Working Group 3 - Chapter 7: Industry - (AR4-WG3-7)
Original at: http://www.ipcc.ch/publications_and_data/ar4/wg3/en/ch7.html
Main AR4 Index | Working Group WG3 Index | Table of Contents | Authors | Executive Summary | Annotated Text | References | Reviewer Comments
With the exception of Chapter and Section headings, all coloured text has been inserted by AccessIPCC. The non-coloured text is the IPCC original.
A number of emails from the Climate Research Unit (CRU) of the University of East Anglia were published on the Internet in November 2009. This has provided a window into the world of climate science.
We have identified a number of key individuals involved in the emails whom we have designated as Persons of Concern [PoC]; a Journal in which a PoC has published has been designated as a Journal of Concern [JoC].
This is not to suggest that we believe such papers are necessarily flawed, but rather that, as Joseph Alcamo noted at Bali in October 2009, "as policymakers and the public begin to grasp the multi-billion dollar price tag for mitigating and adapting to climate change, we should expect a sharper questioning of the science behind climate policy".
References occur in a list at the end of each chapter. Citations are within the normal text of sections and paragraphs.
| Tag | Explanation | Where Used | References | Citations |
|---|---|---|---|---|
| PoC |
Person of Concern Key individual involved in CRU emails as defined in this spreadsheet. |
References, Citations, IPCC Roles | 1 | 1 |
| JoC |
Journal of Concern A Journal which has published articles by one or more PoCs (Person of Concern) |
References, Citations | 4 | 5 |
| MoS |
Model or Simulation Reference appears to be a model or simulation, not observation or experiment |
References, Citations | 21 | 27 |
| NPR |
Non Peer Reviewed Reference has no Journal or no Volume or no Pages or it has Editors. |
References, Citations | 274 | 363 |
| SRC |
Self Reference Concern Author of a chapter containing references to own work. |
References, Citations, IPCC Roles | 43 | 85 |
| ARC |
Paper authored or co-authored by person who is also in list of Authors of another chapter. |
References, Citations | 19 | 22 |
| 2007 |
Paper dated 2007, when IPCC policy stated cutoff was December 2005 |
References, Citations | - | - |
| Ambiguous |
The short inline citation matched with more than one reference; however, AccessIPCC will link to the first reference found. |
Citations | - | 5 |
| NotFound |
The short inline citation was not matched with any reference. Believed to be caused by typing errors. |
Citations | - | 4 |
| Clean |
The reference was probably peer reviewed. |
References, Citations | 44 | 43 |
Coordinating Lead Authors:
Lenny Bernstein (USA), Joyashree Roy (India) [SRC:3],
| Concern | Occurrence |
|---|---|
| SRC 1-4 | 1 |
| Potentially Biased Authors | 1 |
| Impartial Authors | 1 |
Lead Authors:
K. Casey Delhotal (USA), Jochen Harnisch (Germany) [SRC:4], Ryuji Matsuhashi (Japan) [SRC:2], Lynn Price (USA) [SRC:10], Kanako Tanaka (Japan) [SRC:2], Ernst Worrell (The Netherlands) [SRC:25], Francis Yamba (Zambia) [SRC:3], Zhou Fengqi (China),
| Concern | Occurrence |
|---|---|
| SRC >= 5 | 2 |
| SRC 1-4 | 4 |
| Potentially Biased Authors | 6 |
| Impartial Authors | 2 |
Contributing Authors:
Stephane de la Rue du Can (France) [SRC:1], Dolf Gielen (The Netherlands), Suzanne Joosen (The Netherlands), Manaswita Konar (India), Anna Matysek (Australia) [SRC:1], Reid Miner (USA) [SRC:2], Teruo Okazaki (Japan) [SRC:1], Johan Sanders (The Netherlands), Claudia Sheinbaum Parado (Mexico),
| Concern | Occurrence |
|---|---|
| SRC 1-4 | 4 |
| Potentially Biased Authors | 4 |
| Impartial Authors | 5 |
Review Editors:
Olav Hohmeyer (Germany), Shigetaka Seki (Japan),
| Concern | Occurrence |
|---|---|
| Impartial Authors | 2 |
This chapter should be cited as:
Bernstein, L., J. Roy, K. C. Delhotal, J. Harnisch, R. Matsuhashi, L. Price, K. Tanaka, E. Worrell, F. Yamba, Z. Fengqi, 2007: Industry. In Climate Change 2007: Mitigation. Contribution of Working Group III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change [B. Metz, O.R. Davidson, P.R. Bosch, R. Dave, L.A. Meyer (eds)], Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA.
EXECUTIVE SUMMARY
Industrial sector emissions of greenhouse gases (GHGs) include carbon dioxide (CO2) from energy use, from non-energy uses of fossil fuels and from non-fossil fuel sources (e.g., cement manufacture); as well as non-CO2 gases.
- Energy-related CO2 emissions (including emissions from electricity use) from the industrial sector grew from 6.0 GtCO2 (1.6 GtC) in 1971 to 9.9 GtCO2 (2.7 GtC) in 2004. Direct CO2 emissions totalled 5.1 Gt (1.4 GtC), the balance being indirect emissions associated with the generation of electricity and other energy carriers. However, since energy use in other sectors grew faster, the industrial sector’s share of global primary energy use declined from 40% in 1971 to 37% in 2004. In 2004, developed nations accounted for 35%; transition economies 11%; and developing nations 53% of industrial sector energy-related CO2 emissions.
- CO2 emissions from non-energy uses of fossil fuels and from non-fossil fuel sources were estimated at 1.7 Gt (0.46 GtC) in 2000.
- Non-CO2 GHGs include: HFC-23 from HCFC-22 manufacture, PFCs from aluminium smelting and semiconductor processing, SF6 from use in electrical switchgear and magnesium processing and CH4 and N2O from the chemical and food industries. Total emissions from these sources (excluding the food industry, due to lack of data) decreased from 470 MtCO2-eq (130 MtC-eq) in 1990 to 430 MtCO2-eq (120 MtC-eq) in 2000.
Direct GHG emissions from the industrial sector are currently about 7.2 GtCO2-eq (2.0 GtC-eq), and total emissions, including indirect emissions, are about 12 GtCO2-eq (3.3 GtC-eq) (high agreement, much evidence).
Approximately 85% of the industrial sector’s energy use in 2004 was in the energy-intensive industries: iron and steel, non-ferrous metals, chemicals and fertilizers, petroleum refining, minerals (cement, lime, glass and ceramics) and pulp and paper. In 2003, developing countries accounted for 42% of iron and steel production, 57% of nitrogen fertilizer production, 78% of cement manufacture and about 50% of primary aluminium production. Many industrial facilities in developing nations are new and include the latest technology with the lowest specific energy use. However, many older, inefficient facilities remain in both industrialized and developing countries. In developing countries, there continues to be a huge demand for technology transfer to upgrade industrial facilities to improve energy efficiency and reduce emissions (high agreement, much evidence).
Many options exist for mitigating GHG emissions from the industrial sector (high agreement, much evidence). These options can be divided into three categories:
- Sector-wide options, for example more efficient electric motors and motor-driven systems; high efficiency boilers and process heaters; fuel switching, including the use of waste materials; and recycling.
- Process-specific options, for example the use of the bio-energy contained in food and pulp and paper industry wastes, turbines to recover the energy contained in pressurized blast furnace gas, and control strategies to minimize PFC emissions from aluminium manufacture.
- Operating procedures, for example control of steam and compressed air leaks, reduction of air leaks into furnaces, optimum use of insulation, and optimization of equipment size to ensure high capacity utilization.
Mitigation potential and cost in 2030 have been estimated through an industry-by-industry assessment for energy-intensive industries and an overall assessment for other industries. The approach yielded mitigation potentials at a cost of <100 US$/tCO2-eq (<370 US$/tC-eq) of 2.0 to 5.1 GtCO2-eq/yr (0.6 to 1.4 GtC-eq/yr) under the B2 scenario [1] . The largest mitigation potentials are located in the steel, cement, and pulp and paper industries and in the control of non-CO2 gases. Much of the potential is available at <50 US$/tCO2-eq (<180 US$/tC-eq). Application of carbon capture and storage (CCS) technology offers a large additional potential, albeit at higher cost (medium agreement, medium evidence).
