, Hamburg, and New York, 69 pp. [NPR]
Isaev, A.S., G.N. Korovin, S.A. Bartalev, D.V. Ershov, A. Janetos, E.S. Kasischke, H.H. Shugart, N.H.F. French, B.E. Orlick and T.L. Murphy, 2002: Using remote sensing to assess Russian forest fire carbon emissions. Climatic Change, 55, 235-249. [JoC, ARC]
Iverson, L.R. and A.M. Prasad, 2002: Potential redistribution of tree species habitat under five climate change scenarios in the eastern US. Forest Ecol. Manag., 155, 205-222. Clean
Iverson, L.R., M.W. Schwartz and A.M. Prasad, 2004: How fast and far might tree species migrate in the eastern United States due to climate change? Global Ecol. Biogeogr., 13, 209-219. Clean
Izaurralde, R.C., A.M. Thomson, N.J. Rosenberg and R.A. Brown, 2005: Climate change impacts for the conterminous USA: an integrated assessment. Part 6. Distribution and productivity of unmanaged ecosystems. Climatic Change, 69, 107-126. [JoC]
Jackson, D.A. and N.E. Mandrak, 2002: Changing fish biodiversity: predicting the loss of cyprinid biodiversity due to global climate change. Fisheries in a Changing Climate. N.A. McGinn, Ed., American Fisheries Society Symposium 32, Phoenix, Arizona. American Fisheries Society, Bethesda, Maryland, 89-98. [NPR]
Jain, R., 2003: Sustainable development: differing views and policy alternatives. Clean Technologies and Environmental Policy, 4, 197-198. Clean
Jankowski, T., D.M. Livingstone, H. Bührer, R. Forster and P. Niederhaser, 2006: Consequences of the 2003 European heat wave for lake temperature profiles, thermal stability and hypolimnetic oxygen depletion: implications for a warmer world. Limnol. Oceanogr., 51, 815-819. Clean
Jansen, E., J. Overpeck, K.R. Briffa, J.-C. Duplessy, F. Joos, V. Masson-Delmotte, D.O. Olago, B. Otto-Bliesner, Wm. Richard Pelteir, S. Rahmstorf, R. Ramesh, D. Raynaud, D.H. Rind, O. Solomina, R. Villalba and D. Zhang, 2007: Paleoclimate. Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, S. Solomon, D. Qin, M. Manning, Z. Chen, M. Marquis, K.B. Averyt, M. Tignor and H.L. Miller, Eds., Cambridge University Press, Cambridge, 434-496. [NPR, PoC, 2007]
Jansson, R., 2003: Global patterns in endemism explained by past climatic change. P. Roy. Soc. Lond. B Bio, 270, 583-590. Clean
Jasienski, M., S.C. Thomas and F.A. Bazzaz, 1998: Blaming the trees: a critique of research on forest responses to high CO2. Trends Ecol. Evol., 13, 427. Clean
Jasinski, J.P.P. and S. Payette, 2005: The creation of alternative stable states in the southern boreal forest, Quebec, Canada. Ecol. Monogr., 75, 561-583. Clean
Jasper, K., P. Calanca, D. Gyalistras and J. Fuhrer, 2004: Differential impacts of climate change on the hydrology of two alpine river basins. Climate Res., 26, 113-129. [SRC]
Jenkins, M., 2003: Prospects for biodiversity. Science, 302, 1175-1177. [JoC]
Jobbagy, E.G. and R.B. Jackson, 2000: The vertical distribution of soil organic carbon and its relation to climate and vegetation. Ecol. Appl., 10, 423-436. Clean
Johansen, R.J., 2001: Impacts of fire on biological soil crusts. Biological Soil Crusts: Structure, Function, and Management, J. Belnap and O.L. Lange, Eds., Springer, Berlin, 386-397. [NPR]
Johnson, W.C., B.V. Millett, T. Gilmanov, R.A. Voldseth, G.R. Guntenspergen and D.E. Naugle, 2005: Vulnerability of northern prairie wetlands to climate change. BioScience, 55, 863-872. Clean
Johnstone, J.F. and F.S. Chapin, 2003: Non-equilibrium succession dynamics indicate continued northern migration of lodgepole pine. Global Change Biol., 9, 1401-1409. [JoC]
Johnstone, J.F. and F.S. Chapin, 2006: Effects of soil burn severity on post-fire tree recruitment in boreal forests. Ecosystems, 9, 14-31. Clean
Jolly, W.M., M. Dobbertin, N.E. Zimmermann and M. Reichstein, 2005: Divergent vegetation growth responses to the 2003 heat wave in the Swiss Alps. Geophys. Res. Lett., 32, L18409, doi:10.1029/2005GL023252. [JoC]
Jompa, J. and L.J. McCook, 2003: Coral-algal competition: macroalgae with different properties have different effects on corals. Mar. Ecol.–Prog. Ser., 258, 87-95. Clean
Jones, M.B. and A. Donnelly, 2004: Carbon sequestration in temperate grassland ecosystems and the influence of management, climate and elevated CO2. New Phytol., 164, 423-439. Clean
Jones, P., J. Amador, M. Campos, K. Hayhoe, M. Marín, J. Romero and A. Fischlin, 2005: Generating climate change scenarios at high resolution for impact studies and adaptation: focus on developing countries. Tropical Forests and Adaptation to Climate Change: In Search of Synergies, Y. Saloh, C. Robledo and K. Markku, Eds., Island Press, Washington, District of Columbia, 37-55. [NPR, SRC]
Joos, F. and I.C. Prentice, 2004: A paleo-perspective on changes in atmospheric CO2 and climate. Global Carbon Cycle: Integrating Humans, Climate, and the Natural World, Field, C.B. Raupach and M.R. Raupach, Eds., Island Press, Washington, District of Columbia, 165-186. [NPR, PoC]
Juday, G.P., 2005: Forests, land management and agriculture. Arctic Climate Impact Assessment (ACIA): Scientific Report, C. Symon, L. Arris and B. Heal, Eds., Cambridge University Press, Cambridge, 781-862. [NPR]
Jump, A.S. and J. Peñuelas, 2005: Running to stand still: adaptation and the response of plants to rapid climate change. Ecol. Lett., 8, 1010-1020. Clean
Jump, A.S., J.M. Hunt, J.A. Martinez-Izquierdo and J. Peñuelas, 2006: Natural selection and climate change: temperature-linked spatial and temporal trends in gene frequency in Fagus sylvatica. Mol. Ecol., 15, 3469-3480. [NPR]
Kajimoto, T., H. Daimaru, T. Okamoto, T. Otani and H. Onodera, 2004: Effects of snow avalanche disturbance on regeneration of subalpine Abies mariesii forest, northern Japan. Arct. Antarct. Alp. Res., 36, 436-445. Clean
Kaplan, J.O., N.H. Bigelow, I.C. Prentice, S.P. Harrison, P.J. Bartlein, T.R. Christensen, W. Cramer, N.V. Matveyeva, A.D. McGuire, D.F. Murray, V.Y. Razzhivin, B. Smith, D.A. Walker, P.M. Anderson, A.A. Andreev, L.B. Brubaker, M.E. Edwards and A.V. Lozhkin, 2003: Climate change and Arctic ecosystems. 2. Modeling, paleodata-model comparisons, and future projections. J. Geophys. Res. D, 108, 8171, doi:10.1029/2002JD002559. [MoS, SRC]
Kappelle, M., M.M.I. Van Vuuren and P. Baas, 1999: Effects of climate change on biodiversity: a review and identification of key research issues. Biodivers. Conserv., 8, 1383-1397. Clean
Karjalainen, T., A. Pussinen, J. Liski, G.J. Nabuurs, M. Erhard, T. Eggers, M. Sonntag and G.M.J. Mohren, 2002: An approach towards an estimate of the impact of forest management and climate change on the European forest sector carbon budget: Germany as a case study. Forest Ecol. Manag., 162, 87-103. [MoS]
Karlsson, J., A. Jonsson and M. Jansson, 2005: Productivity of high-latitude lakes: climate effect inferred from altitude gradient. Global Change Biol., 11, 710-715. [JoC]
Karnosky, D.F., 2003: Impacts of elevated atmospheric CO2 on forest trees and forest ecosystems: knowledge gaps. Environ. Int., 29, 161-169. Clean
Karnosky, D.F., D.R. Zak, K.S. Pregitzer, C.S. Awmack, J.G. Bockheim, R.E. Dickson, G.R. Hendrey, G.E. Host, J.S. King, B.J. Kopper, E.L. Kruger, M.E. Kubiske, R.L. Lindroth, W.J. Mattson, E.P. McDonald, A. Noormets, E. Oksanen, W.F.J. Parsons, K.E. Percy, G.K. Podila, D.E. Riemenschneider, P. Sharma, R. Thakur, A. Sober, J. Sober, W.S. Jones, S. Anttonen, E. Vapaavuori, B. Mankovska, W. Heilman and J.G. Isebrands, 2003: Tropospheric O3 moderates responses of temperate hardwood forests to elevated CO2: a synthesis of molecular to ecosystem results from the Aspen FACE project. Funct. Ecol., 17, 289-304. Clean
Kasischke, E.S. and B.J. Stocks, Eds., 2000: Fire, Climate Change and Carbon Cycling in the Boreal Forest. Springer-Verlag, New York, 461 pp. [NPR]
Kasischke, E.S. and M.R. Turetsky, 2006: Recent changes in the fire regime across the North American boreal region: spatial and temporal patterns of burning across Canada and Alaska. Geophys. Res. Lett., 33, L09703, doi:10.1029/2006GL025677. [JoC]
Kasischke, E.S., J.H. Hewson, B. Stocks, G. van der Werf and J. Randerson, 2003: The use of ATSR active fire counts for estimating relative patterns of biomass burning: a study from the boreal forest region. Geophys. Res. Lett., 30, 1969, doi:10.1029/2003GL017859. [JoC, MoS]
Kasischke, E.S., E.J. Hyer, P.C. Novelli, L.P. Bruhwiler, N.H.F. French, A.I. Sukhinin, J.H. Hewson and B.J. Stocks, 2005: Influences of boreal fire emissions on Northern Hemisphere atmospheric carbon and carbon monoxide. Global Biogeochem. Cy., 19, GB1012, doi:10.1029/2004GB002300. Clean
Kates, R.W., 2000: Cautionary tales: adaptation and the global poor. Climatic Change, 45, 5-17. [JoC, ARC]
Keddy, P.A., 2000: Wetland Ecology: Principles and Conservation. Cambridge University Press, Cambridge, 614 pp. [NPR]
Keeley, J.E., 2002: Fire management of California shrubland landscapes. Environ. Manage., 29, 395-408. Clean
Keller, A.A. and G. Klein-Macphee, 2000: Impact of elevated temperature on the growth, survival, and trophic dynamics of winter flounder larvae: a mesocosm study. Can. J. Fish. Aquat. Sci., 57, 2382-2392. Clean
Keller, F., S. Goyette and M. Beniston, 2005: Sensitivity analysis of snow cover to climate change scenarios and their impact on plant habitats in alpine terrain. Climatic Change, 72, 299-319. [JoC, SRC]
Keller, J.K., J.R. White, S.D. Bridgham and J. Pastor, 2004: Climate change effects on carbon and nitrogen mineralization in peatlands through changes in soil quality. Global Change Biol., 10, 1053-1064. [JoC]
Kemp, M., 2005: Science in culture: inventing an icon. Nature, 437, 1238-1238. [JoC]
Kennett, S.A., 2002: National policies for biosphere greenhouse gas management: issues and opportunities. Environ. Manage., 30, 595-608. Clean
Keppler, F., J.T.G. Hamilton, M. Brass and T. Rockmann, 2006: Methane emissions from terrestrial plants under aerobic conditions. Nature, 439, 187-191. [JoC]
Kerr, J. and L. Packer, 1998: The impact of climate change on mammal diversity in Canada. Environ. Monit. Assess., 49, 263-270. Clean
Kienast, F., O. Wildi and B. Brzeziecki, 1998: Potential impacts of climate change on species richness in mountain forests: an ecological risk assessment. Biol. Conserv., 83, 291-305. Clean
King, J.A., R.I. Bradley, R. Harrison and A.D. Carter, 2004a: Carbon sequestration and saving potential associated with changes to the management of agricultural soils in England. Soil Use Manage., 20, 394-402. Clean
King, J.S., P.J. Hanson, E. Bernhardt, P. Deangelis, R.J. Norby and K.S. Pregitzer, 2004b: A multiyear synthesis of soil respiration responses to elevated atmospheric CO2 from four forest FACE experiments. Global Change Biol., 10, 1027-1042. [JoC]
Kirschbaum, M. and A. Fischlin, 1996: Climate change impacts on forests. Climate Change 1995: Impacts; Adaptations and Mitigation of Climate Change. Scientific-Technical Analysis. Contribution of Working Group II to the Second Assessment Report of the Intergovernmental Panel of Climate Change., R. Watson, M.C. Zinyowera and R.H. Moss, Eds., Cambridge University Press, Cambridge, 95-129. [NPR, SRC]
Kitayama, K., 1996: Climate of the summit region of Mount Kinabalu (Borneo) in 1992, an El Niño year. Mt. Res. Dev., 16, 65-75. Clean
Klanderud, K., 2005: Climate change effects on species interactions in an alpine plant community. J. Ecol., 93, 127-137. Clean
Klanderud, K. and O. Totland, 2005: Simulated climate change altered dominance hierarchies and diversity of an alpine biodiversity hotspot. Ecology, 86, 2047-2054. [MoS]
Kleypas, J.A. and C. Langdon, 2002: Overview of CO2-induced changes in seawater chemistry. World Coral Reefs in the New Millenium: Bridging Research and Management for Sustainable Development, M.K. Moosa, S. Soemodihardjo, A. Soegiarto, K. Romimohtarto, A. Nontji and S. Suharsono, Eds., Proceedings of the 9th International Coral Reef Symposium, 2, Bali, Indonesia. Ministry of Environment, Indonesian Institute of Sciences, International Society for Reef Studies, 1085-1089. [NPR]
Kleypas, J.A., R.W. Buddemeier, D. Archer, J.P. Gattuso, C. Langdon and B.N. Opdyke, 1999: Geochemical consequences of increased atmospheric carbon dioxide on coral reefs. Science, 284, 118-120. [JoC]
Kleypas, J.A., R.W. Buddemeier and J.P. Gattuso, 2001: The future of coral reefs in an age of global change. Int. J. Earth Sci., 90, 426-437. [MoS]
Kleypas, J.A., R.W. Buddemeier, C.M. Eakin, J.P. Gattuso, J. Guinotte, O. Hoegh-Guldberg, R. Iglesias-Prieto, P.L. Jokiel, C. Langdon, W. Skirving and A.E. Strong, 2005: Comment on “Coral reef calcification and climate change: the effect of ocean warming”. Geophys. Res. Lett., 32, L08601, doi:10.1029/2004GL022329. [JoC, ARC]
Kling, J., K. Hayhoe, L.B. Johnsoin, J.J. Magnuson, S. Polasky, S.K. Robinson, B.J. Shuter, M.M. Wander, D.J. Wuebbles and D.R. Zak, 2003: Confronting Climate Change in the Great Lakes Region: Impacts on our Communities and Ecosystems. Union of Concerned Scientists and the Ecological Society of America, Cambridge, Massachusetts and Washington, District of Columbia, 92 pp. [NPR]
Knorr, W., I.C. Prentice, J.I. House and E.A. Holland, 2005: Long-term sensitivity of soil carbon turnover to warming. Nature, 433, 298-301. [JoC]
Kohler, P., F. Joos, S. Gerber and R. Knutti, 2005: Simulated changes in vegetation distribution, land carbon storage, and atmospheric CO2 in response to a collapse of the North Atlantic thermohaline circulation. Clim. Dynam., 25, 689-708. [PoC, JoC, MoS]
Koop, K., D. Booth, A. Broadbent, J. Brodie, D. Bucher, D. Capone, J. Coll, W. Dennison, M. Erdmann, P. Harrison, O. Hoegh-Guldberg, P. Hutchings, G.B. Jones, A.W.D. Larkum, J. O’Neil, A. Steven, E. Tentori, S. Ward, J. Williamson and D. Yellowlees, 2001: ENCORE: The effect of nutrient enrichment on coral reefs: synthesis of results and conclusions. Mar. Pollut. Bull., 42, 91-120. [ARC]
Körner, C., 1998: Worldwide positions of alpine treelines and their causes. The Impacts of Climate Variability on Forests, M. Beniston and J.L. Innes, Eds., Springer, Berlin, 221-229. [NPR, SRC]
Körner, C., 2003a: Alpine Plant Life: Functional Plant Ecology of High Mountain Ecosystems. Springer, Berlin, 343 pp. [NPR, SRC]
Körner, C., 2003b: Ecological impacts of atmospheric CO2 enrichment on terrestrial ecosystems. Philos. T. Roy. Soc. Lond., 361, 2023-2041. [SRC]
Körner, C., 2003c: Slow in, rapid out: carbon flux studies and Kyoto targets. Science, 300, 1242-1243. [JoC, SRC]
Körner, C., 2004: Through enhanced tree dynamics carbon dioxide enrichment may cause tropical forests to lose carbon. Philos. T. Roy. Soc. Lond., 359, 493-498. [SRC]
Körner, C. and J. Paulsen, 2004: A world-wide study of high altitude treeline temperatures. J. Biogeogr., 31, 713-732. [SRC]
Körner, C., R. Asshoff, O. Bignucolo, S. Hättenschwiler, S.G. Keel, S. Peláez-Riedl, S. Pepin, R.T.W. Siegwolf and G. Zotz, 2005a: Carbon flux and growth in mature deciduous forest trees exposed to elevated CO2. Science, 309, 1360-1362. [JoC, SRC]