Key uncertainties in the projection of mitigation potential and cost in 2030 are the rate of technology development and diffusion, the cost of future technology, future energy and carbon prices, the level of industry activity in 2030, and climate and non-climate policy drivers. Key gaps in knowledge are the base case energy intensity for specific industries, especially in economies-in-transition, and consumer preferences.
Full use of available mitigation options is not being made in either industrialized or developing nations. In many areas of the world, GHG mitigation is not demanded by either the market or government regulations. In these areas, companies will invest in GHG mitigation if other factors provide a return on their investment. This return can be economic, for example energy efficiency projects that provide an economic payout, or it can be in terms of achieving larger corporate goals, for example a commitment to sustainable development. The slow rate of capital stock turnover is also a barrier in many industries, as is the lack of the financial and technical resources needed to implement mitigation options, and limitations in the ability of industrial firms to access and absorb technological information about available options (high agreement, much evidence).
Industry GHG investment decisions, many of which have long-term consequences, will continue to be driven by consumer preferences, costs, competitiveness and government regulation. A policy environment that encourages the implementation of existing and new mitigation technologies could lead to lower GHG emissions. Policy portfolios that reduce the barriers to the adoption of cost-effective, low-GHG-emission technology can be effective (medium agreement, medium evidence).
Achieving sustainable development will require the implementation of cleaner production processes without compromising employment potential. Large companies have greater resources, and usually more incentives, to factor environmental and social considerations into their operations than small and medium enterprises (SMEs), but SMEs provide the bulk of employment and manufacturing capacity in many developing countries. Integrating SME development strategy into the broader national strategies for development is consistent with sustainable development objectives (high agreement, much evidence).
Industry is vulnerable to the impacts of climate change, particularly to the impacts of extreme weather. Companies can adapt to these potential impacts by designing facilities that are resistant to projected changes in weather and climate, relocating plants to less vulnerable locations, and diversifying raw material sources, especially agricultural or forestry inputs. Industry is also vulnerable to the impacts of changes in consumer preference and government regulation in response to the threat of climate change. Companies can respond to these by mitigating their own emissions and developing lower-emission products (high agreement, much evidence).
While existing technologies can significantly reduce industrial GHG emissions, new and lower-cost technologies will be needed to meet long-term mitigation objectives. Examples of new technologies include: development of an inert electrode to eliminate process emissions from aluminium manufacture; use of carbon capture and storage in the ammonia, cement and steel industries; and use of hydrogen to reduce iron and non-ferrous metal ores (medium agreement, medium evidence).
Both the public and the private sectors have important roles in the development of low-GHG-emission technologies that will be needed to meet long-term mitigation objectives. Governments are often more willing than companies to fund the higher risk, earlier stages of the R&D process, while companies should assume the risks associated with actual commercialisation. The Kyoto Protocol’s Clean Development Mechanism (CDM) and Joint Implementation (JI), and a variety of bilateral and multilateral programmes, have the deployment, transfer and diffusion of mitigation technology as one of their goals (high agreement, much evidence).
Voluntary agreements between industry and government to reduce energy use and GHG emissions have been used since the early 1990 s. Well-designed agreements, which set realistic targets, include sufficient government support, often as part of a larger environmental policy package, and include a real threat of increased government regulation or energy/GHG taxes if targets are not achieved, can provide more than business-as-usual energy savings or emission reductions. Some voluntary actions by industry, which involve commitments by individual companies or groups of companies, have achieved substantial emission reductions. Both voluntary agreements and actions also serve to change attitudes, increase awareness, lower barriers to innovation and technology adoption, and facilitate co-operation with stakeholders (medium agreement, much evidence).
7.1 Introduction
This chapter addresses past, ongoing, and short (to 2010 ) and medium-term (to 2030 ) future actions that can be taken to mitigate GHG emissions from the manufacturing and process industries. [2]
Globally, and in most countries, CO2 accounts for more than 90% of CO2-eq GHG emissions from the industrial sector ( Price et al., 2006 [NPR, SRC] ; US EPA, 2006b [NPR] ). These CO2 emissions arise from three sources: (1) the use of fossil fuels for energy, either directly by industry for heat and power generation or indirectly in the generation of purchased electricity and steam; (2) non-energy uses of fossil fuels in chemical processing and metal smelting; and (3) non-fossil fuel sources, for example cement and lime manufacture. Industrial processes also emit other GHGs, e.g.:
- Nitrous oxide (N2O) is emitted as a byproduct of adipic acid, nitric acid and caprolactam production;
- HFC-23 is emitted as a byproduct of HCFC-22 production, a refrigerant, and also used in fluoroplastics manufacture;
- Perfluorocarbons (PFCs) are emitted as byproducts of aluminium smelting and in semiconductor manufacture;
- Sulphur hexafluoride (SF6) is emitted in the manufacture, use and, decommissioning of gas insulated electrical switchgear, during the production of flat screen panels and semiconductors, from magnesium die casting and other industrial applications;
- Methane (CH4) is emitted as a byproduct of some chemical processes; and
- CH4 and N2O can be emitted by food industry waste streams.
Many GHG emission mitigation options have been developed for the industrial sector. They fall into three categories: operating procedures, sector-wide technologies and process-specific technologies. A sampling of these options is discussed in Sections 7.2 – 7.4 . The short- and medium-term potential for and cost of all classes of options are discussed in Section 7.5 , barriers to the application of these options are addressed in Section 7.6 and the implication of industrial mitigation for sustainable development is discussed in Section 7.7 .
Section 7.8 discusses the sector’s vulnerability to climate change and options for adaptation. A number of policies have been designed either to encourage voluntary GHG emission reductions from the industrial sector or to mandate such reductions. Section 7.9 describes these policies and the experience gained to date. Co-benefits of reducing GHG emissions from the industrial sector are discussed in Section 7.10 . Development of new technology is key to the cost-effective control of industrial GHG emissions. Section 7.11 discusses research, development, deployment and diffusion in the industrial sector and Section 7.12 , the long-term (post- 2030 ) technologies for GHG emissions reduction from the industrial sector. Section 7.13 summarizes gaps in knowledge.
7.1.1 Status of the sector
This chapter focuses on the mitigation of GHGs from energy-intensive industries: iron and steel, non-ferrous metals, chemicals (including fertilisers), petroleum refining, minerals (cement, lime, glass and ceramics) and pulp and paper, which account for most of the sector’s energy consumption in most countries (Dasgupta and Roy, 2000 [NotFound] ; IEA, 2003a [NPR] ,b; ( Sinton and Fridley, 2000 ) ). The food processing industry is also important because it represents a large share of industrial energy consumption in many non-industrialized countries. Each of these industries is discussed in detail in Section 7.4 .
Globally, large enterprises dominate these industries. However, small- and medium-sized enterprises (SMEs) are important in developing nations. For example, in India, SMEs have significant shares in the metals, chemicals, food and pulp and paper industries ( GOI, 2005 [NPR] ). There are 39.8 million SMEs in China, accounting for 99% of the country’s enterprises, 50% of asset value, 60% of turnover, 60% of exports and 75% of employment ( APEC, 2002 [NPR] ). While regulations are moving large industrial enterprises towards the use of environmentally sound technology, SMEs may not have the economic or technical capacity to install the necessary control equipment ( Chaudhuri and Gupta, 2003 [NPR] ; Gupta, 2002 [NPR] ) or are slower to innovate ( Swamidass, 2003 [MoS] ). These SME limitations create special challenges for efforts to mitigate GHG emissions. However, innovative R&D for SMEs is also taking place for this sector (See Section 7.7 ).
7.1.2 Development trends
The production of energy-intensive industrial goods has grown dramatically and is expected to continue growing as population and per capita income increase. Since 1970, global annual production of cement increased 271%; aluminium, 223%; steel, 84% ( USGS, 2005 [NPR] ), ammonia, 200% ( IFA, 2005 [NPR] ) and paper, 180% ( FAO, 2006 [NPR] ).