Körner, C., D. Sarris and D. Christodoulakis, 2005b: Long-term increase in climatic dryness in the East Mediterranean as evidenced for the island of Samos. Reg. Environ. Change, 5, 27-36. [SRC]
Kräuchi, N., P. Brang and W. Schonenberger, 2000: Forests of mountainous regions: gaps in knowledge and research needs. Forest Ecol. Manag., 132, 73-82. Clean
Kremen, C., 2005: Managing ecosystem services: what do we need to know about their ecology? Ecol. Lett., 8, 468-479. Clean
Lake, J.C. and M.R. Leishman, 2004: Invasion success of exotic plants in natural ecosystems: the role of disturbance, plant attributes and freedom from herbivores. Biol. Conserv., 117, 215-226. Clean
Lake, P.S., M.A. Palmer, P. Biro, J. Cole, A.P. Covich, C. Dahm, J. Gibert, W. Goedkoop, K. Martens and J. Verhoeven, 2000: Global change and the biodiversity of freshwater ecosystems: impacts on linkages between above-sediment and sediment biota. BioScience, 50, 1099-1107. Clean
Lal, R., 2003: Offsetting global CO2 emissions by restoration of degraded soils and intensification of world agriculture and forestry. Land Degrad. Dev., 14, 309-322. Clean
Lal, R., 2004: Soil carbon sequestration to mitigate climate change. Geoderma, 123, 1-22. Clean
Lambin, E.F., H.J. Geist and E. Lepers, 2003: Dynamics of land-use and land-cover change in tropical regions. Annu. Rev. Ecol. Evol. Syst., 28, 205-241. Clean
Landhäusser, S.M. and R.W. Wein, 1993: Postfire vegetation recovery and tree establishment at the Arctic treeline: climate-change vegetation-response hypotheses. J. Ecol., 81, 665-672. Clean
Langdon, C., W.S. Broecker, D.E. Hammond, E. Glenn, K. Fitzsimmons, S.G. Nelson, T.H. Peng, I. Hajdas and G. Bonani, 2003: Effect of elevated CO2 on the community metabolism of an experimental coral reef. Global Biogeochem. Cy., 17, 1011, doi:10.1029/2002GB001941. Clean
Langenfelds, R.L., R.J. Francey, B.C. Pak, L.P. Steele, J. Lloyd, C.M. Trudinger and C.E. Allison, 2002: Interannual growth rate variations of atmospheric CO2 and its isotope d13C, H2, CH4, and CO between 1992 and 1999 linked to biomass burning. Global Biogeochem. Cy., 16, 1048, doi:10.1029/2001GB001466. Clean
Lara, A., R. Villalba, A. Wolodarsky-Franke, J.C. Aravena, B.H. Luckman and E. Cuq, 2005: Spatial and temporal variation in Nothofagus pumilio growth at tree line along its latitudinal range (35°40’–55° S) in the Chilean Andes. J. Biogeogr., 32, 879-893. Clean
Lauenroth, W.K., H.E. Epstein, J.M. Paruelo, I.C. Burke, M.R. Aguiar and O.E. Sala, 2004: Potential effects of climate change on the temperate zones of North and South America. Rev. Chil. Hist. Nat., 77, 439-453. Clean
Laurance, W.F., A.A. Oliveira, S.G. Laurance, R. Condit, H.E.M. Nascimento, A.C. Sanchez-Thorin, T.E. Lovejoy, A. Andrade, S. D’Angelo, J.E. Riberiro and C.W. Dick, 2004: Pervasive alteration of tree communities in undisturbed Amazonian forests. Nature, 428, 171-175. [JoC]
Lavoie, L. and L. Sirois, 1998: Vegetation changes caused by recent fires in the northern boreal forest of eastern Canada. J. Veg. Sci., 9, 483-492. Clean
Lavorel, S., 1999: Ecological diversity and resilience of Mediterranean vegetation to disturbance. Divers. Distrib., 5, 3-13. Clean
Lawton, R.O., U.S. Nair, R.A. Pielke Sr. and R.M. Welch, 2001: Climatic impact of tropical lowland deforestation on nearby montane cloud forests. Science, 294, 584-587. [JoC]
Le Maitre, D.C., D.M. Richardson and R.A. Chapman, 2004: Alien plant invasions in South Africa: driving forces and the human dimension. S. Afr. J. Sci., 100, 103-112. Clean
Le Roux, P.C., M.A. McGeoch, M.J. Nyakatya and S.L. Chown, 2005: Effects of a short-term climate change experiment on a sub-Antarctic keystone plant species. Global Change Biol., 11, 1628-1639. [JoC]
Leakey, A.D.B., M. Uribelarrea, E.A. Ainsworth, S.L. Naidu, A. Rogers, D.R. Ort and S.P. Long, 2006: Photosynthesis, productivity, and yield of maize are not affected by open-air elevation of CO2 concentration in the absence of drought. Plant Physiol., 140, 779-790. Clean
Learmonth, J.A., C.D. MacLeod, M.B. Santos, G.J. Pierce, H.Q.P. Crick and R.A. Robinson, 2006: Potential effects of climate change on marine mammals. Oceanogr. Mar. Biol., 44, 431-464. Clean
Leemans, R. and B. Eickhout, 2004: Another reason for concern: regional and global impacts on ecosystems for different levels of climate change. Global Environ. Chang., 14, 219-228. [JoC, SRC]
Leemans, R., B. Eickhout, B. Strengers, L. Bouwman and M. Schaeffer, 2002: The consequences of uncertainties in land use, climate and vegetation responses on the terrestrial carbon. Sci. China Ser. C, 45, 126-141. [SRC]
Lehodey, P., F. Chai and J. Hampton, 2003: Modelling climate-related variability of tuna populations from a coupled ocean-biogeochemical-populations dynamics model. Fish. Oceanogr., 12, 483-494. [MoS]
Lelieveld, J., 2006: Climate change: a nasty surprise in the greenhouse. Nature, 443, 405-406. [JoC]
Lelieveld, J., H. Berresheim, S. Borrmann, P.J. Crutzen, F.J. Dentener, H. Fischer, J. Feichter, P.J. Flatau, J. Heland, B. Holzinger, R. Korrmann, M.G. Lawrence, Z. Levin, K.M. Markowicz, N. Milhalopoulos, A. Minikin, V. Ramanathan, M. de Reus, G.J. Roelofs, H.A. Scheeren, J. Sciare, H. Schlager, M. Schultz, P. Siegmund, B. Steil, E.G. Stephanou, P. Stier, M. Traub, C. Warneke, J. Williams and H. Ziereis, 2002: Global air pollution crossroads over the Mediterranean. Science, 298, 794-799. [JoC]
Lemke, P., J. Ren, R. Alley, I. Allison, J. Carrasco, G. Flato, Y. Fujii, G. Kaser, P. Mote, R. Thomas and T. Zhang, 2007: Observations: changes in snow, ice and frozen ground. Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, S. Solomon, D. Qin, M. Manning, Z. Chen, M. Marquis, K.B. Averyt, M. Tignor and H.L. Miller, Eds., Cambridge University Press, Cambridge, 335-383. [NPR, ARC, 2007]
Lenihan, J.M., R. Drapek, D. Bachelet and R.P. Neilson, 2003: Climate change effects on vegetation distribution, carbon, and fire in California. Ecol. Appl., 13, 1667. [SRC]
Lepers, E., E.F. Lambin, A.C. Janetos, R. Defries, F. Achard, N. Ramankutty and R.J. Scholes, 2005: A synthesis of information on rapid land-cover change for the period 1981-2000. BioScience, 55, 115-124. [ARC]
Leuzinger, S., G. Zotz, R. Asshoff and C. Körner, 2005: Responses of deciduous forest trees to severe drought in Central Europe. Tree Physiol., 25, 641-650. [SRC]
Levy, P.E., M.G.R. Cannell and A.D. Friend, 2004: Modelling the impact of future changes in climate, CO2 concentration and land use on natural ecosystems and the terrestrial carbon sink. Global Environ. Chang., 14, 21-30. [JoC, MoS]
Lewis, S.L., Y. Malhi and O.L. Phillips, 2004a: Fingerprinting the impacts of global change on tropical forests. Philos. T. Roy. Soc. Lond. B, 359, 437-462. Clean
Lewis, S.L., O.L. Phillips, T.R. Baker, J. Lloyd, Y. Malhi, S. Almeida, N. Higuchi, W.F. Laurance, D.A. Neill, J.N.M. Silva, J. Terborgh, A.T. Lezama, R.V. Martinez, S. Brown, J. Chave, C. Kuebler, P.N. Vargas and B. Vinceti, 2004b: Concerted changes in tropical forest structure and dynamics: evidence from 50 South American long-term plots. Philos. T. Roy. Soc. Lond. B, 359, 421-436. Clean
Lexer, M.J., K. Honninger, H. Scheifinger, C. Matulla, N. Groll and H. Kromp-Kolb, 2000: The sensitivity of central European mountain forests to scenarios of climatic change: methodological frame for a large-scale risk assessment. Silva Fenn., 34, 113-129. Clean
Li, C., M.D. Flannigan and I.G.W. Corns, 2000: Influence of potential climate change on forest landscape dynamics of west-central Alberta. Can. J. Forest Res., 30, 1905-1912. [ARC]
Li, S.G., J. Asanuma, W. Eugster, A. Kotani, J.J. Liu, T. Urano, T. Oikawa, G. Davaa, D. Oyunbaatar and M. Sugita, 2005: Net ecosystem carbon dioxide exchange over grazed steppe in central Mongolia. Global Change Biol., 11, 1941-1955. [JoC, ARC]
Lichter, J., S.H. Barron, C.E. Bevacqua, A.C. Finzi, K.F. Irving, E.A. Stemmler and W.H. Schlesinger, 2005: Soil carbon sequestration and turnover in a pine forest after six years of atmospheric CO2 enrichment. Ecology, 86, 1835-1847. Clean
Lindroth, R.L., B.J. Kopper, W.F.J. Parsons, J.G. Bockheim, D.F. Karnosky, G.R. Hendrey, K.S. Pregitzer, J.G. Isebrands and J. Sober, 2001: Consequences of elevated carbon dioxide and ozone for foliar chemical composition and dynamics in trembling aspen (Populus tremuloides) and paper birch (Betula papyrifera). Environ. Pollut., 115, 395-404. Clean
Lioubimtseva, E. and J.M. Adams, 2004: Possible implications of increased carbon dioxide levels and climate change for desert ecosystems. Environ. Manage., 33, S388-S404. Clean
Lischke, H., A. Guisan, A. Fischlin, J. Williams and H. Bugmann, 1998: Vegetation responses to climate change in the Alps: modeling studies. Views from the Alps: Regional Perspectives on Climate Change, P. Cebon, U. Dahinden, H.C. Davies, D.M. Imboden and C.C. Jager, Eds., MIT Press, Boston, Massachusetts, 309-350. [NPR, SRC]
Lischke, H., A.F. Lotter and A. Fischlin, 2002: Untangling a Holocene pollen record with forest model simulations and independent climate data. Ecol. Model., 150, 1-21. [MoS, SRC]
Liu, X.D. and B.D. Chen, 2000: Climatic warming in the Tibetan Plateau during recent decades. Int. J. Climatol., 20, 1729-1742. [JoC]
Llope, M., R. Anadon, L. Viesca, M. Quevedo, R. Gonzalez-Quiros and N.C. Stenseth, 2006: Hydrography of the southern Bay of Biscay shelf-break region: integrating the multiscale physical variability over the period 1993-2003. J. Geophys. Res. C, 111, C09021, doi:10.1029/2005JC002963. Clean
Llorens, L. and J. Peñuelas, 2005: Experimental evidence of future drier and warmer conditions affecting flowering of two co-occurring Mediterranean shrubs. Int. J. Plant Sci., 166, 235-245. [MoS]
Llorens, L., J. Peñuelas and M. Estiarte, 2003: Ecophysiological responses of two Mediterranean shrubs, Erica multiflora and Globularia alypum, to experimentally drier and warmer conditions. Physiol. Plant., 119, 231-243. Clean
Llorens, L., J. Peñuelas, M. Estiarte and P. Bruna, 2004: Contrasting growth changes in two dominant species of a Mediterranean shrubland submitted to experimental drought and warming. Ann. Bot., 94, 843-853. Clean
Lloret, F., D. Siscart and C. Dalmases, 2004: Canopy recovery after drought dieback in holm-oak Mediterranean forests of Catalonia (NE Spain). Global Change Biol., 10, 2092-2099. [JoC]
Lloyd, A.H., 2005: Ecological histories from Alaskan tree lines provide insight into future change. Ecology, 86, 1687-1695. [MoS]
Lobo, A. and P. Maisongrande, 2006: Stratified analysis of satellite imagery of SW Europe during summer 2003: the differential response of vegetation classes to increased water deficit. Hydrol. Earth Syst. Sci., 10, 151-164. Clean
Logan, J.A., J. Regniere and J.A. Powell, 2003: Assessing the impacts of global warming on forest pest dynamics. Front. Ecol. Environ., 1, 130-137. Clean
Lorenzoni, I., N.F. Pidgeon and R.E. O’Connor, 2005: Dangerous climate change: the role for risk research. Risk Anal., 25, 1387-1398. [ARC]
Loukos, H., P. Monfray, L. Bopp and P. Lehodey, 2003: Potential changes in skipjack tuna (Katsuwonus pelamis) habitat from a global warming scenario: modelling approach and preliminary results. Fish. Oceanogr., 12, 474-482. [MoS]
Lovejoy, T.E. and L. Hannah, Eds., 2005: Climate Change and Biodiversity. Yale University Press, New Haven, Connecticut, 418 pp. [NPR]
Lucht, W., I.C. Prentice, R.B. Myneni, S. Sitch, P. Friedlingstein, W. Cramer, P. Bousquet, W. Buermann and B. Smith, 2002: Climatic control of the high-latitude vegetation greening trend and Pinatubo effect. Science, 296, 1687-1689. [JoC, SRC]
Lucht, W., S. Schaphoff, T. Erbrecht, U. Heyder and W. Cramer, 2006: Terrestrial vegetation redistribution and carbon balance under climate change. Carbon Balance Manage., 1:6 doi:10.1186/1750-0680-1-6. [SRC]
Luckman, B., 1994: Using multiple high-resolution proxy climate records to reconstruct natural climate variability: an example from the Canadian Rockies. Mountain Environments in Changing Climates, M. Beniston, Ed., Routledge, London, 42-59. [NPR]
Ludwig, D., 2000: Limitations of economic valuation of ecosystems. Ecosystems, 3, 31-35. Clean
Luo, Y.W.S., D. Hui and L.L. Wallace, 2001: Acclimatization of soil respiration to warming in a tall grass prairie. Nature, 413, 622-625. [JoC]
Lusseau, D., R. Williams, B. Wilson, K. Grellier, T.R. Barton, P.S. Hammond and P.M. Thompson, 2004: Parallel influence of climate on the behaviour of Pacific killer whales and Atlantic bottlenose dolphins. Ecol. Lett., 7, 1068-1076. Clean
Luterbacher, J., D. Dietrich, E. Xoplaki, M. Grosjean and H. Wanner, 2004: European seasonal and annual temperature variability, trends, and extremes since 1500. Science, 303, 1499-1503. [JoC]
MacArthur, R.H., 1972: Geographical Ecology Patterns in the Distribution of Species. Harper and Row, New York, 269 pp. [NPR]
Mack, M.C., E.A.G. Schuur, M.S. Bret-Harte, G.R. Shaver and F.S. Chapin, 2004: Ecosystem carbon storage in arctic tundra reduced by long-term nutrient fertilization. Nature, 431, 440-443. [JoC]
Mack, R.N., D. Simberloff, W.M. Lonsdale, H. Evans, M. Clout and F. Bazzaz, 2000: Biotic invasions: causes, epidemiology, global consequences and control. Issues Ecol., 5, 2-22. Clean
Malcolm, J.R., C. Liu, L. Miller, T. Allnut and L. Hansen, Eds., 2002a: Habitats at Risk: Global Warming and Species Loss in Globally Significant Terrestrial Ecosystems. WWF World Wide Fund for Nature, Gland, 40 pp. [NPR, ARC]
Malcolm, J.R., A. Markham, R.P. Neilson and M. Garaci, 2002b: Estimated migration rates under scenarios of global climate change. J. Biogeogr., 29, 835-849. [MoS, SRC]
Malcolm, J.R., C.R. Liu, R.P. Neilson, L. Hansen and L. Hannah, 2006: Global warming and extinctions of endemic species from biodiversity hotspots. Conserv. Biol., 20, 538-548. [SRC]
Malhi, Y. and O.L. Phillips, 2004: Tropical forests and global atmospheric change: a synthesis. Philos. T. Roy. Soc. Lond. B, 359, 549-555. Clean
Malhi, Y., D. Wood, T.R. Baker, J. Wright, O.L. Phillips, T. Cochrane, P. Meir, J. Chave, S. Almeida, L. Arroyo, N. Higuchi, T.J. Killeen, S.G. Laurance, W.F. Laurance, S.L. Lewis, A. Monteagudo, D.A. Neill, P.N. Vargas, N.C.A. Pitman, C.A. Quesada, R. Salomao, J.N.M Silva, A.T. Lezama, J. Terborgh, R.V. Martinez and B. Vinceti, 2006: The regional variation of aboveground live biomass in old-growth Amazonian forests. Global Change Biol., 12, 1107-1138. [JoC]
Maroco, J.P., E. Breia, T. Faria, J.S. Pereira and M.M. Chaves, 2002: Effects of long-term exposure to elevated CO2 and N fertilization on the development of photosynthetic capacity and biomass accumulation in Quercus suber L. Plant Cell Environ., 25, 105-113. Clean
Marra, J., C. Ho and C.C. Trees, 2003: An alternative algorithm for the calculation of primary productivity from remote sensing data. LDEO Technical Report LDEO-2003-1, Lamont-Doherty Earth Observatory, Columbia University, Palisades, New York, 27 pp. [NPR]
Martinez-Meyer, E., A.T. Peterson and W.W. Hargrove, 2004: Ecological niches as stable distributional constraints on mammal species, with implications for Pleistocene extinctions and climate change projections for biodiversity. Global Ecol. Biogeogr., 13, 305-314. [MoS]
Martinez-Vilalta, J. and J. Pinol, 2002: Drought-induced mortality and hydraulic architecture in pine populations of the NE Iberian Peninsula. Forest Ecol. Manag., 161, 247-256. Clean
Marubini, F., H. Barnett, C. Langdon and M.J. Atkinson, 2001: Dependence of calcification on light and carbonate ion concentration for the hermatypic coral Porites compressa. Mar. Ecol.–Prog. Ser., 220, 153-162. [NPR]
Marubini, F., C. Ferrier-Pages and J.P. Cuif, 2003: Suppression of skeletal growth in scleractinian corals by decreasing ambient carbonate-ion concentration: a cross-family comparison. P. Roy. Soc. Lond. B, 270, 179-184. Clean
Masek, J.G., 2001: Stability of boreal forest stands during recent climate change: evidence from Landsat satellite imagery. J. Biogeogr., 28, 967-976. Clean
Matson, P., K.A. Lohse and S.J. Hall, 2002: The globalization of nitrogen deposition: consequences for terrestrial ecosystems. Ambio, 31, 113-119. Clean
Matsui, T., T. Yagihashi, T. Nakaya, H. Taoda, S. Yoshinaga, H. Daimaru and N. Tanaka, 2004: Probability distributions, vulnerability and sensitivity in Fagus crenata forests following predicted climate changes in Japan. J. Veg. Sci., 15, 605-614. [MoS]
May, R., 1999a: How the biosphere is organized. Science, 286, 2091-2091. [JoC]
May, R., 1999b: Unanswered questions in ecology. Philos. T. Roy. Soc. Lond. B, 354, 1951-1959. Clean
McClain, C.R., S.R. Signorini and J.R. Christian, 2004: Subtropical gyre variability observed by ocean-color satellites. Deep-Sea Res. Pt. II, 51, 281-301. Clean
McClanahan, T.R., N.A. Muthiga and S. Mangi, 2001: Coral and algal changes after the 1998 coral bleaching: interaction with reef management and herbivores on Kenyan reefs. Coral Reefs, 19, 380-391. Clean
McClean, C.J., J.C. Lovett, W. Kuper, L. Hannah, J.H. Sommer, W. Barthlott, M. Termansen, G.E. Smith, S. Tokamine and J.R.D Taplin, 2005: African plant diversity and climate change. Ann. Mo. Bot. Gard., 92, 139-152. Clean
McDonald, K.A. and J.H. Brown, 1992: Using montane mammals to model extinctions due to global change. Conserv. Biol., 6, 409-415. [MoS]
McGuire, A.D. and F.S. Chapin, 2006: Climate feedbacks in the Alaskan boreal forest. Alaska’s Changing Boreal Forest, F.S. Chapin, M.W. Oswood, K. van Cleve, L.A. Viereck and D.L. Verbyla, Eds., Oxford University Press, New York, 309-322. [NPR]
McGuire, A.D., F.S. Chapin III, C. Wirth, M. Apps, J. Bhatti, T. Callaghan, T.R. Christensen, J.S. Clein, M. Fukuda, T. Maxima, A. Onuchin, A. Shvidenko and E. Vaganov, 2007: Responses of high latitude ecosystems to global change: Potential consequences for the climate system. Terrestrial Ecosystems in a Changing World, J.G. Canadell, D.E. Pataki and L.F. Pitelka, Eds., Springer-Verlag, Berlin, 297-310. [NPR, SRC, 2007]
McKee, D., D. Atkinson, S.E. Collings, J.W. Eaton, A.B. Gill, I. Harvey, K. Hatton, T. Heyes, D. Wilson and B. Moss, 2003: Response of freshwater microcosm communities to nutrients, fish, and elevated temperature during winter and summer. Limnol. Oceanogr., 48, 707-722. Clean
McLachlan, J.S., J.S. Clark and P.S. Manos, 2005: Molecular indicators of tree migration capacity under rapid climate change. Ecology, 86, 2088-2098. Clean
McLean, R.F., A. Tsyban, V. Burkett, J.O. Codignotto, D.L. Forbes, N. Mimura, R.J. Beamish and V. Ittekkot, 2001: Coastal zones and marine ecosystems. Climate Change 2001: Impacts, Adaptation, and Vulnerability. Contribution of Working Group II to the Third Assessment Report of the Intergovernmental Panel on Climate Change, J.J. McCarthy, O.F. Canziani, N.A. Leary, D.J. Dokken and K.S. White, Eds., Cambridge University Press, Cambridge, 343-379. [NPR, ARC]
McNeely, J.A., 1990: Climate change and biological diversity: policy implications. Landscape: Ecological Impact of Climatic Change, M.M. Boer and R.S. de Groot, Eds., IOS Press, Amsterdam, 406-429. [NPR]
McNeely, J.A. and F. Schutyser, Eds., 2003: Protected Areas in 2023: Scenarios for an Uncertain Future. IUCN: The World Conservation Union, Gland, 51 pp. [NPR, MoS]
McNeil, B.I., R.J. Matear and D.J. Barnes, 2004: Coral reef calcification and climate change: the effect of ocean warming. Geophys. Res. Lett., 31, L22309, doi:10.1029/2004GL021541. [JoC]
McWilliams, J.P., I.M. Côté, J.A. Gill, W.J. Sutherland and A.R. Watkinson, 2005: Accelerating impacts of temperature-induced coral bleaching in the Caribbean. Ecology, 86, 2055-2060. [ARC]
Mearns, L.O., F. Giorgi, P. Whetton, D. Pabon, M. Hulme and M. Lal, 2003: Guidelines for Use of Climate Scenarios Developed from Regional Climate Model Experiments. Intergovernmental Panel on Climate Change (IPCC): Data Distribution Centre (DDC), Norwich, Hamburg and New York, 38 pp. [NPR, MoS, ARC]
Meehl, G.A., T.F. Stocker, W. Collins, P. Friedlingstein, A. Gaye, J. Gregory, A. Kitoh, R. Knutti, J. Murphy, A. Noda, S. Raper, I. Watterson, A. Weaver and Z.-C. Zhao, 2007: Global climate projections. Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, S. Solomon, D. Qin, M. Manning, Z. Chen, M. Marquis, K.B. Averyt, M. Tignor and H.L. Miller, Eds., Cambridge University Press, Cambridge, 747-845. [NPR, ARC, 2007]
Megonigal, J.P., C.D. Vann and A.A. Wolf, 2005: Flooding constraints on tree (Taxodium distichum) and herb growth responses to elevated CO2. Wetlands, 25, 430-438. Clean
Metzger, M.J., M.D.A. Rounsevell, L. Acosta-Michlik, R. Leemans and D. Schröter, 2006: The vulnerability of ecosystem services to land use change. Agr. Ecosyst. Environ., 114, 69-85. [SRC]
Midgley, G.F., L. Hannah, D. Millar, M.C. Rutherford and L.W. Powrie, 2002: Assessing the vulnerability of species richness to anthropogenic climate change in a biodiversity hotspot. Global Ecol. Biogeogr., 11, 445-451. [SRC]
Midgley, G.F., L. Hannah, D. Millar, W. Thuiller and A. Booth, 2003: Developing regional and species-level assessments of climate change impacts on biodiversity in the Cape Floristic Region. Biol. Conserv., 112, 87-97. [SRC]
Midgley, G.F., R.A. Chapman, B. Hewitson, P. Johnston, M. De Wit, G. Ziervogel, P. Mukheibir, L. Van Niekerk, M. Tadross, B.W. Van Wilgen, B. Kgope, P.D. Morant, A. Theron, R.J. Scholes and G.G. Forsyth, 2005: A status quo, vulnerability and adaptation assessment of the physical and socio-economic effects of climate change in the western Cape. Report to the Western Cape Government, Cape Town, South Africa. CSIR Report No. ENV-S-C 2005-073, CSIR Environmentek, Stellenbosch, 170 pp. [NPR, SRC]
Midgley, G.F., G.O. Hughes, W. Thuiller and A.G. Rebelo, 2006: Migration rate limitations on climate change-induced range shifts in Cape Proteaceae. Divers. Distrib., 12, 555-562. [SRC]
Midgley, G.F., W. Thuiller and S.I. Higgins, 2007: Plant species migration as a key uncertainty in predicting future impacts of climate change on ecosystems: progress and challenges. Terrestrial Ecosystems in a Changing World, J.G. Canadell, D.E. Pataki and L.F. Pitelka, Eds., Springer-Verlag, Berlin, 149-160. [NPR, SRC, 2007]
Mieszkowska, N., M.A. Kendall, S.J. Hawkins, R. Leaper, P. Williamson, N.J. Hardman-Mountford and A.J. Southward, 2006: Changes in the range of some common rocky shore species in Britain: a response to climate change? Hydrobiologia, 555, 241-251. Clean
Miles, L.J., 2002: The impact of global climate change on tropical forest biodiversity in Amazonia. PhD thesis, University of Leeds, Leeds, 328pp. [NPR]
Milieu en Natuurplanbureau, 2005: Natuurbalans 2005. Rapportnummer 408763002, SDU Uitgevers – Wageningen Universiteit en Research Centrum, Rijksinstituut voor Integraal Zoetwaterbeheer en Afvalwaterbehandeling, Rijksinstituut voor Kust en Zee, Den Haag, The Netherlands, 197 pp. [NPR]
Millar, C.I., R.D. Westfall, D.L. Delany, J.C. King and L.J. Graumlich, 2004: Response of subalpine conifers in the Sierra Nevada, California, USA, to 20th-century warming and decadal climate variability. Arct. Antarct. Alp. Res., 36, 181-200. Clean
Millennium Ecosystem Assessment, 2005: Millennium Ecosystem Assessment: Living Beyond our Means: Natural Assets and Human Well-being (Statement from the Board). Millennium Ecosystem Assessment, India, France, Kenya, UK, USA, Netherlands, Malaysia, 28 pp. Accessed 13.03.07: http://www.millenniumassessment.org/. [NPR]
Mills, E., 2005: Insurance in a climate of change. Science, 309, 1040-1044. [JoC, ARC]
Mitchell, J.F.B., T.C. Johns, J.M. Gregory and S.F.B. Tett, 1995: Climate response to increasing levels of greenhouse gases and sulphate aerosols. Nature, 376, 501-504. [JoC]
Mitchell, S.W. and F. Csillag, 2001: Assessing the stability and uncertainty of predicted vegetation growth under climatic variability: northern mixed grass prairie. Ecol. Model., 139, 101-121. [MoS]
Mock, G., Ed., 2005: World Resources 2005: The Wealth of the Poor – Managing Ecosystems to Fight Poverty. United Nations Development Programme, United Nations Environment Programme, World Bank, World Resources Institute, Washington, District of Columbia, 266 pp. [NPR]
Moldan, B., S. Percy, J. Riley, T. Hák, J. Rivera and F.L. Toth, 2005: Choosing responses. Ecosystems and Human Well-being: Volume 3: Policy Responses, K. Chopra, R. Leemans, P. Kumar and H. Simons, Eds., Island Press, Washington, District of Columbia, 527-548. [NPR, ARC]
Monahan, A.H., J.C. Fyfe and G.M. Flato, 2000: A regime view of Northern Hemisphere atmospheric variability and change under global warming. Geophys. Res. Lett., 27, 1139-1142. [JoC]
Monson, R.K., A.A. Turnipseed, J.P. Sparks, P.C. Harley, L.E. Scott-Denton, K. Sparks and T.E. Huxman, 2002: Carbon sequestration in a high-elevation, subalpine forest. Global Change Biol., 8, 459-478. [JoC]
Monson, R.K., D.L. Lipson, S.P. Burns, A.A. Turnipseed, A.C. Delany, M.W. Williams and S.K. Schmidt, 2006: Winter forest soil respiration controlled by climate and microbial community composition. Nature, 439, 711-714. [JoC]
Montenegro, G., R. Ginochio, A. Segura, J.E. Keeley and M. Gomez, 2004: Fire regimes and vegetation responses in two Mediterranean-climate regions. Rev. Chil. Hist. Nat., 77, 455-464. Clean
Mooney, H., A. Cropper and W. Reid, 2005: Confronting the human dilemma: how can ecosystems provide sustainable services to benefit society? Nature, 434, 561-562. [JoC]
Moorcroft, P.R., S.W. Pacala and M.A. Lewis, 2006: Potential role of natural enemies during tree range expansions following climate change. J. Theor. Biol., 241, 601-616. Clean
Morgan, J.A., D.R. Lecain, A.R. Mosier and D.G. Milchunas, 2001a: Elevated CO2 enhances water relations and productivity and affects gas exchange in C-3 and C-4 grasses of the Colorado shortgrass steppe. Global Change Biol., 7, 451-466. [JoC]
Morgan, M.G., L.F. Pitelka and E. Shevliakova, 2001b: Elicitation of expert judgments of climate change impacts on forest ecosystems. Climatic Change, 49, 279-307. [JoC]
Morgan, J.A., D.E. Pataki, C. Körner, H. Clark, S.J. Del Grosso, J.M. Grunzweig, A.K. Knapp, A.R. Mosier, P.C.D. Newton, P.A. Niklaus, J.B. Nippert, R.S. Nowak, W.J. Parton, H.W. Polley and M.R. Shaw, 2004: Water relations in grassland and desert ecosystems exposed to elevated atmospheric CO2. Oecologia, 140, 11-25. [SRC]
Moriondo, M., P. Good, R. Durao, M. Bindi, C. Giannakopoulos and J. Corte Real, 2006: Potential impact of climate change on fire risk in the Mediterranean area. Climate Res., 31, 85-95. [ARC]
Moser, D., S. Dullinger, T. Englisch, H. Niklfeld, C. Plutzar, N. Sauberer, H.G. Zechmeister and G. Grabherr, 2005: Environmental determinants of vascular plant species richness in the Austrian Alps. J. Biogeogr., 32, 1117-1127. Clean
Moss, B., D. McKee, D. Atkinson, S.E. Collings, J.W. Eaton, A.B. Gill, I. Harvey, K. Hatton, T. Heyes and D. Wilson, 2003: How important is climate? Effects of warming, nutrient addition and fish on phytoplankton in shallow lake microcosms. J. Appl. Ecol., 40, 782-792. Clean
Moss, R.H. and S.H. Schneider, 2000: Uncertainties in the IPCC TAR: Recommendations to lead authors for more consistent assessment and reporting. Guidance Papers on the Cross Cutting Issues of the Third Assessment Report of the IPCC, R. Pachauri, T. Taniguchi and K. Tanaka, Eds., Geneva, 33-51. [NPR, PoC, ARC]
Mouillot, F., S. Rambal and R. Joffre, 2002: Simulating climate change impacts on fire frequency and vegetation dynamics in a Mediterranean-type ecosystem. Global Change Biol., 8, 423-437. [JoC, MoS]
Mouillot, F., J.P. Ratte, R. Joffre, J.M. Moreno and S. Rambal, 2003: Some determinants of the spatio-temporal fire cycle in a mediterranean landscape (Corsica, France). Landscape Ecol., 18, 665-674. [ARC]
Mouthon, J. and M. Daufresne, 2006: Effects of the 2003 heatwave and climatic warming on mollusc communities of the Saône: a large lowland river and of its two main tributaries (France). Global Change Biol., 12, 441-449. [JoC]
Murdiyarso, D. and L. Lebel, 2007: Southeast Asian fire regimes and land development policy. Terrestrial Ecosystems in a Changing World, J.G. Canadell, D.E. Pataki and L.F. Pitelka, Eds., Springer-Verlag, Berlin, 261-271. [NPR, ARC, 2007]
Musil, C.F., U. Schmiedel and G.F. Midgley, 2005: Lethal effects of experimental warming approximating a future climate scenario on southern African quartz-field succulents: a pilot study. New Phytol., 165, 539-547. [MoS, SRC]
Myers, N., R.A. Mittermeier, C.G. Mittermeier, G.A.B. da Fonseca and J. Kent, 2000: Biodiversity hotspots for conservation priorities. Nature, 403, 853-858. [JoC]
Nabuurs, G.J., O. Masera, K. Andrasko, P. Benitez-Ponce, R. Boer, M. Dutschke, E. Elsiddig, J. Ford-Robertson, P. Frumhoff, T. Karjalainen, O. Krankina, W. Kurz, M. Matsumoto, W. Oyhantcabal, N.H. Ravindranath, M.S. Sanchez and X. Zhang, 2007: Forestry. Climate Change 2007: Mitigation. Contribution of Working Group III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, B. Metz, O. Davidson, P. Bosch, R. Dave and L. Meyer, Eds., Cambridge University Press, UK. [NPR, 2007]
Naiman, R.J., H. Dêcamps and M.E. McClain, 2005: Riparia: Ecology, Conservation and Management of Streamside Communities. Elsevier, Amsterdam, 448 pp. [NPR]
Najjar, R.G., H.A. Walker, E.J. Barron, R.J. Bord, J.R. Gibson, V.S. Kennedy, C.G. Knight, J.P. Megonigal, R.E. O’Connor, C.D. Polsky, N.P. Psuty, B.A. Richards, L.G. Sorenson, E.M. Steele and R.S. Swanson, 2000: The potential impacts of climate change on the mid-Atlantic coastal region. Climate Res., 14, 219-233. Clean