Much of the world’s energy-intensive industry is now located in developing nations. China is the world’s largest producer of steel ( IISI, 2005 [NPR] ), aluminium and cement ( USGS, 2005 [NPR] ). In 2003, developing countries accounted for 42% of global steel production ( IISI, 2005 [NPR] ), 57% of global nitrogen fertilizer production ( IFA, 2004 [NPR] ), 78% of global cement manufacture and about 50% of global primary aluminium production ( USGS, 2005 [NPR] ). Since many facilities in developing nations are new, they sometimes incorporate the latest technology and have the lowest specific emission rates ( BEE, 2006 [NPR] ; IEA, 2006c [NPR] ). This has been demonstrated in the aluminium ( (Navarro et al., 2003 ) ), cement ( BEE, 2003 [NPR] ), fertilizer ( Swaminathan and Sukalac, 2004 [NPR] ) and steel industries (Tata Steel, Ltd., 2005 [NotFound] ). However, due to the continuing need to upgrade existing facilities, there is a huge demand for technology transfer (hardware, software and know-how) to developing nations to achieve energy efficiency and emissions reduction in their industrial sectors (high agreement, much evidence).
New rules introduced both domestically and through the multilateral trade system, foreign buyers, insurance companies, and banks require SMEs to comply with higher technical (e.g., technical barriers to trade), environmental ( ISO, 1996 [NPR] ), and labour standards (ENDS-Directory, 2006 ). These efforts can be in conflict with pressures for economic growth and increased employment, for example in China, where the government’s efforts to ban the use of small-scale coke-producing facilities for energy efficiency and environmental reasons have been unsuccessful due to the high demand for this product ( IEA, 2006a [NPR] ).
Competition within the developing world for export markets, foreign investment, and resources is intensifying. Multinational enterprises seeking out new markets and investments offer both large enterprises ( Rock, 2005 [NPR] ) and capable SMEs the opportunity to insert themselves into global value chains through subcontracting linkages, while at the same time increasing competitive pressure on other enterprises, which could lose their existing markets. Against this backdrop, SMEs, SME associations, support institutions, and governments in transition and developing countries face the challenge of adopting new approaches and fostering SME competitiveness. Integration of SME development strategy in the broader national strategies for technology development, sustainable development and/or poverty reduction and growth is under consideration in transition and developing countries ( GOI, 2004 [NPR] ).
7.1.3 Emission trends
Total industrial sector GHG emissions are currently estimated to be about 12 GtCO2-eq/yr (3.3 GtC-eq/yr) (high agreement, much evidence). Global and sectoral data on final energy use, primary energy use [3] , and energy-related CO2 emissions including indirect emissions related to electricity use, for 1971 to 2004 ( Price et al., 2006 [NPR, SRC] ), are shown in Table 7.1 . In 1971, the industrial sector used 91 EJ of primary energy, 40% of the global total of 227 EJ. By 2004, industrys share of global primary energy use declined to 37%.
Table 7.1: Industrial sector final energy, primary energy and energy-related carbon dioxide emissions, nine world regions, 1971 2004
| Final energy (EJ) | Primary energy (EJ) | Energy-related carbon dioxide, including indirect emissions from electricity use (MtCO2) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 1971 | 1990 | 2004 | 1971 | 1990 | 2004 | 1971 | 1990 | 2004 | |
| Pacific OECD | 6.02 | 8.04 | 10.31 | 8.29 | 11.47 | 14.63 | 524 | 710 | 853 |
| North America | 20.21 | 19.15 | 22.66 | 25.88 | 26.04 | 28.87 | 1,512 | 1,472 | 1512 |
| Western Europe | 14.78 | 14.88 | 16.60 | 19.57 | 20.06 | 21.52 | 1,380 | 1,187 | 1126 |
| Central and Eastern Europe | 3.75 | 4.52 | 2.81 | 5.46 | 7.04 | 3.89 | 424 | 529 | 263 |
| EECCA | 11.23 | 18.59 | 9.87 | 15.67 | 24.63 | 13.89 | 1,095 | 1,631 | 856 |
| Developing Asia | 7.34 | 19.88 | 34.51 | 9.38 | 26.61 | 54.22 | 714 | 2,012 | 4098 |
| Latin America | 2.79 | 5.94 | 8.22 | 3.58 | 7.53 | 10.87 | 178 | 327 | 469 |
| Sub-Saharan Africa | 1.24 | 2.11 | 2.49 | 1.70 | 2.98 | 3.60 | 98 | 178 | 209 |
| Middle East/North Africa | 0.83 | 4.01 | 6.78 | 1.08 | 4.89 | 8.63 | 65 | 277 | 470 |
| World | 68.18 | 97.13 | 114.25 | 90.61 | 131.25 | 160.13 | 5,990 | 8,324 | 9855 |
The developing nations share of industrial CO2 emissions from energy use grew from 18% in 1971 to 53% in 2004 . In 2004, energy use by the industrial sector resulted in emissions of 9.9 GtCO2 (2.7 GtC), 37% of global CO2 emissions from energy use. Direct CO2 emissions totalled 5.1 Gt (1.4 GtC), the balance being indirect emissions associated with the generation of electricity and other energy carriers. In 2000, CO2 emissions from non-energy uses of fossil fuels (e.g., production of petro-chemicals) and from non-fossil fuel sources (e.g., cement manufacture) were estimated to be 1.7 GtCO2 (0.46 GtC) ( Olivier and Peters, 2005 [ARC] ). As shown in Table 7.3 , industrial emissions of non-CO2 gases totalled about 0.4 GtCO2-eq (0.1 GtC-eq) in 2000 and are projected to be at about the same level in 2010 . Direct GHG emissions from the industrial sector are currently about 7.2 GtCO2-eq (2.0 GtC-eq), and total emissions, including indirect emissions, are about 12 GtCO2-eq (3.3 GtC-eq).
Table 7.2: Projected industrial sector final energy, primary energy and energy-related CO2 emissions, based on SRES Scenarios, 2010 2030 . A1B Scenario
| Final energy (EJ) | Primary energy (EJ) | Energy-related carbon dioxide, including indirect emissions from electricity use (MtCO2) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 2010 | 2020 | 2030 | 2010 | 2020 | 2030 | 2010 | 2020 | 2030 | |
| Pacific OECD | 10.04 | 10.68 | 11.63 | 14.19 | 14.25 | 14.52 | 1,170 | 1,169 | 1,137 |
| North America | 24.95 | 26.81 | 28.34 | 32.32 | 32.84 | 32.94 | 1,875 | 1,782 | 1,650 |
| Western Europe | 16.84 | 18.68 | 20.10 | 24.76 | 25.45 | 25.47 | 1,273 | 1,226 | 1,158 |
| Central and Eastern Europe | 6.86 | 7.74 | 8.57 | 9.28 | 10.28 | 10.99 | 589 | 608 | 594 |
| EECCA | 20.82 | 24.12 | 27.74 | 28.83 | 32.20 | 35.43 | 1,764 | 1,848 | 1,853 |
| Developing Asia | 39.49 | 54.00 | 72.50 | 62.09 | 84.64 | 109.33 | 4,827 | 6,231 | 7,340 |
| Latin America | 18.20 | 26.58 | 33.13 | 29.14 | 38.72 | 51.09 | 1,492 | 2,045 | 2,417 |
| Sub-Saharan Africa | 7.01 | 10.45 | 13.70 | 13.27 | 19.04 | 27.40 | 833 | 1,286 | 1,534 |
| Middle East/North Africa | 14.54 | 22.21 | 29.17 | 20.34 | 29.20 | 39.32 | 1,342 | 1,888 | 2,224 |
| World | 158.75 | 201.27 | 244.89 | 234.32 | 286.63 | 346.48 | 15,165 | 18,081 | 19,908 |
| Final energy (EJ) | Primary energy (EJ) | Energy-related carbon dioxide including indirect emissions from electricity use (MtCO2) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 2010 | 2020 | 2030 | 2010 | 2020 | 2030 | 2010 | 2020 | 2030 | |
| Pacific OECD | 10.83 | 11.64 | 11.38 | 14.27 | 14.17 | 12.83 | 980 | 836 | 688 |
| North America | 20.23 | 20.82 | 21.81 | 28.64 | 29.28 | 29.18 | 1,916 | 1,899 | 1,725 |
| Western Europe | 14.98 | 14.66 | 14.35 | 19.72 | 18.56 | 17.69 | 1,270 | 1,154 | 1,063 |
| Central and Eastern Europe | 3.42 | 4.30 | 5.03 | 4.44 | 5.28 | 6.06 | 327 | 380 | 424 |
| EECCA | 12.65 | 14.74 | 16.96 | 16.06 | 19.06 | 22.33 | 1,093 | 1,146 | 1,208 |
| Developing Asia | 40.68 | 53.62 | 67.63 | 55.29 | 72.42 | 90.54 | 4,115 | 4,960 | 5,785 |
| Latin America | 11.46 | 15.08 | 18.24 | 15.78 | 20.10 | 24.84 | 950 | 1,146 | 1,254 |
| Sub-Saharan Africa | 2.75 | 4.96 | 10.02 | 4.33 | 7.53 | 14.51 | 260 | 345 | 665 |
| Middle East/North Africa | 8.12 | 9.67 | 12.48 | 13.90 | 15.51 | 19.22 | 791 | 888 | 1,080 |
| World | 125.13 | 149.49 | 177.90 | 172.44 | 201.92 | 237.19 | 11,703 | 12,755 | 13,892 |
| Note: Biomass energy included, EECCA = countries of Eastern Europe, the Caucasus and Central Asia. Source: Price et al. (2006). | |||||||||
Table 7.2 shows the results for the industrial sector of the disaggregation of two of the emission scenarios (see footnote 1), A1B and B2, produced for the IPCC Special Report on Emissions Scenarios SRES ( IPCC, 2000b [NPR] ) into four subsectors and nine world regions ( Price et al., 2006 [NPR, SRC] ). These projections show energy-related industrial CO2 emissions of 14 and 20 GtCO2 in 2030 for the B2 and A1B scenarios, respectively. In both scenarios, CO2 emissions from industrial energy use are expected to grow significantly in the developing countries, while remaining essentially constant in the A1 scenario and declining in the B2 scenario for the industrialized countries and countries with economies-in-transition.