Naki?enovi? , N., J. Alcamo, G. Davis, B. de Vries, J. Fenhann, S. Gaffin, K. Gregory, A. Grübler, T.Y. Jung, T. Kram, E. Lebre la Rovere, L. Michaelis, S. Mori, T. Morita, W. Pepper, H. Pitcher, L. Price, K. Riahi, A. Roehrl, H.H. Rogner, A. Sankovski, M. Schlesinger, P. Shukla, S. Smith, R. Swart, S. van Rooijen, N. Victor and Z. Dadi, Eds., 2000: Emissions Scenarios: A Special Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, 599 pp. [NPR, ARC]
National Research Council, 1999: Perspectives on Biodiversity: Valuing its Role in an Everchanging World. National Academies Press, Washington, District of Columbia, 168 pp. [NPR]
Neilson, R.P. and R.J. Drapek, 1998: Potentially complex biosphere responses to transient global warming. Global Change Biol., 4, 505-521. [JoC, SRC]
Neilson, R.P., I.C. Prentice, B. Smith, T. Kittel and D. Viner, 1998: Simulated changes in vegetation distribution under global warming. Regional Impacts of Climatic Change: An Assessment of Vulnerability. A Special Report of the Intergovernmental Panel on Climate Change (IPCC) Working Group II, R.T. Watson, M.C. Zinyowera, R.H. Moss and D.J. Dokken, Eds., Cambridge University Press, Annex C, 439-456. [NPR, SRC]
Neilson, R.P., L.F. Pitelka, A.M. Solomon, R. Nathan, G.F. Midgley, J.M.V. Fragoso, H. Lischke and K. Thompson, 2005: Forecasting regional to global plant migration in response to climate change. BioScience, 55, 749-759. [MoS, SRC]
Nelson, G.C., 2005: Drivers of ecosystem change: summary chapter. Ecosystems and Human Well-being: Volume 1: Current State and Trends, R. Hassan, R. Scholes and N. Ash, Eds., Island Press, Washington, District of Columbia, 73-76. [NPR]
Nelson, J.A., J.A. Morgan, D.R. Lecain, A. Mosier, D.G. Milchunas and B.A. Parton, 2004: Elevated CO2 increases soil moisture and enhances plant water relations in a long-term field study in semi-arid shortgrass steppe of Colorado. Plant Soil, 259, 169-179. Clean
Nemani, R.R., C.D. Keeling, H. Hashimoto, W.M. Jolly, S.C. Piper, C.J. Tucker, R.B. Myneni and S.W. Running, 2003: Climate-driven increases in global terrestrial net primary production from 1982 to 1999. Science, 300, 1560-1563. [JoC, ARC]
Nepstad, D., P. Lefebvre, U.L. Da Silva, J. Tomasella, P. Schlesinger, L. Solorzano, P. Moutinho, D. Ray and J.G. Benito, 2004: Amazon drought and its implications for forest flammability and tree growth: a basin-wide analysis. Global Change Biol., 10, 704-717. [JoC]
Nepstad, D.C., A. Verissimo, A. Alencar, C. Nobre, E. Lima, P. Lefebvre, P. Schlesinger, C. Potter, P. Moutinho, E. Mendoza, M. Cochrane and V. Brooks, 1999: Large-scale impoverishment of Amazonian forests by logging and fire. Nature, 398, 505-508. [JoC, ARC]
Neuvonen, S., 2004: Spatial and temporal variation in biodiversity in the European North. SCANNET: Scandinavian/North European Network of Terrestrial Field Bases Final Report Work Package 6, Kevo Subarctic Research Istitute University of Turku, Turku, 44 pp. [NPR]
Ni, J., 2001: Carbon storage in terrestrial ecosystems of China: estimates at different spatial resolutions and their responses to climate change. Climatic Change, 49, 339-358. [JoC, MoS]
Ni, J., S.P. Harrison, I.C. Prentice, J.E. Kutzbach and S. Sitch, 2006: Impact of climate variability on present and Holocene vegetation: a model-based study. Ecol. Model., 191, 469-486. [MoS]
Nicholls, R.J., F.M.J. Hoozemans and M. Marchand, 1999: Increasing flood risk and wetland losses due to global sea-level rise: regional and global analyses. Global Environ. Chang., 9, S69-S87. [JoC, ARC]
Nicholson, S., 2000: Land surface processes and Sahel climate. Rev. Geophys., 38, 117-139. Clean
Nicholson, S.E., 2001: Climatic and environmental change in Africa during the last two centuries. Climate Res., 17, 123-144. Clean
Nicholson, S.E., 2002: What are the key components of climate as a driver of desertification? Global Desertification: Do Humans Cause Deserts? J.F. Reynolds and D.M. Stafford-Smith, Eds., Dahlem University Press, Berlin, 41-57. [NPR]
Nicholson, S.E., C.J. Tucker and M.B. Ba, 1998: Desertification, drought, and surface vegetation: an example from the West African Sahel. B. Am. Meteorol. Soc., 79, 815-829. [JoC]
Nicholson, S.E., B. Some and B. Kone, 2000: An analysis of recent rainfall conditions in West Africa, including the rainy seasons of the 1997 El Niño and the 1998 La Niña years. J. Climate, 13, 2628-2640. [JoC]
Niklaus, P.A. and C.H. Körner, 2004: Synthesis of a six-year study of calcareous grassland responses to in situ CO2 enrichment. Ecol. Monogr., 74, 491-511. [SRC]
Niklaus, P.A., P.W. Leadley, C.H. Körner and B. Schmid, 2001: A long-term field study on biodiversity * elevated CO2 interactions in grassland. Ecol. Monogr., 71, 341-356. [SRC]
Nippert, J., A. Knapp and J. Briggs, 2006: Intra-annual rainfall variability and grassland productivity: can the past predict the future? Plant Ecol., 184, 75-87. [MoS]
Nnadozie, K.C., 1998: Legal and policy approaches at linking the desertification and climate change agenda in Nigeria. Paper presented at the Workshop on Climate Change, Biodiversity, and Desertification held during the 12th Session of the Global Biodiversity Forum (GBF12-Dakar/CCD COP2), Meridien President Dakar, Senegal, 15 pp. [NPR]
Norby, R.J. and Y.Q. Luo, 2004: Evaluating ecosystem responses to rising atmospheric CO2 and global warming in a multi-factor world. New Phytol., 162, 281-293. Clean
Norby, R.J., P.J. Hanson, E.G., O’Neill, T.J. Tschaplinski, J.F. Weltzin, R.A. Hansen, W.X. Cheng, S.D. Wullschleger, C.A. Gunderson, N.T. Edwards and D.W. Johnson, 2002: Net primary productivity of a CO2-enriched deciduous forest and the implications for carbon storage. Ecol. Appl., 12, 1261-1266. Clean
Norby, R.J., J. Ledford, C.D. Reilly, N.E. Miller and E.G. O’Neill, 2004: Fine-root production dominates response of a deciduous forest to atmospheric CO2 enrichment. P. Natl. Acad. Sci. USA, 101, 9689-9693. [JoC]
Norby, R.J., E.H. Delucia, B. Gielen, C. Calfapietra, C.P. Giardina, J.S. King, J. Ledford, H.R. McCarthy, D.J.P. Moore, R. Ceulemans, P. De Angelis, A.C. Finzi, D.F. Karnosky, M.E. Kubiske, M. Lukac, K.S. Pregitzer, G.E. Scarascia-Mugnozza, W.H. Schlesinger and R. Oren, 2005: Forest response to elevated CO2 is conserved across a broad range of productivity. P. Natl. Acad. Sci. USA, 102, 18052-18056. [JoC]
Norby, R.J., L.E. Rustad, J.S. Dukes, D.S. Ojima, W.J. Parton, S.J. Del Grosso, R.E. McMurtrie and D.P. Pepper, 2007: Ecosystem responses to warming and interacting global change factors. Terrestrial Ecosystems in a Changing World, J.G. Canadell, D.E. Pataki and L.F. Pitelka, Eds., Springer-Verlag, Berlin, 45-58. [NPR, 2007]
Novelli, P.C., K.A. Masarie, P.M. Lang, B.D. Hall, R.C. Myers and J.W. Elkins, 2003: Reanalysis of tropospheric CO trends: effects of the 1997–1998 wildfires. J. Geophys. Res. D, 108, 4464, doi:10.1029/2002JD003031. Clean
Novick, K.A., P.C. Stoy, G.G. Katul, D.S. Ellsworth, M.B.S. Siqueira, J. Juang and R. Oren, 2004: Carbon dioxide and water vapor exchange in a warm temperate grassland. Oecologia, 138, 259-274. Clean
Nowak, R.S., S.F. Zitzer, D. Babcock, V. Smith-Longozo, T.N. Charlet, J.S. Coleman, J.R. Seemann and S.D. Smith, 2004: Elevated atmospheric CO2 does not conserve soil water in the Mojave Desert. Ecology, 85, 93-99. Clean
Nunes, M.C.S., M.J. Vasconcelos, J.M.C. Pereira, N. Dasgupta and R.J. Alldredge, 2005: Land cover type and fire in Portugal: do fires burn land cover selectively? Landscape Ecol., 20, 661-673. Clean
O’Brien, K.L. and R.M. Leichenko, 2000: Double exposure: assessing the impacts of climate change within the context of economic globalization. Global Environ. Chang., 10, 221-232. [JoC, ARC]
O’Reilly, C.M., S.R. Alin, P.D. Plisnier, A.S. Cohen and B.A. McKee, 2003: Climate change decreases aquatic ecosystem productivity of Lake Tanganyika, Africa. Nature, 424, 766-768. [JoC, ARC]
Occhipinti-Ambrogi, A. and D. Savini, 2003: Biological invasions as a component of global change in stressed marine ecosystems. Mar. Pollut. Bull., 46, 542-551. Clean
Ogaya, R. and J. Peñuelas, 2003: Comparative seasonal gas exchange and chlorophyll fluorescence of two dominant woody species in a holm oak forest. Flora, 198, 132-141. Clean
Ogaya, R. and J. Peñuelas, 2004: Phenological patterns of Quercus ilex, Phillyrea latifolia, and Arbutus unedo growing under a field experimental drought. Ecoscience, 11, 263-270. Clean
Ogaya, R., J. Peñuelas, J. Martinez-Vilalta and M. Mangiron, 2003: Effect of drought on diameter increment of Quercus ilex, Phillyrea latifolia, and Arbutus unedo in a holm oak forest of NE Spain. Forest Ecol. Manag., 180, 175-184. Clean
Ogden, J., L. Basher and M. McGlone, 1998: Fire, forest regeneration and links with early human habitation: evidence from New Zealand. Ann. Bot., 81, 687-696. Clean
Ogutu, J.O. and N. Owen-Smith, 2003: ENSO, rainfall and temperature influences on extreme population declines among African savanna ungulates. Ecol. Lett., 6, 412-419. Clean
Olsson, L., L. Eklundh and J. Ardo, 2005: A recent greening of the Sahel: trends, patterns and potential causes. J. Arid Environ., 63, 556-566. Clean
Opdam, P. and D. Wascher, 2004: Climate change meets habitat fragmentation: linking landscape and biogeographical scale level in research and conservation. Biol. Conserv., 117, 285-297. Clean
Oren, R., D.S. Ellsworth, K.H. Johnsen, N. Phillips, B.E. Ewers, C. Maier, K.V.R. Schäfer, H. McCarthy, G. Hendrey, S.G. McNulty and G.G. Katul, 2001: Soil fertility limits carbon sequestration by forest ecosystems in a CO2-enriched atmosphere. Nature, 411, 469-472. [JoC]
Orr, J.C., V.J. Fabry, O. Aumont, L. Bopp, S.C. Doney, R.A. Feely, A. Gnanadesikan, N. Gruber, A. Ishida, F. Joos, R.M. Key, K. Lindsay, E. Maier-Reimer, R. Matear, P. Monfray, A. Mouchet, R.G. Najjar, G.K. Plattner, K.B. Rodgers, C.L. Sabine, J.L. Sarmiento, R. Schlitzer, R.D. Slater, I.J. Totterdell, M.F. Weirig, Y. Yamanaka and A. Yool, 2005: Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms. Nature, 437, 681-686. [PoC, JoC]
Osborne, C.P., P.L. Mitchell, J.E. Sheehy and F.I. Woodward, 2000: Modelling the recent historical impacts of atmospheric CO2 and climate change on Mediterranean vegetation. Global Change Biol., 6, 445-458. [JoC, MoS]
Osmond, B., G. Ananyev, J. Berry, C. Langdon, Z. Kolber, G. Lin, R. Monson, C. Nichol, U. Rascher, U. Schurr, S. Smith and D. Yakir, 2004: Changing the way we think about global change research: scaling up in experimental ecosystem science. Global Change Biol., 10, 393-407. [JoC]
Ostendorf, B., D.W. Hilbert and M.S. Hopkins, 2001: The effect of climate change on tropical rainforest vegetation pattern. Ecol. Model., 145, 211-224. Clean
Overpeck, J., J. Cole and P. Bartlein, 2005: A “paleoperspective” on climate variability and change. Climate Change and Biodiversity, T.E. Lovejoy and L. Hannah, Eds., Yale University Press, New Haven, Connecticut, 91-108. [NPR, PoC]
Overpeck, J.T., B.L. Otto-Bliesner, G.H. Miller, D.R. Muhs, R.B. Alley and J.T. Kiehl, 2006: Paleoclimatic evidence for future ice-sheet instability and rapid sea-level rise. Science, 311, 1747-1750. [PoC, JoC, MoS]
Pagiola, S., K. von Ritter and J.T. Bishop, 2004: Assessing the economic value of ecosystem conservation. Environment Department Paper No. 101, The World Bank Environment Department, Washington, District of Columbia, 66 pp. [NPR]
Parmesan, C., T.L. Root and M.R. Willig, 2000: Impacts of extreme weather and climate on terrestrial biota. B. Am. Meteorol. Soc., 81, 443-450. [JoC, ARC]
Parry, M.L., Ed., 2000: Assessment of potential effects and adaptations to climate change in Europe: The Europe Acacia Project. Report of concerted action of the environment programme of the Research Directorate General of the Commission of the European Communities, Jackson Environmental Institute, University of East Anglia, Norwich, 320 pp. [NPR, ARC]
Pauli, H., M. Gottfried, T. Dirnböck, S. Dullinger and G. Grabherr, 2003: Assessing the long-term dynamics of endemic plants at summit habitats. Alpine Biodiversity in Europe, L. Nagy, G. Grabherr, C. Körner and D.B.A. Thompson, Eds., Springer-Verlag, Berlin, 195-207. [NPR]
Pauly, D., J. Alder, A. Bakun, S. Heileman, K.H. Kock, P. Mace, W. Perrin, K. Stergiou, U.R. Sumaila, M. Vierros, K. Freire and Y. Sadovy, 2005: Marine fisheries systems. Ecosystems and Human Well-being: Volume 1: Current State and Trends, R. Hassan, R. Scholes and N. Ash, Eds., Island Press, Washington, District of Columbia, 477-511. [NPR]
Pausas, J.G. and D. Abdel Malak, 2004: Spatial and temporal patterns of fire and climate change in the eastern Iberian Peninsula (Mediterranean Basin). Ecology, Conservation and Management of Mediterranean Climate Ecosystems of the World, M. Arianoutsou and V.P. Papanastasis, Eds., 10th International Conference on Mediterranean Climate Ecosystems, Rhodes, Greece. Millpress, Rotterdam, 1-6. [NPR]
Pearce, D., 1998: Auditing the Earth: the value of the world’s ecosystem services and natural capital. Environment, 40, 23-28. Clean
Pearson, R.G., 2006: Climate change and the migration capacity of species. Trends Ecol. Evol., 21, 111-113. Clean
Pearson, R.G. and T.P. Dawson, 2003: Predicting the impacts of climate change on the distribution of species: are bioclimate envelope models useful? Global Ecol. Biogeogr., 12, 361-371. [MoS]
Pearson, R.G., T.P. Dawson and C. Liu, 2004: Modelling species distributions in Britain: a hierarchical integration of climate and land-cover data. Ecography, 27, 285-298. [MoS, ARC]
Peck, L.S., K.E. Webb and D.M. Bailey, 2004: Extreme sensitivity of biological function to temperature in Antarctic marine species. Funct. Ecol., 18, 625-630. Clean
Pederson, G.T., S.T. Gray, D.B. Fagre and L.J. Graumlich, 2006: Long-duration drought variability and impacts on ecosystem services: a case study from Glacier National Park, Montana. Earth Interactions, 10, 1-28. Clean
Peng, C. and M.J. Apps, 2000: Simulating global soil-CO2 flux and its response to climate change. J. Environ. Sci., 12, 257-265. [MoS]
Peñuelas, J. and M. Boada, 2003: A global change-induced biome shift in the Montseny mountains (NE Spain). Global Change Biol., 9, 131-140. [JoC]
Peñuelas, J., F. Lloret and R. Montoya, 2001: Severe drought effects on Mediterranean woody flora in Spain. Forest Sci., 47, 214-218. Clean
Perry, A.L., P.J. Low, J.R. Ellis and J.D. Reynolds, 2005: Climate change and distribution shifts in marine fishes. Science, 308, 1912-1915. [JoC]
Peters, R.L. and J.D.S. Darling, 1985: The greenhouse effect and nature reserves: global warming would diminish biological diversity by causing extinctions among reserve species. BioScience, 35, 707-717. Clean
Peterson, A.T., 2003: Projected climate change effects on Rocky Mountain and Great Plains birds: generalities of biodiversity consequences. Global Change Biol., 9, 647-655. [JoC]
Peterson, A.T., M.A. Ortega-Huerta, J. Bartley, V. Sanchez-Cordero, J. Soberon, R.H. Buddemeier and D.R.B. Stockwell, 2002: Future projections for Mexican faunas under global climate change scenarios. Nature, 416, 626-629. [JoC, MoS]