Table 7.3 shows projections of non-CO2 GHG emissions from the industrial sector to 2030 extrapolated from data to 2020 ( US EPA 2006a [NPR] ,b). US EPA provides the only comprehensive data set with baselines and mitigation costs over this time frame for all gases and all sectors. However, baselines differ substantially for sectors covered by other studies, for example IPCC/TEAP ( 2005 ). As a result of mitigation actions, non-CO2 GHG emissions decreased from 1990 to 2000, and there are many programmes underway to further reduce these emissions (See Sections 7.4.2 and 7.4.8. ). Therefore Table 7.3 shows the US EPAs technology adoption scenario, which assumes continued compliance with voluntary industrial targets. Table 7.4 shows these emissions by industrial process. [4]
Table 7.3: Projected industrial sector emissions of non-CO2 GHGs, MtCO2-eq/yr
| Region | 1990 | 2000 | 2010 | 2030 |
|---|---|---|---|---|
| Pacific OECD | 38 | 53 | 47 | 49 |
| North America | 147 | 117 | 96 | 147 |
| Western Europe | 159 | 96 | 92 | 109 |
| Central and Eastern Europe | 31 | 21 | 22 | 27 |
| EECCA | 37 | 20 | 21 | 26 |
| Developing Asia | 34 | 91 | 118 | 230 |
| Latin America | 17 | 18 | 21 | 38 |
| Sub-Saharan Africa | 6 | 10 | 11 | 21 |
| Middle East/North Africa | 2 | 3 | 10 | 20 |
| World | 470 | 428 | 438 | 668 |
Table 7.4: Projected baseline industrial sector emissions of non-CO2 GHGs
| Industrial sector | Emissions (MtCO2-eq/yr) | |||
|---|---|---|---|---|
| 1990 | 2000 | 2010 | 2030 | |
| N2O emissions from adipic/nitric acid production | 223 | 154 | 164 | 190 |
| HFC/PFC emissions from substitutes for ozone-depleting substancesa | 0 | 52 | 93 | 198 |
| HFC-23 emissions from HCFC-22 production | 77 | 96 | 45 | 106 |
| SF6 emission from use of electrical equipment (excluding manufacture) | 42 | 27 | 46 | 74 |
| PFC emission from aluminium production | 98 | 58 | 39 | 51 |
| PFC and SF6 emissions from semiconductor manufacture | 9 | 23 | 35 | 20 |
| SF6 emissions from magnesium production | 12 | 9 | 4 | 9 |
| N2O emission from caprolactam manufacture | 8 | 10 | 13 | 20 |
| Total | 470 | 428 | 438 | 668 |
| a Emissions from refrigeration equipment used in industrial processes included; emissions from all other refrigeration and air conditioning applications excluded. Source: US EPA, 2006a,b. | ||||
7.2 Industrial mitigation matrix
A wide range of technologies have the potential for reducing industrial GHG emissions (high agreement, much evidence). They can be grouped into categories, for example energy efficiency, fuel switching and power recovery. Within each category, some technologies, such as the use of more efficient electric motors and motor systems, are broadly applicable across all industries; while others, such as top-gas pressure recovery in blast furnaces, are process-specific. Table 7.5 presents selected examples of both classes of technologies for a number of industries. The table is not comprehensive and does not cover all industries or GHG mitigation technologies.
Table 7.5: Selected examples of industrial technology for reducing greenhouse-gas emissions (not comprehensive). Technologies in italics are under demonstration or development
| Sector | Energy efficiency | Fuel switching | Power recovery | Renewables | Feedstock change | Product change | Material efficiency | Non-CO2 GHG | CO2 sequestration |
|---|---|---|---|---|---|---|---|---|---|
| Sector wide | Benchmarking; Energy management systems; Efficient motor systems, boilers, furnaces, lighting and HVAC; Process integration | Coal to natural gas and oil | Cogeneration | Biomass, Biogas, PV, Wind turbines, Hydropower | Recycled inputs | Oxy-fuel combustion, CO2 separation from flue gas | |||
| Iron & Steel | Smelt reduction, Near net shape casting, Scrap preheating, Dry coke quenching | Natural gas, oil or plastic injection into the BF | Top-gas pressure recovery, Byproduct gas combined cycle | Charcoal | Scrap | High strength steel | Recycling, High strength steel, Reduction process losses | n.a. | Hydrogen reduction, Oxygen use in blast furnaces |
| Non-Ferrous Metals | Inert anodes, Efficient cell designs | Scrap | Recycling, thinner film and coating | PFC/SF6 controls | |||||
| Chemicals | Membrane separations, Reactive distillation | Natural gas | Pre-coupled gas turbine, Pressure recovery turbine, H2 recovery | Recycled plastics, biofeedstock | Linear low density polyethylene, high-performance Plastics | Recycling, Thinner film and coating, Reduced process losses | N2O, PFCs, CFCs and HFCs control | Application to ammonia, ethylene oxide processes | |
| Petroleum Refining | Membrane separation Refinery gas | Natural gas | Pressure recovery turbine, hydrogen recovery | Biofuels | Bio-feedstock | Increased efficiency transport sector | Control technology for N2O/CH4 | From hydrogen production | |
| Cement | Precalciner kiln, Roller mill, fluidized bed kiln | Waste fuels, Biogas, Biomass | Drying with gas turbine, power recovery | Biomass fuels, Biogas | Slags, pozzolanes | Blended cement Geo-polymers | n.a. | O2 combustion in kiln | |
| Glass | Cullet preheating Oxyfuel furnace | Natural gas | Air bottoming cycle | n.a. | Increased cullet use | High-strength thin containers | Re-usable containers | n.a. | O2 combustion |
| Pulp and Paper | Efficient pulping, Efficient drying, Shoe press, Condebelt drying | Biomass, Landfill gas | Black liquor gasification combined cycle | Biomass fuels (bark, black liquor) | Recycling, Non-wood fibres | Fibre orientation, Thinner paper | Reduction cutting and process losses | n.a. | O2 combustion in lime kiln |
| Food | Efficient drying, Membranes | Biogas, Natural gas | Anaerobic digestion, Gasification | Biomass, Biogas, Solar drying | Reduction process losses, Closed water use |
7.3 Industrial sector-wide operating procedures and technologies
This section discusses sector-wide mitigation options. Barriers to the implementation of these options are discussed in Section 7.6 .
7.3.1 Management practices, including benchmarking
Management tools are available to reduce GHG emissions, often without capital investment or increased operating costs. Staff training in both skills and the company’s general approach to energy efficiency for use in their day-to-day practices has been shown to be beneficial ( Caffal, 1995 [NPR] ). Programmes, for example reward systems that provide regular feedback on staff behaviour, have had good results.
Even when energy is a significant cost for an industry, opportunities for improvement may be missed because of organizational barriers. Energy audit and management programmes create a foundation for improvement and provide guidance for managing energy throughout an organization. Several countries have instituted voluntary corporate energy management standards, for example Canada (Natural Resources Canada, n.d.), Denmark ( Gudbjerg, 2005 [NPR] ) and the USA ( ANSI, 2005 [NPR] ). Others, for example India, through the Bureau of Energy Efficiency ( GOI 2004 [NPR] , 2005 ), promote energy audits. Integration of energy management systems into broader industrial management systems, allowing energy use to be managed for continuous improvement in the same manner as labour, waste and other inputs are managed, is highly beneficial ( McKane et al., 2005 [NPR] ). Documentation of existing practices and planned improvements is essential to achieving a transition from energy efficiency programmes and projects dependent on individuals to processes and practices that are part of the corporate culture. Software tools are available to help identify energy saving opportunities (US DOE, n.d.-a; US EPA, n.d.).
Energy Audits and Management Systems. Companies of all sizes use energy audits to identify opportunities for reducing energy use, which in turn reduces GHG emissions. For example, in 2000, Exxon Mobil implemented its Global Energy Management System with the goal of achieving a 15% reduction in energy use in its refineries and chemical plants ( Eidt, 2004 [NPR] Okazaki et al. 2004 [NPR, SRC] ) estimate that approximately 10% of total energy consumption in steel making could be saved through improved energy and materials management. ( Mozorov and Nikiforov 2002 ) ) reported an even larger 21.6% efficiency improvement in a Russian iron and steel facility. For SMEs in Germany, ( Schleich 2004 ) ) reported that energy audits help overcome several barriers to energy efficiency, including missing information about energy consumption patterns and energy saving measures. Schleich also found that energy audits conducted by engineering firms were more effective than those conducted by utilities or trade associations.
GHG Inventory and Reporting Systems. Understanding the sources and magnitudes of its GHG emissions gives industry the capability to develop business strategies to adapt to changing government and consumer requirements. Protocols for inventory development and reporting have been developed; the Greenhouse Gas Protocol developed by the World Resources Institute and World Business Council for Sustainable Development (WRI/WBCSD, 2004 ) is the most broadly used. The Protocol defines an accounting and reporting standard that companies can use to ensure that their measurements are accurate and complete. Several industries (e.g., aluminium, cement, chemical and pulp and paper) have developed specific calculation tools to implement the Protocol. Other calculation tools have been developed to estimate GHG emissions from office-based business operations and to quantify the uncertainty in GHG measurement and estimation (WRI/WBCSD, 2005 ). Within the European Union, GHG reporting guidelines have been developed for companies participating in the EU Emission Trading System.
GHG Management Systems. Environmental quality management systems such as ISO, Environmental quality management systems such as ISO,1 ( ISO, 1996 [NPR] ), are being used by many companies to build capacity for GHG emission reduction. For example, the US petroleum industry developed their own standard based on systems developed by various companies ( API, 2005 [NPR] ). The GHG emissions reduction opportunities identified by these management systems are evaluated using normal business criteria, and those meeting the current business or regulatory requirements are adopted. Those not adopted represent additional capacity that could be used if business, government, or consumer requirements change.
Benchmarking. Companies can use benchmarking to compare their operations with those of others, to industry average, or to best practice, to determine whether they have opportunities to improve energy efficiency or reduce GHG emissions. Benchmarking is widely used in industry, but benchmarking programmes must be carefully designed to comply with laws ensuring fair competition, and companies must develop their own procedures for using the information generated through these programmes. The petroleum industry has the longest experience with energy efficiency benchmarking through the use of an industry-accepted index developed by a private company ( Barats, 2005 [NPR] ). Many benchmarking programmes are developed through trade associations or ad hoc consortia of companies, and their details are often proprietary. However, ten Canadian potash operations published the details of their benchmarking exercise ( CFI, 2003 [NPR] ), which showed that increased employee awareness and training was the most frequently identified opportunity for improved energy performance. The success of the aluminium industry’s programmes is discussed in Section 7.4.2 .
Several governments have supported the development of benchmarking programmes in various forms, for example Canada, Flanders (Belgium), the Netherlands, Norway and the USA. As part of its energy and climate policy the Dutch government has reached an agreement with its energy-intensive industry that is explicitly based on industry’s energy efficiency performance relative to that of comparable industries worldwide. Industry is required to achieve world best practice in terms of energy efficiency. In return, the government refrains from implementing additional climate policies. By 2002 this programme involved companies using 94% of the energy consumed by industry in the Netherlands. Phylipsen et al. 2002 [Ambiguous] ) critiqued the agreement, and conclude that it would avoid emissions of 4 to 9 MtCO2 (1.1 to 2.5 MtC) in 2012 compared to a business-as-usual scenario, but that these emission reductions were smaller than those that would be achieved by a continuation of the Long-Term Agreements with industry (which ended in 2000 ) that called for a 2%/yr improvement in energy efficiency. The Flemish covenant, agreed in 2002, uses a similar approach. As of 1 January 2005, 177 companies had joined the covenant, which projects cumulative emissions saving of 2.45 MtCO2 (0.67 MtC) in 2012 (Government of Flanders, 2005 ).
In the USA, EPA’s Energy STAR for Industry programme has developed a benchmarking system for selected industries, for example automotive assembly plants, cement and wet corn milling ( (Boyd, 2005 ) ). The system is used by programme participants to evaluate the performance of their individual plants against a distribution of the energy performance of US peers. Other benchmarking programmes compare individual facilities to world best practice ( Galitsky et al., 2004 [Ambiguous] ).
7.3.2 Energy efficiency
IEA 2006a [NPR] ) reports ‘The energy intensity of most industrial processes is at least 50% higher than the theoretical minimum determined by the laws of thermodynamics. Many processes have very low energy efficiency and average energy use is much higher than the best available technology would permit.’ This provides a significant opportunity for reducing energy use and its associated CO2 emissions.
The major factors affecting energy efficiency of industrial plants are: choice and optimization of technology, operating procedures and maintenance, and capacity utilization, that is the fraction of maximum capacity at which the process is operating. Many studies ( US DOE, 2004 [NPR] ; IGEN/BEE; n.d.) have shown that large amounts of energy can be saved and CO2 emissions avoided by strict adherence to carefully designed operating and maintenance procedures. Steam and compressed air leaks, poorly maintained insulation, air leaks into boilers and furnaces and similar problems all contribute to excess energy use. Quantification of the amount of CO2 emission that could be avoided is difficult, because, while it is well known that these problems exist, the information on their extent is case-specific. Low capacity utilization is associated with more frequent shut-downs and poorer thermal integration, both of which lower energy efficiency and raise CO2 emissions.
In view of the low energy efficiency of industries in many developing counties, in particular Africa ( UNIDO, 2001 [NPR] ), application of industry-wide technologies and measures can yield technical and economic benefits, while at the same time enhance environmental integrity. Application of housekeeping and general maintenance on older, less-efficient plants can yield energy savings of 10–20%. Low-cost/minor capital measures (combustion efficiency optimisation, recovery and use of exhaust gases, use of correctly sized, high efficiency electric motors and insulation, etc.) show energy savings of 20–30%. Higher capital expenditure measures (automatic combustion control, improved design features for optimisation of piping sizing, and air intake sizing, and use of variable speed drive motors, automatic load control systems and process residuals) can result in energy savings of 40–50% ( UNIDO, 2001 [NPR] ,( Bakaya-Kyahurwa, 2004 ) ).