Petit, J.R., J. Jouzel, D. Raynaud, N.I. Barkov, J.M. Barnola, I. Basile, M. Benders, J. Chappellaz, M. Davis, G. Delaygue, M. Delmotte, V.M. Kotlyakov, M. Legrand, V.Y. Lipenkov, C. Lorius, L. Pépin, C. Ritz, E. Saltzman and M. Stievenard, 1999: Climate and atmospheric history of the past 420,000 years from the Vostok ice core, Antarctica. Nature, 399, 429-436. [JoC]
Peylin, P., P. Bousquet, C. Le Quere, S. Sitch, P. Friedlingstein, G. McKinley, N. Gruber, P. Rayner and P. Ciais, 2005: Multiple constraints on regional CO2 flux variations over land and oceans. Global Biogeochem. Cy., 19, GB1011, doi:10.1029/2003GB002214. Clean
Phillips, O.L., R.V. Martinez, L. Arroyo, T.R. Baker, T. Killeen, S.L. Lewis, Y. Malhi, A.M. Mendoza, D. Neill, P.N. Vargas, M. Alexiades, C. Ceron, A. Di Fiore, T. Erwin, A. Jardim, W. Palacios, M. Saldias and B. Vinceti, 2002: Increasing dominance of large lianas in Amazonian forests. Nature, 418, 770-774. [JoC]
Phillips, O.L., T.R. Baker, L. Arroyo, N. Higuchi, T.J. Killeen, W.F. Laurance, S.L. Lewis, J. Lloyd, Y. Malhi, A. Monteagudo, D.A. Neill, P.N. Vargas, J.N.M. Silva, J. Terborgh, R.V. Martinez, M. Alexiades, S. Almeida, S. Brown, J. Chave, J.A. Comiskey, C.I. Czimczik, A. Di Fiore, T. Erwin, C. Kuebler, S.G. Laurance, H.E.M. Nascimento, J. Olivier, W. Palacios, S. Patino, N.C.A. Pitman, C.A. Quesada, M. Salidas, A.T. Lezama and B. Vinceti, 2004: Pattern and process in Amazon tree turnover, 1976-2001. Philos. T. Roy. Soc. Lond. B, 359, 381-407. Clean
Phoenix, G.K. and J.A. Lee, 2004: Predicting impacts of Arctic climate change: past lessons and future challenges. Ecol. Res., 19, 65-74. [MoS]
Piao, S.L., J.Y. Fang and J.S. He, 2006: Variations in vegetation net primary production in the Qinghai-Xizang Plateau, China, from 1982 to 1999. Climatic Change, 74, 253-267. [JoC]
Piatkowski, U., D.F. Vergani and Z.B. Stanganelli, 2002: Changes in the cephalopod diet of southern elephant seal females at King George Island, during El Niño-La Niña events. J. Mar. Biol. Assoc. UK, 82, 913-916. Clean
Pickering, C., R. Good and K. Green, 2004: Potential Effects of Global Warming on the Biota of the Australian Alps. Australian Greenhouse Office, Australian Government, Canberra, 51 pp. [NPR]
Pielke, R.A., Sr., 2005: Land use and climate change. Science, 310, 1625-1626. [JoC]
Pierce, J.L., G.A. Meyer and A.J. Timothy Jull, 2004: Fire-induced erosion and millennial-scale climate change in northern ponderosa pine forests. Nature, 432, 87-90. [JoC]
Platt, T., C. Fuentes-Yaco and K.T. Frank, 2003: Spring algal bloom and larval fish survival. Nature, 423, 398-399. [JoC]
Podur, J., D.L. Martell and K. Knight, 2002: Statistical quality control analysis of forest fire activity in Canada. Can. J. Forest Res., 32, 195-205. Clean
Podur, J., D.L. Martell and F. Csillag, 2003: Spatial patterns of lightning-caused forest fires in Ontario, 1976-1998. Ecol. Model., 164, 1-20. Clean
Poff, L.N., M.M. Brinson and J.W.J. Day, 2002: Aquatic Ecosystems and Global Climate Change: Potential Impacts on Inland Freshwater and Coastal Wetland Ecosystems in the United States. Pew Center on Global Climate Change, Arlington, Virginia, 44 pp. [NPR]
Pounds, J.A., 2001: Climate and amphibian declines. Nature, 410, 639-640. [JoC]
Pounds, J.A. and R. Puschendorf, 2004: Ecology: clouded futures. Nature, 427, 107-109. [JoC, MoS]
Pounds, J.A., M.P.L. Fogden and J.H. Campbell, 1999: Biological response to climate change on a tropical mountain. Nature, 398, 611-615. [JoC]
Pounds, J.A., M.R. Bustamante, L.A. Coloma, J.A. Consuegra, M.P.L. Fogden, P.N. Foster, E. La Marca, K.L. Masters, A. Merino-Viteri, R. Puschendorf, S.R. Ron, G.A. Sanchez-Azofeifa, C.J. Still and B.E. Young, 2006: Widespread amphibian extinctions from epidemic disease driven by global warming. Nature, 439, 161-167. [JoC]
Prentice, I.C., P.J. Bartlein and T. Webb III, 1991: Vegetation and climate changes in eastern North America since the last glacial maximum. Ecology, 72, 2038-2056. Clean
Prentice, I.C., G.D. Farquhar, M.J.R. Fasham, M.L. Goulden, M. Heimann, V.J. Jaramillo, H.S. Kheshgi, C. Le Quéré, R.J. Scholes and D.W.R. Wallace, 2001: The carbon cycle and atmospheric carbon dioxide. Climate Change 2001: The Scientific Basis. Contribution of Working Group I to the Third Assessment Report of the Intergovernmental Panel on Climate Change, J.T. Houghton, Y. Ding, D.J. Griggs, M. Noguer, P.J. van der Linden, X. Dai, K. Maskell and C.A. Johnson, Eds., Cambridge University Press, Cambridge, 183-237. [NPR]
Prentice, I.C., A. Bondeau, W. Cramer, S.P. Harrison, T. Hickler, W. Lucht, S. Sitch, B. Smith and M.T. Sykes, 2007: Dynamic global vegetation modelling: quantifying terrestrial ecosystem responses to large-scale environmental change. Terrestrial Ecosystems in a Changing World, J.G. Canadell, D.E. Pataki and L.F. Pitelka, Eds., Springer-Verlag, Berlin, 175-192. [NPR, SRC, 2007]
Preston, B.L., 2006: Risk-based reanalysis of the effects of climate change on U.S. cold-water habitat. Climatic Change, 76, 91-119. [JoC]
Price, D.T., N.E. Zimmermann, P.J. van der Meer, M.J. Lexer, P. Leadley, I.T.M. Jorritsma, J. Schaber, D.F. Clark, P. Lasch, S. McNulty, J.G. Wu and B. Smith, 2001: Regeneration in gap models: Priority issues for studying forest responses to climate change. Climatic Change, 51, 475-508. [JoC, MoS]
Price, J.T. and T.L. Root, 2001: Climate change and Neotropical migrants. T. N. Am. Wildl. Nat. Resour., 66, 371-379. [SRC]
Price, J.T. and P. Glick, 2002: The Birdwatcher’s Guide to Global Warming. National Wildlife Federation and American Bird Conservancy, Washington, District of Columbia, 36 pp. [NPR, SRC]
Price, J.T. and T.L. Root, 2005: Potential Impacts of climate change on Neotropical migrants: management implications. Bird Conservation Implementation and Integration in the Americas, C.J. Ralph and T.D. Rich, Eds., USDA Forest Service, Arcata, California, 1123-1128. [NPR, SRC]
Prince, S.D., E.B. De Colstoun and L.L. Kravitz, 1998: Evidence from rain-use efficiencies does not indicate extensive Sahelian desertification. Global Change Biol., 4, 359-374. [JoC]
Prospero, J.M. and P.J. Lamb, 2003: African droughts and dust transport to the Caribbean: climate change implications. Science, 302, 1024-1027. [JoC]
Rabalais, N.N., R.E. Turner and W.J. Wiseman, 2002: Gulf of Mexico Hypoxia, a.k.a. “The Dead Zone”. Annu. Rev. Ecol. Syst., 33, 235-263. Clean
Rambal, S., J.M. Ourcival, R. Joffre, F. Mouillot, Y. Nouvellon, M. Reichstein and A. Rocheteau, 2003: Drought controls over conductance and assimilation of a Mediterranean evergreen ecosystem: scaling from leaf to canopy. Global Change Biol., 9, 1813-1824. [JoC]
Ramsay, M.A. and I. Stirling, 1988: Reproductive biology and ecology of female polar bears (Ursus maritimus). J. Zool., 214, 601-634. Clean
Randall, D., R. Wood, S. Bony, R. Coleman, T. Fichefet, J. Fyfe, V. Kattsov, A. Pitman, J. Shukla, J. Srinivasan, R.J. Stouffer, A. Sumi and K. Taylor, 2007: Climate models and their evaluation. Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, S. Solomon, D. Qin, M. Manning, Z. Chen, M. Marquis, K.B. Averyt, M. Tignor and H.L. Miller, Eds., Cambridge University Press, Cambridge, 589-662. [NPR, 2007]
Randerson, J.T., G.R. van der Werf, G.J. Collatz, L. Giglio, C.J. Still, P. Kasibhatla, J.B. Miller, J.W.C. White, R.S. Defries and E.S. Kasischke, 2005: Fire emissions from C-3 and C-4 vegetation and their influence on interannual variability of atmospheric CO2 and d13(CO2). Global Biogeochem. Cy., 19, GB2019, doi:10.1029/2004GB002366. Clean
Rasmussen, K., B. Fog and J.E. Madsen, 2001: Desertification in reverse? Observations from northern Burkina Faso. Global Environ. Chang., 11, 271-282. [JoC]
Rasmussen, L., C. Beier and A. Bergstedt, 2002: Experimental manipulations of old pine forest ecosystems to predict the potential tree growth effects of increased CO2 and temperature in a future climate. Forest Ecol. Manag., 158, 179-188. [MoS]
Raven, J., K. Caldeira, H. Elderfield, O. Hoegh-Guldberg, P. Liss, U. Riebesell, J. Shepherd, C. Turley and A. Watson, 2005: Ocean acidification due to increasing atmospheric carbon dioxide. Policy document 12/05, The Royal Society, The Clyvedon Press Ltd, Cardiff, 68 pp. [NPR, SRC]
Rebetez, M. and M. Dobbertin, 2004: Climate change may already threaten Scots pine stands in the Swiss Alps. Theor. Appl. Climatol., 79, 1-9. Clean
Reich, P.B., S.E. Hobbie, T. Lee, D.S. Ellsworth, J.B. West, D. Tilman, J.M.H. Knops, S. Naeem and J. Trost, 2006: Nitrogen limitation constrains sustainability of ecosystem response to CO2. Nature, 440, 922-925. [JoC]
Reichstein, M., J.D. Tenhunen, O. Roupsard, J.M. Ourcival, S. Rambal, F. Miglietta, A. Peressotti, M. Pecchiari, G. Tirone and R. Valentini, 2002: Severe drought effects on ecosystem CO2 and H2O fluxes at three Mediterranean evergreen sites: revision of current hypotheses? Global Change Biol., 8, 999-1017. [JoC]
Reid, W.V., H.A. Mooney, A. Cropper, D. Capistrano, S.R. Carpenter, K. Chopra, P. Dasgupta, T. Dietz, A.K. Duraiappah, R. Hassan, R. Kasperson, R. Leemans, R.M. May, A.J. McMichael, P. Pingali, C. Samper, R. Scholes, R.T. Watson, A.H. Zakri, Z. Shidong, N.J. Ash, E. Bennett, P. Kumar, M.J. Lee, C. Raudsepp-Hearne, H. Simons, J. Thonell and M.B. Zurek, Eds., 2005: Ecosystems and Human Well-being: Synthesis. Island Press, Washington, District of Columbia, 155 pp. [NPR, SRC]
Reij, C., G. Tappan and A. Belemvire, 2005: Changing land management practices and vegetation on the Central Plateau of Burkina Faso (1968-2002). J. Arid Environ., 63, 642-659. Clean
Reynaud, S., N. Leclercq, S. Romaine-Lioud, C. Ferrier-Pages, J. Jaubert and J.P. Gattuso, 2003: Interacting effects of CO2 partial pressure and temperature on photosynthesis and calcification in a scleractinian coral. Global Change Biol., 9, 1660-1668. [JoC]
Richards, M., 2003: Poverty reduction, equity and climate change: global governance synergies or contradictions? Globalisation and Poverty Programme. Government Report, Overseas Development Institute, London, 14 pp. Accessed 13.03.07: http://www.odi.org.uk. [NPR]
Richardson, A.J. and D.S. Schoeman, 2004: Climate impact on plankton ecosystems in the Northeast Atlantic. Science, 305, 1609-1612. [JoC]
Riebesell, U., I. Zondervan, B. Rost, P.D. Tortell, R.E. Zeebe and F.M.M. Morel, 2000: Reduced calcification of marine plankton in response to increased atmospheric CO2. Nature, 407, 364-367. [JoC]
Rietkerk, M., S.C. Dekker, P.C. de Ruiter and J. van de Koppel, 2004: Self-organized patchiness and catastrophic shifts in ecosystems. Science, 305, 1926-1929. [JoC]
Rillig, M.C., S.F. Wright, M.R. Shaw and C.B. Field, 2002: Artificial climate warming positively affects arbuscular mycorrhizae but decreases soil aggregate water stability in an annual grassland. Oikos, 97, 52-58. [ARC]
Roberts, C.M., C.J. McClean, J.E.N. Veron, J.P. Hawkins, G.R. Allen, D.E. McAllister, C.G. Mittermeier, F.W. Schueler, M. Spalding, F. Wells, C. Vynne and T.B. Werner, 2002: Marine biodiversity hotspots and conservation priorities for tropical reefs. Science, 295, 1280-1284. [JoC]
Robinson, R.A., J.A. Learmonth, A.M. Hutson, C.D. Macleod, T.H. Sparks, D.I. Leech, G.J. Pierce, M.M. Rehfisch and H.Q.P. Crick, 2005: Climate change and migratory species. BTO Research Report, Department for Environment, Food and Rural Affairs (Defra), London, 414 pp. [NPR, ARC]
Root, T.L. and S.H. Schneider, 1995: Ecology and climate: research strategies and implications. Science, 269, 334-341. [PoC, JoC, ARC]
Root, T.L., J.T. Price, K.R. Hall, S.H. Schneider, C. Rosenzweig and J.A. Pounds, 2003: Fingerprints of global warming on wild animals and plants. Nature, 421, 57-60. [PoC, JoC, SRC]
Ross, D.J., K.R. Tate, P.C.D. Newton and H. Clark, 2002: Decomposability of C3 and C4 grass litter sampled under different concentrations of atmospheric carbon dioxide at a natural CO2 spring. Plant Soil, 240, 275-286. Clean
Rouault, M., J.L. Mélice, C.J.C. Reason and J.R.E. Lutijeharms, 2005: Climate variability at Marion Island, Southern Ocean, since 1960. J. Geophys. Res. C, 110, C05007, doi:10.1029/2004JC002492. Clean
Rounsevell, M.D.A., P.M. Berry and P.A. Harrison, 2006: Future environmental change impacts on rural land use and biodiversity: a synthesis of the ACCELERATES project. Environ. Sci. Policy, 9, 93-100. [MoS, SRC]
Rustad, L.E., J.L. Campbell, G.M. Marion, R.J. Norby, M.J. Mitchell, A.E. Hartley, J.H.C. Cornelissen and J. Gurevitch, 2001: A meta-analysis of the response of soil respiration, net nitrogen mineralization, and aboveground plant growth to experimental ecosystem warming. Oecologia, 126, 543-562. Clean
Rutherford, M.C., G.F. Midgley, W.J. Bond, L.W. Powrie, R. Roberts and J. Allsopp, Eds., 2000: Plant Biodiversity: Vulnerability and Adaptation Assessment. Department of Environmental Affairs and Tourism, Pretoria, 59 pp. [NPR, SRC]
Sabine, C.L., M. Heimann, P. Artaxo, D.C.E. Bakker, C.T.A. Chen, C.B. Field and N. Gruber, 2004: Current status and past trends of the global carbon cycle. Global Carbon Cycle: Integrating Humans, Climate, and the Natural World, C.B. Field and M.R. Raupach, Eds., Island Press, Washington, Distict of Columbia, 17-44. [NPR, ARC]
Sadovy, Y., 2005: Trouble on the reef: the imperative for managing vulnerable and valuable fisheries. Fish Fish., 6, 167-185. Clean
Sala, O.E., 2005: Biodiversity across scenarios. Ecosystems and Human Well-being: Volume 2: Scenarios, S. Carpenter, P. Pingali, E. Bennett and M. Zurek, Eds., Island Press, Washington, District of Columbia, 375-408. [NPR]
Sala, O.E., I.F.S. Chapin, J.J. Armesto, E. Berlow, J. Bloomfield, R. Dirzo, E. Huber Sanwald, L.F. Huenneke, R.B. Jackson, A. Kinzig, R. Leemans, D.H. Lodge, H.A. Mooney, M. Oesterheld, N. Leroy Poff, M.T. Sykes, B.H. Walker, M. Walker and D.H. Wall, 2000: Global biodiversity scenarios for the year 2100. Science, 287, 1770-1774. [JoC, SRC]
Salvador, R., F. Lloret, X. Pons and J. Pinol, 2005: Does fire occurrence modify the probability of being burned again? A null hypothesis test from Mediterranean ecosystems in NE Spain. Ecol. Model., 188, 461-469. Clean
Sanchez, E., C. Gallardo, M.A. Gaertner, A. Arribas and M. Castro, 2004: Future climate extreme events in the Mediterranean simulated by a regional climate model: a first approach. Global Planet. Change, 44, 163-180. [MoS]
Sankaran, M., 2005: Fire, grazing and the dynamics of tall-grass savannas in the Kalakad-Mundanthurai Tiger Reserve, South India. Conserv. Soc., 3, 4-25. Clean
Sankaran, M., N.P. Hanan, R.J. Scholes, J. Ratnam, D.J. Augustine, B.S. Cade, J. Gignoux, S.I. Higgins, X. le Roux, F. Ludwig, J. Ardo, F. Banyikwa, A. Bronn, G. Bucini, K.K. Caylor, M.B. Coughenour, A. Diouf, W. Ekaya, C.J. Feral, E.C. February, P.G.H. Frost, P. Hiernaux, H. Hrabar, K.L. Metzger, H.H.T. Prins, S. Ringrose, W. Sea, J. Tews, J. Worden and N. Zambatis, 2005: Determinants of woody cover in African savannas. Nature, 438, 846-849. [JoC]