Electric motor driven systems provide a large potential for improvement of industry-wide energy efficiency. de Keulenaer et al., 2004 [NPR] ) report that motor-driven systems account for approximately 65% of the electricity consumed by EU-25 industry. Xenergy ( 1998 ) gave similar figures for the USA, where motor-driven systems account for 63% of industrial electricity use. The efficiency of motor-driven systems can be increased by improving the efficiency of the electric motor through reducing losses in the motor windings, using better magnetic steel, improving the aerodynamics of the motor and improving manufacturing tolerances. However, the motor is only one part of the system, and maximizing efficiency requires properly sizing of all components, improving the efficiency of the end-use devices (pumps, fans, etc.), reducing electrical and mechanical transmission losses, and the use of proper operation and maintenance procedures. Implementing high-efficiency motor driven systems, or improving existing ones, in the EU-25 could save about 30% of the energy consumption, up to 202 TWh/yr, and avoid emissions of up to 100 MtCO2/yr (27.2 MtC/yr) ( De Keulenaer et al., 2004 [NPR] ). In the USA, use of more efficient electric motor systems could save over 100 TWh/yr by 2010, and avoid emissions of 90 MtCO2/yr (24.5 MtC/yr) (Xenergy, 1998 ). A study of the use of variable speed drives in selected African food processing plants, petroleum refineries, and municipal utility companies with a total motor capacity of 70,000 kW resulted in a potential saving of 100 ktCO2-eq/yr (27 ktC/yr), or between 30–40%, at an internal rate of return of 40% ( CEEEZ, 2003 [NPR] IEA 2006b [NPR] ) estimates the global potential to be >20–25%, but a number of barriers have limited the optimization of motor systems (See Section 7.6 ).
Typical estimates indicate that about 20% of compressed air is lost through leakage. US DOE has developed best practices to identify and eliminate sources of leakage (US DOE, n.d.-a). IEA 2006a [NPR] ) estimates that steam generation consumes about 15% of global final industrial energy use. The efficiency of current steam boilers can be as high as 85%, while research in the USA aims to develop boilers with an efficiency of 94%. However, in practice, average efficiencies are often much lower. Efficiency measures exist for both boilers and distribution systems. Besides general maintenance, these include improved insulation, combustion controls and leak repair in the boiler, improved steam traps and condensate recovery. Studies in the USA identified energy-efficiency opportunities with economically attractive potentials up to 18–20% ( Einstein et al., 2001 [SRC] ; US DOE, 2002 [NPR] ). Boiler systems can also be upgraded to cogeneration systems.
Efficient high-pressure boilers using process residuals like bagasse are now available ( Cornland et al., 2001 [NPR, SRC] ) and can be used to replace traditional boilers (15–25 bar) in the sugar industry. The high-pressure steam is used to generate electricity for own use with a surplus available for export to the grid (see also 7.3.4). For example, a boiler with a 60 MW steam turbine system in a 400 t/hour sugar factory could provide a potential surplus of 40 MW of zero-carbon electricity, saving 400 ktCO2/yr ( Yamba and Matsika, 2003 [NPR, SRC] ). Similar technology installed at an Indian sugar mill increased the crushing period from 150 to 180 days, and exported an average of 10 MW of zero carbon electricity to the grid ( Sobhanbabu, 2003 [NPR] ).
Furnaces and process heaters, many of which are tailored for specific applications, can be further optimized to reduce energy use and emissions. Efficiency improvements are found in most new furnaces ( Berntsson et al., 1997 [NPR] ). Research is underway to further optimize combustion processes by improving furnace and burner designs, preheating combustion air, optimizing combustion controls ( Martin et al., 2000 [NPR, SRC] ); and using oxygen enrichment or oxy-fuel burners (See Section 7.3.7 ). These techniques are already being applied in specific applications.
7.3.3 Fuel switching, including the use of waste materials
While some industrial processes require specific fuels (e.g., metallurgical coke for iron ore reduction) [5] , many industries use fuel for steam generation and/or process heat, with the choice of fuel being determined by cost, fuel availability and environmental regulations. The TAR ( IPCC, 2001a [NPR] ) limited its consideration of industrial fuel switching to switches within fossil fuels (replacing coal with oil or natural gas), and concluded, based on a comparison of average and lowest carbon intensities for eight industries, that such switches could reduce CO2 emissions by 10–20%. These values are still applicable. A variety of industries are using methane from landfills as a boiler fuel ( US EPA, 2005 [NPR] ).
Waste materials (tyres, plastics, used oils and solvents and sewerage sludge) are being used by a number of industries. Even though many of these materials are derived from fossil fuels, they can reduce CO2 emissions compared to an alternative in which they were landfilled or burned without energy recovery. The steel industry has developed technology to use wastes such as plastics ( Ziebek and Stanek, 2001 [MoS] ) as alternative fuel and feedstock’s. Pretreated plastic wastes have been recycled in coke ovens and blast furnaces ( (Okuwaki, 2004 ) ), reducing CO2 emissions by reducing both emissions from incineration and the demand for fossil fuels. In Japan, use of plastics wastes in steel has resulted in a net emissions reduction of 0.6 MtCO2-eq/yr ( Okazaki et al., 2004 [NPR, SRC] ). Incineration of wastes (e.g., tyres, municipal and hazardous waste) in cement kilns is one of the most efficient methods of disposing of these materials ( (Cordi and Lombardi, 2004; ) ( Houillon and Jolliet, 2005 ) ). Heidelberg Cement 2006 [NPR] ) reported using 78% waste materials (tyres, animal meal and grease, and sewerage sludge) as fuel for one of its cement kilns. The cement industry, particularly in Japan, is investing to allow the use of municipal waste as fuel ( (Morimoto et al., 2006 ) ). Cement companies in India are using non-fossil fuels, including agricultural wastes, sewage, domestic refuse and used tyres, as well as wide range of waste solvents and other organic liquids; coupled with improved burners and burning systems ( Jain, 2005 [NPR] ).
Humphreys and Mahasenan 2002 [NPR] ) estimated that global CO2 emissions could be reduced by 12% through increased use of waste fuels. However, IEA 2006a [NPR] ) notes that use of waste materials is limited by their availability, Also, use of these materials for fuel must address their variable composition, and comply with all applicable environmental regulations, including control of airborne toxic materials.
7.3.4 Heat and power recovery
Energy recovery provides major energy efficiency and mitigation opportunities in virtual all industries. Energy recovery techniques are old, but large potentials still exist ( (Bergmeier, 2003 ) ). Energy recovery can take different forms: heat, power and fuel recovery. Fuel recovery options are discussed in the specific industry sectors in Section 7.4 . While water (steam) is the most used energy recovery medium, the use of chemical heat sinks in heat pumps, organic Rankine cycles and chemical recuperative gas turbines, allow heat recovery at lower temperatures. Energy-efficient process designs are often based on increased internal energy recovery, making it hard to define the technology or determine the mitigation potential.
Heat is used and generated at specific temperatures and pressures and discarded afterwards. The discarded heat can be re-used in other processes onsite, or used to preheat incoming water and combustion air. New, more efficient heat exchangers or more robust (e.g., low-corrosion) heat exchangers are being developed continuously, improving the profitability of enhanced heat recovery. In industrial sites the use of low-temperature waste heat is often limited, except for preheating boiler feed water. Using heat pumps allows recovery of the low-temperature heat for the production of higher temperature steam.
While there is a significant potential for heat recovery in most industrial facilities, it is important to design heat recovery systems that are energy-efficient and cost-effective (i.e., process integration). Even in new designs, process integration can identify additional opportunities for energy efficiency improvement. Typically, cost-effective energy savings of 5 to 40% are found in process integration analyses in almost all industries ( Martin et al., 2000 [NPR, SRC] ; IEA-IETS, n.d.). The wide variation makes it hard to estimate the overall potential for energy-efficiency improvement and GHG mitigation. However, Martin et al. 2000 [NPR, SRC] ) estimated the potential fuel savings from process integration in US industry to be 10% above the gain for conventional heat recovery systems. Einstein et al. 2001 [SRC] ) and the US DOE 2002 [NPR] ) estimated an energy savings potential of 5 to 10% above conventional heat recovery techniques.
Power can be recovered from processes operating at elevated pressures using even small pressure differences to produce electricity through pressure recovery turbines. Examples of pressure recovery opportunities are blast furnaces, fluid catalytic crackers and natural gas grids (at sites where pressure is reduced before distribution and use). Power recovery may also include the use of pressure recovery turbines instead of pressure relief valves in steam networks and organic Rankine cycles from low-temperature waste streams. Bailey and Worrell 2005 [NPR, SRC] ) found a potential savings of 1 to 2% of all power produced in the USA, which would mitigate 21 MtCO2 (5.7 MtC).
Cogeneration (also called Combined Heat and Power, CHP) involves using energy losses in power production to generate heat for industrial processes and district heating, providing significantly higher system efficiencies. Cogeneration technology is discussed in Section 4.3.5 . Industrial cogeneration is an important part of power generation in Germany and the Netherlands, and is the majority of installed cogeneration capacity in many countries. Laurin et al. 2004 [NPR] ) estimated that currently installed cogeneration capacity in Canada provided a net emission reduction of almost 30 MtCO2/yr (8.18 MtC/yr). Cogeneration is also well established in the paper, sugar and chemical industries in India, but not in the cement industry due to lack of indigenously proven technology suitable for high dust loads. The Indian government is recommending adoption of technology already in use in China, Japan and Southeast Asian countries ( Raina, 2002 [NPR] ).
There is still a large potential for cogeneration. Mitigation potential for industrial cogeneration is estimated at almost 150 MtCO2 (40 MtC) for the USA ( (Lemar, 2001 ) ), and 334 MtCO2 (91.1 MtC) for Europe ( De Beer et al., 2001 [NPR] ). Studies also have been performed for specific countries, for example Brazil ( (Szklo et al., 2004 ) ), although the CO2 emissions mitigation impact is not always specified.
7.3.5 Renewable energy
The use of biomass is well established in some industries. The pulp and paper industry uses biomass for much of its energy needs (See Section 7.4.6 .). In many developing countries the sugar industry uses bagasse and the edible oils industry uses byproduct wastes to generate steam and/or electricity (See Section 7.4.7 .). The use of bagasse for energy is likely to grow as more becomes available as a byproduct of sugar-based ethanol production ( Kaltner et al., 2005 [NPR] ). When economically attractive, other industries use biomass fuels, for example charcoal in blast furnaces in Brazil ( Kim and Worrell, 2002a [SRC] ). These applications will reduce CO2 emissions, but will only achieve zero net CO2 emissions if the biomass is grown sustainably.
Industry also can use solar or wind generated electricity, if it is available. The potential for this technology is discussed in Section 4.3.3 . The food and jute industries make use of solar energy for drying in appropriate climates ( Das and Roy, 1994 [NPR, SRC] ). The African Rural Energy Enterprise Development initiative is promoting the use of solar food driers in Mali and Tanzania to preserve fresh produce for local use and for the commercial market ( AREED, 2000 [NPR] ). Concentrating solar power could be used to provide process heat for industrial purposes, though there are currently no commercial applications (IEA-SolarPACES, n.d.).
7.3.6 Materials efficiency and recycling
Materials efficiency refers to the reduction of energy use by the appropriate choice of materials and recycling. Many of these options are applicable to the transport and building sectors and are discussed in Chapter 5 , section 5.3.1 and Chapter 6 , section 6.4 . Recycling is the best-documented material efficiency option for the industrial sector. Recycling of steel in electric arc furnaces accounts about a third of world production and typically uses 60–70% less energy ( De Beer et al., 1998 [NPR] ). This technology, and options for further energy savings, are discussed in Section 7.4.1 . Recycling aluminium requires only 5% of the energy of primary aluminium production. Recycled aluminium from used products and sources outside the aluminium industry now constitutes 33% of world supply and is forecast to rise to 40% by 2025 ( IAI, 2006b [NPR, MoS] , Martcheck, 2006 [MoS] ). Recycling is also an important energy saving factor in other non-ferrous metal industries, as well as the glass and plastics industries (GOI, various issues). Recycling occurs both internally within plants and externally in the waste management sector (See Section 10.4.5 ).
Materials substitution, for example the addition of wastes (blast furnace slag, fly ash) and geo-polymers to clinker to reduce CO2 emissions from cement manufacture (See Section 7.4.5.1 ), is also applicable to the industrial sector. Some materials substitution options, for example the production of lightweight materials for vehicles, can increase GHG emissions from the industrial sector, which will be more than offset by the reduction of emissions from other sectors (See Section 7.4.9 ). Use of bio-materials is a special case of materials substitution. No projections of the GHG mitigation potential of this option were found in the literature.
7.3.7 Carbon dioxide Capture and Storage (CCS), including oxy-fuel combustion
CCS involves generating a stream with a high concentration of CO2, then either storing it geologically, in the ocean, or in mineral carbonates, or using it for industrial purposes. The IPCC Special Report on CCS ( IPCC, 2005b [NPR] ) provides a full description of this technology, including its potential application in industry. It also discusses industrial uses of CO2, including its temporary retention in beverages, which are small compared to total industrial emissions of CO2.
Large quantities of hydrogen are produced as feedstock for petroleum refining, and the production of ammonia and other chemicals. Hydrogen manufacture produces a CO2-rich by-product stream, which is a potential candidate for CCS technology. IPCC 2005b [NPR] ) estimated the representative cost of CO2 storage from hydrogen manufacture at 15 US$/tCO2 (55 US$/tC). Transport (250 km pipeline) injection and monitoring would add another 2 to 16 US$/tCO2 (7 to 60 US$/tC) to costs.
CO2 emissions from steel making are also a candidate for CCS technology. IEA 2006a [NPR] ) estimates that CCS could reduce CO2 emissions from blast furnaces and DRI (direct reduction iron) plants by about 0.1 GtCO2 (0.03 GtC) in 2030 at a cost of 20 to 30 US$/tCO2 (73 to 110 US$/tC). Smelt reduction also allow the integration of CCS into the production of iron. CCS has also been investigated for the cement industry. ( Anderson and Newell 2004 ) ) estimate that it is possible to reduce CO2 emissions by 65 to 70%, at costs of 50 to 250 US$/tCO2 (183–917 US$/tC). IEA 2006a [NPR] ) estimates the potential for this application at up to 0.25 GtCO2 (0.07 GtC) in 2030 .
Oxy-fuel combustion can be used to produce a CO2-rich flue gas, suitable for CCS, from any combustion process. In the past, oxy-fuel combustion has been considered impractical because of its high flame temperature. However, Gross et al. 2003 [NPR] ), report on the development of technology that allows oxy-fuel combustion to be used in industrial furnaces with conventional materials. Tests in an aluminium remelting furnace showed up to 73% reduction in natural gas use compared to a conventional air-natural gas furnace. When the energy required to produce oxygen is taken into account, overall energy saving is reduced to 50 to 60% (Jupiter Oxygen Corp., 2006 ). Lower but still impressive energy efficiency improvements have been obtained in other applications, up to 50% in steel remelting furnaces, up to 45% in small glass-making furnaces, and up to 15% in large glass-making furnaces ( NRC, 2001 [NPR] ). The technology has also been demonstrated using coal and waste oils as fuel. Since much less nitrogen is present in the combustion chamber, NOx emissions are very low, even without external control, and the system is compatible with integrated pollution removal technology for the control of mercury, sulphur and particulate emissions as well as CO2 ( Ochs et al., 2005 [NPR] ).
Industry does not currently use CCS as a mitigation option, because of its high cost. However, assuming that the R&D currently underway on lowering CCS cost is successful, application of this technology to industrial CO2 sources should begin before 2030 and be wide-spread after that date.
7.4 Process-specific technologies and measures
This section discusses process specific mitigation options. Barriers to the implementation of these options are discussed in Section 7.6 . The section focuses on energy intensive industries: iron and steel, non-ferrous metals, chemicals, petroleum refining, minerals (cement, lime and glass) and pulp and paper. IEA 2006a [NPR] ) reported that these industries (ex-petroleum refining) accounted for 72% of industrial final energy use in 2003 . With petroleum refining, the total is about 85%. A subsection covers the food industry, which is not a major contributor to global industrial GHG emissions, but is a large contributor to these emissions in many developing countries. Subsections also cover other industries and inter-industry options, where the use of one industry’s waste as a feedstock or energy source by another industry can reduce overall emissions (See Section 7.4.9 ). All the industries discussed in this section can benefit from application of the sector-wide technologies (process optimization, energy efficiency, etc.) discussed in Section 7.3 . The application of these technologies will not be discussed again.