Sanz-Elorza, M., E.D. Dana, A. Gonzalez and E. Sobrino, 2003: Changes in the high-mountain vegetation of the central Iberian peninsula as a probable sign of global warming. Ann. Bot., 92, 273-280. Clean
Sardans, J. and J. Peñuelas, 2004: Increasing drought decreases phosphorus availability in an evergreen Mediterranean forest. Plant Soil, 267, 367-377. Clean
Sarmiento, J., 2000: That sinking feeling. Nature, 408, 155-156. [JoC]
Sarmiento, J.L. and N. Gruber, 2002: Sinks for anthropogenic carbon. Phys. Today, 55, 30-36. Clean
Sarmiento, J.L., T.M.C. Hughes, R.J. Stouffer and S. Manabe, 1998: Simulated response of the ocean carbon cycle to anthropogenic climate warming. Nature, 393, 245-249. [JoC, MoS]
Sarmiento, J.L., N. Gruber, M. Brzezinksi and J. Dunne, 2004a: High latitude controls of thermocline nutrients and low latitude biological productivity. Nature, 426, 56-60. [JoC]
Sarmiento, J.L., R. Slater, R. Barber, L. Bopp, S.C. Doney, A.C. Hirst, J. Kleypas, R. Matear, U. Mikolajewicz, P. Monfray, V. Soldatov, S.A. Spall and R. Stouffer, 2004b: Response of ocean ecosystems to climate warming. Global Biogeochem. Cy., 18, GB3003, doi:10.1029/2003GB002134. Clean
Sazonova, T.S., V.E. Romanovsky, J.E. Walsh and D.O. Sergueev, 2004: Permafrost dynamics in the 20th and 21st centuries along the East Siberian transect. J. Geophys. Res. D, 109, D01108, doi:10.1029/2003JD003680. [ARC]
Scavia, D., J.C. Field, D.F. Boesch, R.W. Buddemeier, V. Burkett, D.R. Cayan, M. Fogarty, M.A. Harwell, R.W. Howarth, C. Mason, D.J. Reed, T.C. Royer, A.H. Sallenger and J.G. Titus, 2002: Climate change impacts on U.S. coastal and marine ecosystems. Estuaries, 25, 149-164. [ARC]
Schäfer, K.V.R., R. Oren, D.S. Ellsworth, C.T. Lai, J.D. Herrick, A.C. Finzi, D.D. Richter and G.G. Katul, 2003: Exposure to an enriched CO2 atmosphere alters carbon assimilation and allocation in a pine forest ecosystem. Global Change Biol., 9, 1378-1400. [JoC]
Schallenberg, M., C.J. Hall and C.W. Burns, 2001: Climate change alters zooplankton community structure and biodiversity in coastal wetlands. Report of Freshwater Ecology Group, University of Otago, Hamilton, 9 pp. [NPR]
Schaphoff, S., W. Lucht, D. Gerten, S. Sitch, W. Cramer and I.C. Prentice, 2006: Terrestrial biosphere carbon storage under alternative climate projections. Climatic Change, 74, 97-122. [JoC, MoS, SRC]
Schär, C., P.L. Vidale, D. Lüthi, C. Frei, C. Häberli, M.A. Liniger and C. Appenzeller, 2004: The role of increasing temperature variability in European summer heatwaves. Nature, 427, 332-336. [JoC]
Scharlemann, J.P.W., R.E. Green and A. Balmford, 2004: Land-use trends in Endemic Bird Areas: global expansion of agriculture in areas of high conservation value. Global Change Biol., 10, 2046-2051. [JoC]
Scheffer, M., S. Carpenter, J.A. Foley, C. Folke and B. Walker, 2001: Catastrophic shifts in ecosystems. Nature, 413, 591-596. [JoC]
Schellnhuber, H.J., 2002: Coping with Earth system complexity and irregularity. Challenges of a Changing Earth, W. Steffen, D. Jäger and C. Bradshaw, Eds., Springer, Berlin, 151-156. [NPR]
Schimel, D. and D. Baker, 2002: Carbon cycle: the wildfire factor. Nature, 420, 29-30. [JoC]
Schimel, D.S., T.G.F. Kittel, S. Running, R. Monson, A. Turnipseed and D. Anderson, 2002: Carbon sequestration studied in Western US Mountains. EOS Transactions, 83, 445-449. [ARC]
Schimper, A.F.W., 1903: Plant Geography on a Physiological Basis. Clarendon Press, Oxford, 839 pp. [NPR]
Schlyter, P., I. Stjernquist, L. Bärring, A.M. Jönsson and C. Nilsson, 2006: Assessment of the impacts of climate change and weather extremes on boreal forests in northern Europe, focusing on Norway spruce. Climate Res., 31, 75-84. Clean
Schneider, S.H., 2004: Abrupt non-linear climate change, irreversibility and surprise. Global Environ. Chang., 14, 245-258. [PoC, JoC, ARC]
Schoennagel, T., T.T. Veblen and W.H. Romme, 2004: The interaction of fire, fuels, and climate across rocky mountain forests. BioScience, 54, 661-676. Clean
Scholes, R.J. and S.R. Archer, 1997: Tree–grass interactions in savannas. Annu. Rev. Ecol. Syst., 28, 517-544. Clean
Scholes, R.J., G. von Maltitz, M. de Wit, G.O. Hughes, G. Midgley and B. Erasmus, 2004: Helping Biodiversity Adapt to Climate Change. LUMONET: Science Development Network, Finnish Environment Institute, Helsinki, 2 pp. [NPR, SRC]
Scholze, M., W. Knorr, N.W. Arnell and I.C. Prentice, 2006: A climate change risk analysis for world ecosystems. P. Natl. Acad. Sci. USA, 103, 13116-13120. [JoC, ARC]
Schröder, A., L. Persson and A.M. De Roos, 2005: Direct experimental evidence for alternative stable states: a review. Oikos, 110, 3-19. Clean
Schrope, M.K., J.P. Chanton, L.H. Allen and J.T. Baker, 1999: Effect of CO2 enrichment and elevated temperature on methane emissions from rice, Oryza sativa. Global Change Biol., 5, 587-599. [NPR]
Schröter, D., W. Cramer, R. Leemans, I.C. Prentice, M.B. Araújo, N.W. Arnell, A. Bondeau, H. Bugmann, T.R. Carter, C.A. Gracia, A.C. de la Vega-Leinert, M. Erhard, F. Ewert, M. Glendining, J.I. House, S. Kankaanpää, R.J.T. Klein, S. Lavorel, M. Lindner, M.J. Metzger, J. Meyer, T.D. Mitchell, I. Reginster, M. Rounsevell, S. Sabaté, S. Sitch, B. Smith, J. Smith, P. Smith, M.T. Sykes, K. Thonicke, W. Thuiller, G. Tuck, S. Zaehle and B. Zierl, 2005: Ecosystem service supply and vulnerability to global change in Europe. Science, 310, 1333-1337. [JoC, SRC]
Schulze, E.D. and A. Freibauer, 2005: Carbon unlocked from soils. Nature, 437, 205-206. [JoC]
Schumacher, S. and H. Bugmann, 2006: The relative importance of climatic effects, wildfires and management for future forest landscape dynamics in the Swiss Alps. Global Change Biol., 12, 1435-1450. [JoC, MoS, SRC]
Schumacher, S., H. Bugmann and D.J. Mladenoff, 2004: Improving the formulation of tree growth and succession in a spatially explicit landscape model. Ecol. Model., 180, 175-194. [MoS, SRC]
Schumacher, S., B. Reineking, J. Sibold and H. Bugmann, 2006: Modelling the impact of climate and vegetation on fire regimes in mountain landscapes. Landscape Ecol., 21, 539-554. [MoS, SRC]
Schwinning, S. and O.E. Sala, 2004: Hierarchy of responses to resource pulses in arid and semi-arid ecosystems. Oecologia, 141, 211-220. Clean
Scott, D. and C. Lemieux, 2005: Climate change and protected area policy and planning in Canada. Forest Chron., 81, 696-703. [ARC]
Semazzi, F.H.M. and Y. Song, 2001: A GCM study of climate change induced by deforestation in Africa. Climate Res., 17, 169-182. Clean
Serreze, M.C., F. Carse, R.G. Barry and J.C. Rogers, 1997: Icelandic low cyclone activity: climatological features, linkages with the NAO, and relationships with recent changes in the Northern Hemisphere Circulation. J. Appl. Syst. Anal., 10, 453-464. [ARC]
Shafer, S.L., P.J. Bartlein and R.S. Thompson, 2001: Potential changes in the distributions of western North America tree and shrub taxa under future climate scenarios. Ecosystems, 4, 200-215. [MoS]
Shaw, M.R., E.S. Zavaleta, N.R. Chiariello, E.E. Cleland, H.A. Mooney and C.B. Field, 2002: Grassland responses to global environmental changes suppressed by elevated CO2. Science, 298, 1987-1990. [JoC, ARC]
Sheppard, C.R.C., 2003: Predicted recurrences of mass coral mortality in the Indian Ocean. Nature, 425, 294-297. [JoC, MoS]
Shiyatov, S.G., 2003: Rates of change in the upper treeline ecotone in the polar Ural Mountains. PAGES News, 11, 8-10. Clean
Shvidenko, A., C.V. Barber and R. Persson, 2005: Forest and woodland systems. Ecosystems and Human Well-being: Volume 1: Current State and Trends, R. Hassan, R. Scholes and N. Ash, Eds., Island Press, Washington, District of Columbia, 585-621. [NPR, ARC]
Siegenthaler, U., T.F. Stocker, E. Monnin, D. Luthi, J. Schwander, B. Stauffer, D. Raynaud, J.M. Barnola, H. Fischer, V. Masson-Delmotte and J. Jouzel, 2005: Stable carbon cycle-climate relationship during the late Pleistocene. Science, 310, 1313-1317. [JoC]
Simmons, R.E., P. Barnard, W.R.J. Dean, G.F. Midgley, W. Thuiller and G. Hughes, 2004: Climate change and birds: perspectives and prospects from southern Africa. Ostrich, 75, 295-308. [SRC]
Sims, D.W., V.J. Wearmouth, M.J. Genner, A.J. Southward and S.J. Hawkins, 2004: Low-temperature-driven early spawning migration of a temperate marine fish. J. Anim. Ecol., 73, 333-341. Clean
Singh, H.S., 2003: Vulnerability and adaptability of tidal forests in response to climate change in India. Indian Forester, 129, 749-756. Clean
Siqueira, M.F. and A.T. Peterson, 2003: Consequences of global climate change for geographic distributions of cerrado tree species. Biota Neotropica, 3, 1-14. Clean
Sitch, S., B. Smith, I.C. Prentice, A. Arneth, A. Bondeau, W. Cramer, J.O. Kaplan, S. Levis, W. Lucht, M.T. Sykes, K. Thonicke and S. Venevsky, 2003: Evaluation of ecosystem dynamics, plant geography and terrestrial carbon cycling in the LPJ dynamic global vegetation model. Global Change Biol., 9, 161-185. [JoC, MoS, SRC]
Sitch, S., V. Brovkin, W. von Bloh, D. van Vuuren, B. Assessment and A. Ganopolski, 2005: Impacts of future land cover changes on atmospheric CO2 and climate. Global Biogeochem. Cy., 19, GB2013, doi:10.1029/2004GB002311. [MoS, ARC]
Sitch, S., A.D. McGuire, J. Kimball, N. Gedney, J. Gamon, R. Emgstrom, A. Wolf, Q. Zhuang and J. Clein, 2007: Assessing the carbon balance of circumpolar Arctic tundra with remote sensing and process-based modeling approaches. Ecol. Appl., 17, 213-234. [MoS, 2007]
Skinner, W.R., M.D. Flannigan, B.J. Stocks, D.L. Martell, B.M. Wotton, J.B. Todd, J.A. Mason, K.A. Logan and E.M. Bosch, 2002: A 500 hPa synoptic wildland fire climatology for large Canadian forest fires, 1959-1996. Theor. Appl. Climatol., 71, 157-169. Clean
Skre, O., R. Baxter, R.M.M. Crawford, T.V. Callaghan and A. Fedorkov, 2002: How will the tundra-taiga interface respond to climate change? Ambio Special Report, 12, 37-46. [SRC]
Smith, J.B. and J.K. Lazo, 2001: A summary of climate change impact assessments from the US Country Studies Program. Climatic Change, 50, 1-29. [JoC, ARC]
Smith, L.C., Y. Sheng, G.M. MacDonald and L.D. Hinzman, 2005: Disappearing arctic lakes. Science, 308, 1429. [JoC]
Smith, M.D. and A.K. Knapp, 1999: Exotic plant species in a C-4-dominated grassland: invasibility, disturbance and community structure. Oecologia, 120, 605-612. Clean
Smith, R.D. and E. Malthby, 2003: Using the Ecosystem Approach to Implement the Convention on Biological Diversity: Key Issues and Case Studies. Island Press, Chicago, 118 pp. [NPR]
Smith, S.D., T.E. Huxman, S.F. Zitzer, T.N. Charlet, D.C. Housman, J.S. Coleman, L.K. Fenstermaker, J.R. Seemann and R.S. Nowak, 2000: Elevated CO2 increases productivity and invasive species success in an arid ecosystem. Nature, 408, 79-82. [JoC]
Smith, V.R., 2002: Climate change in the sub-Antarctic: an illustration from Marion Island. Climatic Change, 52, 345-357. [JoC]
Snyder, P.K., C. Delire and J.A. Foley, 2004: Evaluating the influence of different vegetation biomes on the global climate. Clim. Dynam., 23, 279-302. [JoC]
Solorzano, S., M.A. Castillo-Santiago, D.A. Navarrete-Gutierrez and K. Oyama, 2003: Impacts of the loss of neotropical highland forests on the species distribution: a case study using resplendent quetzal an endangered bird species. Biol. Conserv., 114, 341-349. Clean
Sorenson, L.G., R. Goldberg, T.L. Root and M.G. Anderson, 1998: Potential effects of global warming on waterfowl populations breeding in the Northern Great Plains. Climatic Change, 40, 343-369. [JoC, ARC]
Spehn, E. and C. Körner, 2005: A global assessment of mountain biodiversity and its function. Global Change and Mountain Regions: An Overview of Current Knowledge, U.M. Huber, H.K.M. Bugmann and M.A. Reasoner, Eds., Springer, Berlin, 393-400. [NPR, SRC]
Stabeno, P.J., N.A. Bond, N.B. Kachel, S.A. Salo and J.D. Schumacher, 2001: On the temporal variability of the physical environment over the south-eastern Bering Sea. Fish. Oceanogr., 10, 81-98. Clean
Stachowicz, J.J., J.R. Terwin, R.B. Whitlatch and R.W. Osman, 2002: Linking climate change and biological invasions: ocean warming facilitates nonindigenous species invasions. P. Natl. Acad. Sci. USA, 99, 15497-15500. [JoC]
Stampfli, A. and M. Zeiter, 2004: Plant regeneration directs changes in grassland composition after extreme drought: a 13-year study in southern Switzerland. J. Ecol., 92, 568-576. Clean
Stapp, P., G.A. Polls and F.S. Pirero, 1999: Stable isotopes reveal strong marine and El Niño effects on island food webs. Nature, 401, 467-469. [JoC]
Steffen, W., A. Sanderson, P.D. Tyson, J. Jäger, P.A. Matson, B. Moore, F. Oldfield, K. Richardson, H.-J. Schellnhuber, B.L. Turner II and R.J. Wasson, 2004: Global Change and the Earth System: A Planet Under Pressure. Springer, Berlin, 336 pp. [NPR]
Stenseth, N.C. and J.W. Hurrell, 2005: Global climate change: building links between the climate and ecosystem impact research communities. Climate Res., 29, 181-182. Clean
Stenseth, N.C., A. Mysterud, G. Ottersen, J.W. Hurrell, K.S. Chan and M. Lima, 2002: Ecological effects of climate fluctuations. Science, 297, 1292-1296. [JoC]
Stenseth, N.C., G. Ottersen, J.W. Hurrell, A. Mysterud, M. Lima, K.S. Chan, N.G. Yoccoz and B. Adlandsvik, 2003: Studying climate effects on ecology through the use of climate indices: the North Atlantic Oscillation, El Niño Southern Oscillation and beyond. P. Roy. Soc. Lond. B, 270, 2087-2096. Clean
Stern, N., 2007: The Economics of Climate Change: The Stern Review. Cambridge University Press, Cambridge, 692 pp. [NPR, 2007]
Stevens, G.C. and J.F. Fox, 1991: The causes of treeline. Annu. Rev. Ecol. Syst., 22, 177-191. Clean
Still, C.J., P.N. Foster and S.H. Schneider, 1999: Simulating the effects of climate change on tropical montane cloud forests. Nature, 398, 608-610. [PoC, JoC, MoS, ARC]
Stirling, I. and A.E. Derocher, 1993: Possible impacts of climatic warming on polar bears. Arctic, 46, 240-245. Clean
Stirling, I., N.J. Lunn and J. Iacozza, 1999: Long-term trends in the population ecology of polar bears in western Hudson Bay in relation to climatic change. Arctic, 52, 294-306. [SRC]
Stock, W.D., F. Ludwig, C. Morrow, G.F. Midgley, S.J.E. Wand, N. Allsopp and T.L. Bell, 2005: Long-term effects of elevated atmospheric CO2 on species composition and productivity of a southern African C-4 dominated grassland in the vicinity of a CO2 exhalation. Plant Ecol., 178, 211-224. [SRC]
Stocks, B.J., M.A. Fosberg, T.J. Lynham, L. Mearns, B.M. Wotton, Q. Yang, J.Z. Jin, K. Lawrence, G.R. Hartley, J.A. Mason and D.W. McKenney, 1998: Climate change and forest fire potential in Russian and Canadian boreal forests. Climatic Change, 38, 1-13. [JoC, ARC]
Stocks, B.J., J.A. Mason, J.B. Todd, E.M. Bosch, B.M. Wotton, B.D. Amiro, M.D. Flannigan, K.G. Hirsch, K.A. Logan, D.L. Martell and W.R. Skinner, 2002: Large forest fires in Canada, 1959-1997. J. Geophys. Res. D, 108, 8149, doi:10.1029/2001JD000484. Clean
Stokstad, E., 2001: U.N. report suggests slowed forest losses. Science, 291, 2294. [JoC]
Strengers, B., R. Leemans, B.J. Eickhout, B. de Vries and A.F. Bouwman, 2004: The land-use projections and resulting emissions in the IPCC SRES scenarios as simulated by the IMAGE 2.2 model. GeoJournal, 61, 381-393. [MoS, SRC]
Sturm, M., C. Racine and K. Tape, 2001: Increasing shrub abundance in the Arctic. Nature, 411, 546-547. [JoC]
Sturm, M., T. Douglas, C. Racine and G.E. Liston, 2005: Changing snow and shrub conditions affect albedo with global implications. J. Geophys. Res. G, 110, G01004, doi:10.1029/2005JG000013. Clean
Sumner, G.N., R. Romero, V. Homar, C. Ramis, S. Alonso and E. Zorita, 2003: An estimate of the effects of climate change on the rainfall of Mediterranean Spain by the late twenty-first century. Clim. Dynam., 20, 789-805. [JoC, MoS]
Symon, C., L. Arris and B. Heal, Eds., 2005: Arctic Climate Impact Assessment (ACIA): Scientific Report. Cambridge University Press, Cambridge, 1042 pp. [NPR]
Szmant, A.M., 2001: Why are coral reefs world-wide becoming overgrown by algae? ‘Algae, algae everywhere, and nowhere a bite to eat!’ Coral Reefs, 19, 299-302. Clean
Tape, K., M. Sturm and C. Racine, 2006: The evidence for shrub expansion in Northern Alaska and the Pan-Arctic. Global Change Biol., 12, 686-702. [JoC]
Theurillat, J.P. and A. Guisan, 2001: Potential impact of climate change on vegetation in the European Alps: a review. Climatic Change, 50, 77-109. [JoC, SRC]
Theurillat, J.P., F. Felber, P. Geissler, J.M. Gobat, M. Fierz, A. Fischlin, P. Küpfer, A. Schlüssel, C. Velluti, G.F. Zhao and J. Williams, 1998: Sensitivity of plant and soil ecosystems of the Alps to climate change. Views from the Alps: Regional Perspectives on Climate Change, P. Cebon, U. Dahinden, H.C. Davies, D.M. Imboden and C.C. Jager, Eds., MIT Press, Boston, Massachusetts, 225-308. [NPR, SRC]
Thirgood, S., A. Mosser, S. Tham, G. Hopcraft, E. Mwangomo, T. Mlengeya, M. Kilewo, J. Fryxell, A.R.E. Sinclair and M. Borner, 2004: Can parks protect migratory ungulates? The case of the Serengeti wildebeest. Anim. Conserv., 7, 113-120. Clean
Thomas, C.D., A. Cameron, R.E. Green, M. Bakkenes, L.J. Beaumont, Y.C. Collingham, B.F.N. Erasmus, M.F. de Siqueira, A. Grainger, L. Hannah, L. Hughes, B. Huntley, A.S. van Jaarsveld, G.F. Midgley, L. Miles, M.A. Ortega-Huerta, A.T. Peterson, O.L. Phillips and S.E. Williams, 2004a: Extinction risk from climate change. Nature, 427, 145-148. [JoC, SRC]
Thomas, C.D., S.E. Williams, A. Cameron, R.E. Green, M. Bakkenes, L.J. Beaumont, Y.C. Collingham, B.F.N. Erasmus, M.F de Siqueira, A. Grainger, L. Hannah, L. Hughes, B. Huntley, A.S. van Jaarsveld, G.F. Midgley, L. Miles, M.A. Ortega-Huerta, A.T. Peterson and O.L. Philipps, 2004b: Biodiversity conservation: uncertainty in predictions of extinction risk/Effects of changes in climate and land use/Climate change and extinction risk (reply). Nature, 430, 34. [JoC, MoS, SRC]
Thomas, D.S.G. and H.C. Leason, 2005: Dunefield activity response to climate variability in the southwest Kalahari. Geomorphology, 64, 117-132. Clean
Thonicke, K., S. Venevsky, S. Sitch and W. Cramer, 2001: The role of fire disturbance for global vegetation dynamics: coupling fire into a Dynamic Global Vegetation Model. Global Ecol. Biogeogr., 10, 661-677. [MoS, SRC]
Thuiller, W., S. Lavorel and M.B. Araujo, 2005a: Niche properties and geographical extent as predictors of species sensitivity to climate change. Global Ecol. Biogeogr., 14, 347. [MoS, SRC]
Thuiller, W., S. Lavorel, M.B. Araujo, M.T. Sykes and I.C. Prentice, 2005b: Climate change threats to plant diversity in Europe. P. Natl. Acad. Sci. USA, 102, 8245-8250. [JoC, SRC]
Thuiller, W., O. Broenniman, G. Hughes, J.R.M. Alkemades, G.F. Midgley and F. Corsi, 2006a: Vulnerability of African mammals to anthropogenic climate change under conservative land transformation assumptions. Global Change Biol., 12, 424-440. [JoC, SRC]
Thuiller, W., G.F. Midgley, G.O. Hughes, B. Bomhard, G. Drew, M.C. Rutherford and F.I. Woodward, 2006b: Endemic species and ecosystem sensitivity to climate change in Namibia. Global Change Biol., 12, 759-776. [JoC, SRC]
Timoney, K.P., 2003: The changing disturbance regime of the boreal forest of the Canadian Prairie Provinces. Forest Chron., 79, 502-516. Clean
Tinner, W. and A.F. Lotter, 2001: Central European vegetation response to abrupt climate change at 8.2 ka. Geology, 29, 551-554. Clean
Tol, R.S.J., 2001: Equitable cost-benefit analysis of climate change policies. Ecol. Econ., 36, 71-85. [ARC]
Toman, M., 1998a: Research frontiers in the economics of climate change. Environ. Resour. Econ., 11, 603-621. Clean
Toman, M., 1998b: Why not to calculate the value of the world’s ecosystem services and natural capital. Ecol. Econ., 25, 57-60. Clean
Tompkins, E.L. and W.N. Adger, 2003: Building resilience to climate change through adaptive management of natural resources. Working Paper 27, Tyndall Centre for Climate Change Research, Norwich, 19 pp. [NPR]
Trenberth, K.E., P.D. Jones, P.G. Ambenje, R. Bojariu, D.R. Easterling, A.M.G. Klein Tank, D.E. Parker, J.A. Renwick, F. Rahimzadeh, M.M. Rusticucci, B.J. Soden and P.-M. Zhai, 2007: Observations: surface and atmospheric climate change. Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, S. Solomon, D. Qin, M. Manning, Z. Chen, M. Marquis, K.B. Averyt, M. Tignor and H.L. Miller, Eds., Cambridge University Press, Cambridge, 235-336. [NPR, PoC, 2007]
Trigo, R.M., M.G. Pereira, J.M.C. Pereira, B. Mota, T.J. Calado, C.C. da Camara and F.E. Santo, 2005: The exceptional fire season of summer 2003 in Portugal. Geophys. Res. Abstracts, 7, 09690. Clean
Trigo, R.M., J.M.C. Pereira, M.G. Pereira, B. Mota, T.J. Calado, C.C. Dacamara and F.E. Santo, 2006: Atmospheric conditions associated with the exceptional fire season of 2003 in Portugal. Int. J. Climatol., 26, 1741-1757. [JoC]
Tuchman, N.C., R.G. Wetzel, S.T. Rier, K.A. Wahtera and J.A. Teeri, 2002: Elevated atmospheric CO2 lowers leaf litter nutritional quality for stream ecosystem food webs. Global Change Biol., 8, 163-170. [JoC]
Tuchman, N.C., K.A. Wahtera, R.G. Wetzel, N.M. Russo, G.M. Kilbane, L.M. Sasso and J.A. Teeri, 2003a: Nutritional quality of leaf detritus altered by elevated atmospheric CO2: effects on development of mosquito larvae. Freshwater Biol., 48, 1432-1439. Clean
Tuchman, N.C., K.A. Wahtera, R.G. Wetzel and J.A. Teeri, 2003b: Elevated atmospheric CO2 alters leaf litter quality for stream ecosystems: an in situ leaf decomposition study. Hydrobiologia, 495, 203-211. Clean
Turley, C., J. Blackford, S. Widdicombe, D. Lowe and P. Nightingale, 2006: Reviewing the impact of increased atmospheric CO2 on oceanic pH and the marine ecosystem. Avoiding Dangerous Climate Change, H.J. Schellnhuber, W. Cramer, N. Naki?enovi?, T.M.L. Wigley and G. Yohe, Eds., Cambridge University Press, Cambridge, 65-70. [NPR, SRC]
Tyrrell, T., P.M. Holligan and C.D. Mobley, 1999: Optical impacts of oceanic coccolithophore blooms. J. Geophys. Res. E, 104, 3223-3241, doi:10.1029/1998JC900052. Clean
UNEP, 2002: Global Environmental Outlook 3: Past, Present and Future Perspectives. A report from the GEO Project, United Nations Environment Programme, Nairobi, 416 pp. [NPR, MoS]
Usher, M.B., T.V. Callaghan, G. Gilchrist, B. Heal, G.P. Juday, H. Loeng, M.A.K. Muir and P. Prestrud, 2005: Principles of conserving the Arctic’s biodiversity. Arctic Climate Impact Assessment (ACIA): Scientific Report, C. Symon, L. Arris and B. Heal, Eds., Cambridge University Press, Cambridge, 539-596. [NPR, SRC]
Van Auken, O.W., 2000: Shrub invasions of North American semiarid grasslands. Annu. Rev. Ecol. Syst., 31, 197-215. Clean
Van Dam, R., H. Gitay, M. Finlayson, N.J. Davidson and B. Orlando, 2002: Climate Change and Wetlands: Impacts, Adaptation and Mitigation. Ramsar COP8 DOC. 11, Ramsar Bureau, The Ramsar Convention on Wetlands, Gland, 64 pp. [NPR]
van den Bergh, J.C.J.M., 2004: Optimal climate policy is a utopia: from quantitative to qualitative cost-benefit analysis. Ecol. Econ., 48, 385-393. Clean
van den Born, G.J., R. Leemans and M. Schaeffer, 2004: Climate change scenarios for dryland West Africa 1990-2050. The Impact of Climate Change on Drylands, A.J. Dietz, R. Ruben and A. Verhagen, Eds., Kluwer Academic, Dordrecht, 43-48. [NPR, SRC]
van der Werf, G.R., J.T. Randerson, G.J. Collatz, L. Giglio, P.S. Kasibhatla, A.F. Arellano, S.C. Olsen and E.S. Kasischke, 2004: Continental-scale partitioning of fire emissions during the 1997 to 2001 El Niño/La Niña period. Science, 303, 73-76. [JoC]
van Groenigen, K.J., J. Six, B.A. Hungate, M.A. de Graaff, N. van Breemen and C. van Kessel, 2006: Element interactions limit soil carbon storage. P. Natl. Acad. Sci. USA, 103, 6571-6574. [JoC]
Van Kessel, C., J. Nitschelm, W.R. Horwath, D. Harris, F. Walley, A. Luscher and U. Hartwig, 2000: Carbon-13 input and turn-over in a pasture soil exposed to long-term elevated atmospheric CO2. Global Change Biol., 6, 123-135. [JoC]
van Vliet, A. and R. Leemans, 2006: Rapid species responses to changes in climate requires stringent climate protection target. Avoiding Dangerous Climate Change, H.J. Schellnhuber, W. Cramer, N. Naki?enovi?, T.M.L. Wigley and G. Yohe, Eds., Cambridge University Press, Cambridge, 135-141. [NPR, SRC]
van Wijk, M.T., K.E. Clemmensen, G.R. Shaver, M. Williams, T.V. Callaghan, F.S. Chapin, J.H.C. Cornelissen, L. Gough, S.E. Hobbie, S. Jonasson, J.A. Lee, A. Michelsen, M.C. Press, S.J. Richardson and H. Rueth, 2004: Long-term ecosystem level experiments at Toolik Lake, Alaska, and at Abisko, Northern Sweden: generalizations and differences in ecosystem and plant type responses to global change. Global Change Biol., 10, 105-123. [JoC, SRC]
Van Wilgen, B.W., N. Govender, H.C. Biggs, D. Ntsala and X.N. Funda, 2004: Response of Savanna fire regimes to changing fire-management policies in a large African National Park. Conserv. Biol., 18, 1533-1540. Clean
Väre, H., R. Lampinen, C. Humphries and P. Williams, 2003: Taxonomic diversity of vascular plants in the European alpine areas. Alpine Biodiversity in Europe: A Europe-wide Assessment of Biological Richness and Change, L. Nagy, G. Grabherr, C.K. Körner and D.B.A. Thompson, Eds., Springer Verlag, Berlin, 133-148. [NPR]
Vazquez, A. and J.M. Moreno, 2001: Spatial distribution of forest fires in Sierra de Gredos (Central Spain). Forest Ecol. Manag., 147, 55-65. [ARC]
Veenendaal, E.M., O. Kolle and J. Lloyd, 2004: Seasonal variation in energy fluxes and carbon dioxide exchange for a broad-leaved semi-arid savanna (Mopane woodland) in Southern Africa. Global Change Biol., 10, 318-328. [JoC]
Velichko, A.A., Ed., 2002: Dynamics of Terrestrial Landscape Components and Inland and Marginal Seas of Northern Eurasia During the Last 130,000 Years. GEOS Press, Moscow, 296 pp. [NPR, SRC]
Velichko, A.A., N. Catto, A.N. Drenova, V.A. Klimanov, K.V. Kremenetski and V.P. Nechaev, 2002: Climate changes in East Europe and Siberia at the late glacial–Holocene transition. Quatern. Int., 91, 75-99. [SRC]
Velichko, A.A., O.K. Borisova, E.M. Zelikson and T.D. Morozova, 2004: Changes in vegetation and soils of the East European Plain to be expected in the 21st century due to the anthropogenic changes in climate. Geogr. Polonica, 77, 35-45. [SRC]
Velichko, A.A., E.Y. Novenko, V.V. Pisareva, E.M. Zelikson, T. Boettger and F.W. Junge, 2005: Vegetation and climate changes during the Eemian interglacial in Central and Eastern Europe: comparative analysis of pollen data. Boreas, 34, 207-219. [SRC]
Verburg, P., R.E. Hecky and H. Kling, 2003: Ecological consequences of a century of warming in Lake Tanganyika. Science, 301, 505-507. [JoC]
Vergani, D.F., Z.B. Stanganelli and D. Bilenca, 2004: Effects of El Niño and La Niña events on the sex ratio of southern elephant seals at King George Island. Mar. Ecol.–Prog. Ser., 268, 293-300. Clean
Vicente-Serrano, S.M., J.C. Gonzalez-Hidalgo, M. de Luis and J. Raventos, 2004: Drought patterns in the Mediterranean area: the Valencia region (eastern Spain). Climate Res., 26, 5-15. Clean
Villalba, R. and T.T. Veblen, 1997: Regional patterns of tree population age structures in northern Patagonia: climatic and disturbance influences. J. Ecol., 85, 113-124. Clean
Villers-Ruiz, L. and I. Trejo-Vazquez, 1998: Impacto del cambio climático en los bosques y áreas naturales protegidas de México (Impact of climatic change in forests and natural protected areas of Mexico). Interciencia, 23, 10-19. Clean
Virtanen, T., S. Neuvonen, A. Nikula, M. Varama and P. Niemelä, 1996: Climate change and the risks of Neodiprion sertifer outbreaks on Scots pine. Silva Fenn., 30, 169-177. Clean
Visser, M.E., C. Both and M.M. Lambrechts, 2004: Global climate change leads to mistimed avian reproduction. Adv. Ecol. Res., 35, 89-110. Clean
Vitousek, P.M., H.A. Mooney, J. Lubchenco and J.M. Melillo, 1997: Human domination of Earth’s ecosystems. Science, 277, 494-499. [JoC]
Vlassova, T.K., 2002: Human impacts on the tundra–taiga zone dynamics: the case of the Russian Lesotundra. Ambio Special Report , 12, 30-36. Clean
Volney, W.J.A. and R.A. Fleming, 2000: Climate change and impacts of boreal forest insects. Agr. Ecosyst. Environ., 82, 283-294. Clean
Von Maltitz, G.P., R.J. Scholes, B. Erasmus and A. Letsoalo, 2006: Adapting conservation strategies to accommodate impacts of climate change in southern Africa. Working paper No. 35, Assessments of Impacts and Adaptations to Climate Change (AIACC), Washington, District of Columbia, 53 pp. [NPR]
Wagner, W., K. Scipal, C. Pathe, D. Gerten, W. Lucht and B. Rudolf, 2003: Evaluation of the agreement between the first global remotely sensed soil moisture data with model and precipitation data. J. Geophys. Res. D, 108, 4611, doi:10.1029/2003JD003663. [MoS, SRC]
Walker, M.D., C.H. Wahren, R.D. Hollister, G.H.R. Henry, L.E. Ahlquist, J.M. Alatalo, M.S. Bret-Harte, M.P. Calef, T.V. Callaghan, A.B. Carroll, H.E. Epstein, I.S. Jonsdottir, J.A. Klein, B. Magnusson, U. Molau, S.F. Oberbauer, S.P. Rewa, C.H. Robinson, G.R. Shaver, K.N. Suding, C.C. Thompson, A. Tolvanen, O. Totland, P.L. Turner, C.E. Tweedie, P.J. Webber and P.A. Wookey, 2006: Plant community responses to experimental warming across the tundra biome. P. Natl. Acad. Sci. USA, 103, 1342-1346. [JoC, SRC]
Walsh, J.E., 1995: Long-term observations for monitoring of the cryosphere. Climatic Change, 31, 369-394. [JoC, ARC]
Walsh, J.E. and M.S. Timlin, 2003: Northern Hemisphere sea ice simulations by global climate models. Polar Res., 22, 75-82. [MoS, ARC]
Walter, K.M., S.A. Zimov, J.P. Chanton, D. Verbyla and F.S. Chapin, 2006: Methane bubbling from Siberian thaw lakes as a positive feedback to climate warming. Nature, 443, 71-75. [JoC]
Walther, G.R., C.A. Burga and P.J. Edwards, Eds., 2001: “Fingerprints” of Climate Change: Adaptive Behavior and Shifting Species Ranges. Kluwer, New York, 329 pp. [NPR]
Walther, G.R., E. Post, P. Convey, A. Menzel, C. Parmesan, T.J.C. Beebee, J.M. Fromentin, O. Hoegh-Guldberg and F. Bairlein, 2002: Ecological responses to recent climate change. Nature, 416, 389-395. [JoC, ARC]
Wang, G.L. and E.A.B. Eltahir, 2002: Impact of CO2 concentration changes on the biosphere-atmosphere system of West Africa. Global Change Biol., 8, 1169-1182. [JoC]
Wang, S.Q., J.Y. Liu, G.R. Yu, Y.Y. Pan, Q.M. Chen, K.R. Li and J.Y. Li, 2004a: Effects of land use change on the storage of soil organic carbon: a case study of the Qianyanzhou Forest Experimental Station in China. Climatic Change, 67, 247-255. [JoC]
Wang, X.L.L., F.W. Zwiers and V.R. Swail, 2004b: North Atlantic Ocean wave climate change scenarios for the twenty-first century. J. Climate, 17, 2368-2383. [JoC]
Wanner, H., R. Rickli, E. Salvisberg, C. Schmutz and M. Schüepp, 1997: Global climate change and variability and its influence on Alpine climate: concepts and observations. Theor. Appl. Climatol., 58, 221-243. Clean
Warner, T.T., 2004: Desert Meteorology. Cambridge University Press, Cambridge, 595 pp. [NPR]
Warren, A., 2005: The policy implications of Sahelian change. J. Arid Environ., 63, 660-670. Clean
Warren, R., 2006: Impacts of global climate change at different annual mean global temperature increases. Avoiding Dangerous Climate Change, H.J. Schellnhuber, W. Cramer, N. Naki?enovi?, T.M.L. Wigley and G. Yohe, Eds., Cambridge University Press, Cambridge, 93-131. [NPR, SRC]
Watson, R.T., I.R. Noble, B. Bolin, N.H. Ravindranath, D.J. Verardo and D.J. Dokken, Eds., 2000: Land Use, Land-Use Change, and Forestry: A Special Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, 377 pp. [NPR]
Weltzin, J.F., J. Pastor, C. Harth, S.D. Bridgham, K. Updegraff and C.T. Chapin, 2000: Response of bog and fen plant communities to warming and water-table manipulations. Ecology, 81, 3464-3478. Clean
Weltzin, J.F., C. Harth, S.D. Bridgham, J. Pastor and M. Vonderharr, 2001: Production and microtopography of bog bryophytes: response to warming and water-table manipulations. Oecologia, 128, 557-565. Clean
Weltzin, J.F., S.D. Bridgham, J. Pastor, J.Q. Chen and C. Harth, 2003: Potential effects of warming and drying on peatland plant community composition. Global Change Biol., 9, 141-151. [JoC]
Westerling, A.L., H.G. Hidalgo, D.R. Cayan and T.W. Swetnam, 2006: Warming and earlier spring increase western US forest wildfire activity. Science, 313, 940-943. [JoC, ARC]
Weyhenmeyer, G.A., 2004: Synchrony in relationships between the North Atlantic Oscillation and water chemistry among Sweden’s largest lakes. Limnol. Oceanogr., 49, 1191-1201. Clean
Weyhenmeyer, G.A., M. Meili and D.M. Livingstone, 2004: Nonlinear temperature response of lake ice breakup. Geophys. Res. Lett., 31, L07203, doi:10.1029/2004GL019530. [JoC]
White, A., M.G.R. Cannell and A.D. Friend, 2000a: CO2 stabilization, climate change and the terrestrial carbon sink. Global Change Biol., 6, 817-833. [JoC]
White, T.A., B.D. Campbell, P.D. Kemp and C.L. Hunt, 2000b: Sensitivity of three grassland communities to simulated extreme temperature and rainfall events. Global Change Biol., 6, 671-684. [JoC, MoS]
Whitehead, H., 1997: Sea surface temperature and the abundance of sperm whale calves off the Galapagos Islands: implications for the effects of global warming. Report of the International Whaling Commission, 47, 941-944. Clean
Whitlock, C., S.L. Shafer and J. Marlon, 2003: The role of climate and vegetation change in shaping past and future fire regimes in the northwestern US and the implications for ecosystem management. Forest Ecol. Manag., 178, 5-21. [MoS]
Whittingham, E., J. Campbell and P. Townsley, 2003: Poverty and Reefs. DFID-IMM-IOC/UNESCO, Exeter, 260 pp. [NPR]
Wichmann, M.C., F. Jeltsch, W.R.J. Dean, K.A. Moloney and C. Wissel, 2003: Implication of climate change for the persistence of raptors in arid savanna. Oikos, 102, 186-202. Clean
Wikelski, M. and C. Thom, 2000: Marine iguanas shrink to survive El Niño: changes in bone metabolism enable these adult lizards to reversibly alter their length. Nature, 403, 37-38. [JoC]
Wilby, R.L., S.P. Charles, E. Zorita, B. Timbal, P. Whetton and L.O. Mearns, 2004: Guidelines for Use of Climate Scenarios Developed from Statistical Downscaling Methods: IPCC Task Group on Data and Scenario Support for Impact and Climate Analysis (TGICA). Intergovernmental Panel on Climate Change (IPCC): Data Distribution Centre (DDC), Norwich, Hamburg and New York, 27 pp. [NPR, ARC]
Wiley, J.W. and J.M. Wunderle, Jr., 1994: The effects of hurricanes on birds, with special reference to Caribbean islands. Bird Conserv. Int., 3, 319-349. Clean
Wilkinson, C., Ed., 2004: Status of Coral Reefs of the World: 2004. Vols. 1 and 2. Global Coral Reef Monitoring Network and Australian Institute of Marine Science, Townsville, 303+557 pp. [NPR]
Williams, A.A.J., D.J. Karoly and N. Tapper, 2001: The sensitivity of Australian fire danger to climate change. Climatic Change, 49, 171-191. [JoC, ARC]
Williams, M.A., C.W. Rice, A. Omay and C. Owensby, 2004a: Carbon and nitrogen pools in a tallgrass prairie soil under elevated carbon dioxide. Soil Sci. Soc. Am. J., 68, 148-153. Clean
Williams, P., L. Hannah, S. Andelman, G. Midgley, M. Araujo, G. Hughes, L. Manne, E. Martinez-Meyer and R. Pearson, 2005: Planning for climate change: identifying minimum-dispersal corridors for the Cape proteaceae. Conserv. Biol., 19, 1063-1074. [SRC]
Williams, R.J., L.B. Hutley, G.D. Cook, J. Russell-Smith, A. Edwards and X.Y. Chen, 2004b: Assessing the carbon sequestration potential of mesic savannas in the Northern Territory, Australia: approaches, uncertainties and potential impacts of fire. Funct. Plant Ecol., 31, 415-422. Clean
Williams, S.E., E.E. Bolitho and S. Fox, 2003: Climate change in Australian tropical rainforests: an impending environmental catastrophe. P. Roy. Soc. Lond. B, 270, 1887-1892. Clean
Willig, M.R., D.M. Kaufman and R.D. Stevens, 2003: Latitudinal gradients of biodiversity: pattern, process, scale, and synthesis. Annu. Rev. Ecol. Evol. Syst., 34, 273-309. Clean
Wilmking, M., G.P. Juday, V.A. Barber and H.S.J. Zald, 2004: Recent climate warming forces contrasting growth responses of white spruce at treeline in Alaska through temperature thresholds. Global Change Biol., 10, 1724-1736. [JoC]
Winder, M. and D.E. Schindler, 2004: Climatic effects on the phenology of lake processes. Global Change Biol., 10, 1844-1856. [JoC]
Wingfield, J.C., G. Ramos-Fernandez, A.N.D. La Mora and H. Drummond, 1999: The effects of an “El Niño” Southern Oscillation event on reproduction in male and female blue-footed boobies, Sula nebouxii. Gen. Comp. Endocrl., 114, 163-172. [NPR]
Winnett, S.M., 1998: Potential effects of climate change on US forests: a review. Climate Res., 11, 39-49. Clean
Winslow, J.C., J. Hunt, E. Raymond and S.C. Piper, 2003: The influence of seasonal water availability on global C3 versus C4 grassland biomass and its implications for climate change research. Ecol. Model., 163, 153-173. Clean
Woodward, F.I. and M.R. Lomas, 2001: Integrating fluxes from heterogeneous vegetation. Global Ecol. Biogeogr., 10, 595-601. Clean
Woodward, F.I. and M.R. Lomas, 2004a: Simulating vegetation processes along the Kalahari transect. Global Change Biol., 10, 383-392. [JoC, MoS]
Woodward, F.I. and M.R. Lomas, 2004b: Vegetation dynamics: simulating responses to climatic change. Biol. Rev., 79, 643-670. [MoS]
Woodwell, G.M., 2004: Mountains: top down. Ambio Special Report, 13, 35-38. Clean
Wooldridge, S., T. Done, R. Berkelmans, R. Jones and P. Marshall, 2005: Precursors for resilience in coral communities in a warming climate: a belief network approach. Mar. Ecol.–Prog. Ser., 295, 157-169. [ARC]
Worm, B., E.B. Barbier, N. Beaumont, J.E. Duffy, C. Folke, B.S. Halpern, J.B.C. Jackson, H.K. Lotze, F. Micheli, S.R. Palumbi, E. Sala, K.A. Selkoe, J.J. Stachowicz and R. Watson, 2006: Impacts of biodiversity loss on ocean ecosystem services. Science, 314, 787-790. [JoC]
Wright, S.J., O. Calderón, A. Hernandéz and S. Paton, 2004: Are lianas increasing in importance in tropical forests? A 17-year record from Panama. Ecology, 85, 484-489. Clean
Wullschleger, S.D. and R.J. Norby, 2001: Sap velocity and canopy transpiration in a sweetgum stand exposed to free-air CO2 enrichment (FACE). New Phytol., 150, 489-498. Clean
Xenopoulos, M.A., D.M. Lodge, J. Alcamo, M. Marker, K. Schulze and D.P. Van Vuuren, 2005: Scenarios of freshwater fish extinctions from climate change and water withdrawal. Global Change Biol., 11, 1557-1564. [JoC, ARC]
Xu, D.Y. and H. Yan, 2001: A study of the impacts of climate change on the geographic distribution of Pinus koraiensis in China. Environ. Int., 27, 201-205. Clean
Yamano, H. and M. Tamura, 2004: Detection limits of coral reef bleaching by satellite remote sensing: simulation and data analysis. Remote Sens. Environ., 90, 86-103. [MoS]
Yoccoz, N.G. and R.A. Ims, 1999: Demography of small mammals in cold regions: the importance of environmental variability. Animal Responses to Global Change in the North, A. Hofgaard, J.P. Ball, K. Danell and T.V. Callaghan, Eds., Ecological Bulletin 47, Blackwell, Oxford, 137-144. [NPR]
Zalakevicius, M. and S. Svazas, 2005: Global climate change and its impact on wetlands and waterbird populations. Acta Zool. Lituanica, 15, 215-217. Clean
Zebisch, M., F. Wechsung and H. Kenneweg, 2004: Landscape response functions for biodiversity: assessing the impact of land-use changes at the country level. Landscape Urban Plan., 67, 157-172. Clean
Zeebe, R.E. and D. Wolf-Gladrow, 2001: CO2 in Seawater: Equilibrium, Kinetics, Isotopes. Elsevier, Amsterdam, 346 pp. [NPR]
Zepp, R.G., T.V. Callaghan and D.J. Erickson, 2003: Interactive effects of ozone depletion and climate change on biogeochemical cycles. Photochem. Photobiol. Sci., 2, 51-61. [SRC]
Zha, Y., J. Gao and Y. Zhang, 2005: Grassland productivity in an alpine environment in response to climate change. Area, 37, 332-340. Clean
Zhang, W., K.M. Parker, Y. Luo, S. Wan, L.L. Wallace and S. Hu, 2005: Soil microbial responses to experimental warming and clipping in a tallgrass prairie. Global Change Biol., 11, 266-277. [JoC]
Zheng, X., Z. Zhou, Y. Wang, J. Zhu, Y. Wang, J. Yue, Y. Shi, K. Kobayashi, K. Inubushi, Y. Huang, S.H. Han, Z.J. Xu, B.H. Xie, K. Butterbach-Bahl and L.X. Yang, 2006: Nitrogen-regulated effects of free-air CO2 enrichment on methane emissions from paddy rice fields. Global Change Biol., 12, 1717-1732. [JoC]
Zierl, B. and H. Bugmann, 2005: Global change impacts on hydrological processes in Alpine catchments. Water Resour. Res., 41, 1-13. [SRC]
Zimov, S.A., E.A.G. Schuur and F.S. Chapin, 2006: Permafrost and the global carbon budget. Science, 312, 1612-1613. [JoC]
Ziska, L.H., T.B. Moya, R. Wassmann, O.S. Namuco, R.S. Lantin, J.B. Aduna, E. Abao, K.F. Bronson, H.U. Neue and D. Olszyk, 1998: Long-term growth at elevated carbon dioxide stimulates methane emission in tropical paddy rice. Global Change Biol., 4, 657-665. [JoC]
Zöckler, C. and I. Lysenko, 2000: Waterbirds on the Edge: First Circumpolar Assessment of Climate Change Impact on Arctic Breeding Water Birds. WCMC Biodiversity Series No. 11, UNEP and World Conservation Monitoring Centre (WCMC), Cambridge, 27 pp.[NPR]
| Tag | Explanation | Occurrences | Percentages |
|---|---|---|---|
| PoC |
Person of Concern Key individual involved in CRU emails as defined in this spreadsheet. |
16 | 1% |
| JoC |
Journal of Concern A Journal which has published articles by one or more PoCs (Person of Concern) |
273 | 29% |
| MoS |
Model or Simulation Reference appears to be a model or simulation, not observation or experiment. |
130 | 14% |
| NPR |
Non Peer Reviewed Reference has no Journal or no Volume or no Pages or it has Editors. |
174 | 18% |
| SRC |
Self Reference Concern Author of a chapter containing references to own work. |
148 | 16% |
| ARC |
Paper authored or co-authored by person who is also in list of Authors of another chapter. |
113 | 12% |
| 2007 |
Paper dated 2007, when IPCC policy stated cutoff was December 2005 |
18 | 1% |
| Clean |
The reference was probably peer reviewed. |
303 | 33% |
NOTE: In the Summary of Reviewer Comments,
Accepted includes only those responses that were unambiguously Accepted.
There are other categories of responses that we have not yet quantified
due to inconsistencies of usage.
| Reviewer | Total Comments | Accepted | IPCC Roles | Papers |
|---|---|---|---|---|
| Government of Finland | 149 | 38 | 0 | 0 |
| Guasch, LL | 119 | 19 | 0 | 0 |
| Government of Switzerland | 118 | 17 | 0 | 0 |
| Hanson, CE | 106 | 16 | 1 | 2 |
| House, Joanna | 89 | 1 | 0 | 0 |
| Government of Korea | 67 | 10 | 0 | 0 |
| Harvey, D | 60 | 8 | 0 | 1 |
| Government of Germany | 50 | 6 | 0 | 0 |
| Government of Australia | 47 | 6 | 0 | 0 |
| Prentice, IC | 34 | 0 | 0 | 26 |
| Wood, PJ | 33 | 1 | 0 | 0 |
| Fleming, Richard | 32 | 0 | 0 | 0 |
| Hawkins, SJ | 31 | 9 | 0 | 10 |
| Berry, PM | 29 | 3 | 0 | 8 |
| Government of Netherlands | 27 | 0 | 0 | 0 |
| Crick, Humphrey | 27 | 5 | 0 | 0 |
| Government of USA | 26 | 8 | 0 | 0 |
| Goklany, IM | 26 | 1 | 0 | 15 |
| Government of Poland | 24 | 1 | 0 | 0 |
| Masters, Gregory | 17 | 0 | 0 | 0 |
| Kasischke, Eric | 15 | 2 | 0 | 0 |
| Government of Argentina | 15 | 0 | 0 | 0 |
| Government of Canada | 11 | 5 | 0 | 0 |
| Beier, C | 11 | 2 | 0 | 8 |
| Harley, Mike | 10 | 1 | 0 | 0 |
| Bernier, Pierre | 10 | 0 | 0 | 0 |
| Hansen, LJ | 8 | 0 | 0 | 4 |
| Thomas, CD | 7 | 0 | 0 | 20 |
| Epstein, PR | 6 | 1 | 0 | 11 |
| MacMynowski, DP | 6 | 0 | 2 | 3 |
| Fearnside, PM | 6 | 0 | 1 | 6 |
| Balzter, H | 6 | 0 | 0 | 1 |
| Government of UK | 6 | 1 | 0 | 0 |
| Pauli, H | 5 | 2 | 0 | 8 |
| Luo, Tianxiang | 5 | 3 | 0 | 0 |
| Abbink, Oscar | 5 | 0 | 0 | 0 |
| Government of China | 5 | 1 | 0 | 0 |
| Government of Norway | 5 | 1 | 0 | 0 |
| Marshall, P | 5 | 0 | 0 | 4 |
| Government of France | 4 | 0 | 0 | 0 |
| Rodriguez, AG | 3 | 0 | 0 | 0 |
| Anadon, R | 3 | 3 | 0 | 2 |
| Chapin, FS | 3 | 0 | 0 | 23 |
| Penuelas, J | 3 | 1 | 0 | 25 |
| Government of Sweden | 3 | 0 | 0 | 0 |
| Lapin, Milan | 3 | 0 | 0 | 0 |
| Morgan, Jack | 2 | 1 | 0 | 0 |
| Bernstein, Lenny | 2 | 0 | 0 | 0 |
| Government of Austria | 2 | 0 | 0 | 0 |
| Tanaka, N | 2 | 0 | 0 | 3 |
| Spencer, T | 1 | 1 | 1 | 4 |
| Tsunekawa, Atsushi | 1 | 0 | 0 | 0 |
| Shinoda, Masato | 1 | 0 | 0 | 0 |
| Barring, L | 1 | 0 | 0 | 6 |
| Totals | 1292 | 174 | 5 | 190 |
| Reviewer Type | Total Comments | Accepted | % Accepted |
|---|---|---|---|
| Govt | 559 | 94 | 16% |
| IPCC | 359 | 47 | 13% |
| Indep | 374 | 33 | 8% |
| Total | 1292 | 174 | 13% |
Main AR4 Index | Working Group WG2 Index | Table of Contents | Authors | Executive Summary | Annotated Text | References | Reviewer Comments