7.4.1 Iron and steel
Steel is by far the worlds most important metal, with a global production of 1129 Mt in 2005 . In 2004, the most important steel producers were China (26%), EU-25 (19%), Japan (11%), USA (10%) and Russia (6%) ( IISI, 2005 [NPR] ). Three routes are used to make steel. In the primary route (about 60%), used in almost 50 countries, iron ore is reduced to iron in blast furnaces using mostly coke or coal, then processed into steel. In the second route (about 35%), scrap steel is melted in electric-arc furnaces to produce crude steel that is further processed. This process uses only 30 to 40% of the energy of the primary route, with CO2 emissions reduction being a function of the source of electricity ( De Beer et al., 1998 [NPR] ). The remaining steel production (about 5%), uses natural gas to produce direct reduced iron (DRI). DRI cannot be used in primary steel plants, and is mainly used as an alternative iron input in electric arc furnaces, which can result in a reduction of up to 50% in CO2 emissions compared with primary steel making ( IEA, 2006a [NPR] ). Use of DRI is expected to increase in the future ( Hidalgo et al., 2005 [MoS] ).
Global steel industry CO2 emissions are estimated to be 1500 to 1600 MtCO2 (410 to 440 MtC), including emissions from coke manufacture and indirect emissions due to power consumption, or about 6 to 7% of global anthropogenic emissions ( Kim and Worrell, 2002a [SRC] ). The total is higher for some countries, for example steel production accounts for over 10% of Chinas energy use and about 10% of its anthropogenic CO2 emissions ( Price et al., 2002 [SRC] ). Emissions per tonne of steel vary widely between countries: 1.25 tCO2 (0.35 tC) in Brazil, 1.6 tCO2 (0.44 tC) in Korea and Mexico, 2.0 tCO2 (0.54 tC) in the USA, and 3.1 to 3.8 tCO2 (0.84 to 1.04 tC) in China and India ( Kim and Worrell, 2002a [SRC] ). The differences are based on the production routes used, product mix, production energy efficiency, fuel mix, carbon intensity of the fuel mix, and electricity carbon intensity.
Energy Efficiency. Iron and steel production is a combination of batch processes. Steel industry efforts to improve energy efficiency include enhancing continuous production processes to reduce heat loss, increasing recovery of waste energy and process gases, and efficient design of electric arc furnaces, for example scrap preheating, high-capacity furnaces, foamy slagging and fuel and oxygen injection. Continuous casting, introduced in the 1970 s and 1980 s, saves both energy and material, and now accounts for 88% of global steel production ( IISI, 2005 [NPR] ). Figure 7.1 shows the technical potential [6] for CO2 emission reductions by region in 2030 for full diffusion of eight cost-effective and/or well developed energy savings technologies under the SRES B2 scenario, using a methodology developed by Tanaka et al. 2005 [MoS, SRC] , 2006 ).
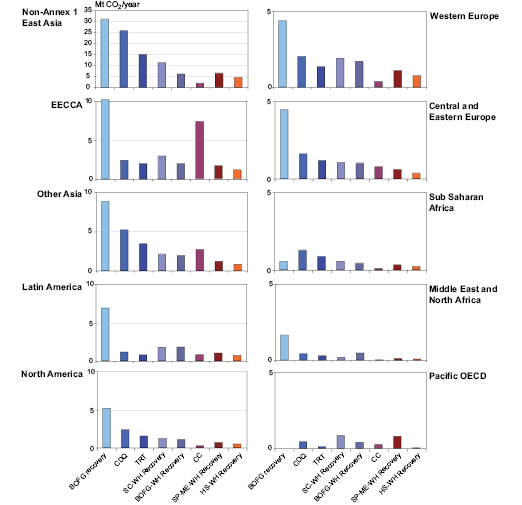
Figure 7.1: CO2 reduction potential of eight energy saving technologies in 2030
CDQ = Coke Dry Quenching, HS = Hot Stove, TRT = Top Pressure Recovery Turbine, SC = Sinter Cooling, CC = Continuous Casting, SP = Sinter Plant, BOFG = Basic Oxygen Furnace Gas, ME = Main Exhaust, WH = Waste Heat
Note: B2 Scenario, CO2 emission reduction based on energy saving assuming 100% diffusion in 2030 less current diffusion rates.
Source: Tanaka, 2006 [NPR, SRC] .
The potential for energy efficiency improvement varies based on the production route used, product mix, energy and carbon intensities of fuel and electricity, and the boundaries chosen for the evaluation. Tanaka et al. 2006 [NPR, SRC] ) also used a Monte Carlo approach to estimate the uncertainty in their projections of technical potential for three steel making technologies. Kim and Worrell 2002a [SRC] ) estimated economic potential by taking industry structure into account. They benchmarked the energy efficiency of steel production to the best practice performance in five countries with over 50% of world steel production, finding potential CO2 emission reductions due to energy efficiency improvement varying from 15% (Japan) to 40% (China, India and the USA). While China has made significant improvements in energy efficiency, reducing energy consumption per tonne of steel from 29.3 GJ in 1990 to 23.0 GJ in 2000 [7] ( Price et al., 2002 [SRC] ), there is still considerable potential for energy efficiency improvement and CO2 emission mitigation ( Kim and Worrell, 2002a [SRC] ). Planned improvements include greater use of continuous casting and near-net shape casting, injection of pulverized coal, increased heat and energy recovery and improved furnace technology ( Zhou et al., 2003 [NPR, ARC] ). A study in 2000 estimated the 2010 global technical potential for energy efficiency improvement with existing technologies at 24% ( De Beer et al., 2000a [NPR, SRC] ) and that an additional 5% could be achieved by 2020 using advanced technologies such as smelt reduction and near net shape casting.
ULCOS (Ultra-Low CO2 Steel making), a consortium of 48 European companies and organizations, has as its goal the development of steel making technology that reduces CO2 emission by at least 50%. The technologies being evaluated, including CCS, biomass and hydrogen reduction, show a potential for controlling emissions to 0.5 to 1.5 tCO2/t (0.14 to 0.41 tC/t) steel ( Birat, 2005 [NPR] ). Economics may limit the achievable emission reduction potential. A study of the US steel industry found a 2010 technical potential for energy-efficiency improvement of 24% ( Worrell et al., 2001a [SRC] ), but economic potential, using a 30% hurdle rate, was only 18%, even accounting for the full benefits of the energy efficiency measures ( Worrell et al., 2003 [SRC] ). A similar study of the European steel industry found an economic potential of less than 13% ( De Beer et al., 2001 [NPR] ). These studies focused mainly on retrofit options. However, potential savings could be realized by a combination of capital stock turnover and retrofit of existing equipment. A recent analysis of the efficiency improvement of electric arc furnaces in the US steel industry found that the average efficiency improvement between 1990 and 2002 was 1.3%/yr, of which 0.7% was due to capital stock turnover and 0.5% due to retrofit of existing furnaces ( Worrell and Biermans, 2005 [SRC] ). Future efficiency developments will aim at further process Data is pluralintegration. The most important are near net shape casting ( Martin et al., 2000 [NPR, SRC] ), with current applications at numerous plants around the world; and smelt reduction, which integrates ore agglomeration, coke making and iron production in a single process, offering an energy-efficient alternative at small to medium scales ( De Beer et al., 1998 [NPR] ).
Fuel Switching. Coal (in the form of coke) is the main fuel in the iron and steel industry because it provides both the reducing agent and the flow characteristics required by blast furnaces in the production of iron. Steel-making processes produce large volumes of byproducts (e.g., coke oven and blast furnace gas) that are used as fuel. Hence, a change in coke use will affect the energy balance of an integrated iron and steel plant.
Technology enabling the use of oil, natural gas and pulverized coal to replace coke in iron-making has long been available. Use of this technology has been dictated by the relative costs of the fuels and the process limitations in iron-making furnaces. Use of oil and natural gas could reduce CO2 emissions. More recently, the steel industry has developed technologies that use wastes, such as plastics, as alternative fuel and raw materials ( Ziebek and Stanek, 2001 [MoS] ). Pretreated plastic wastes have been recycled in coke ovens and blast furnaces ( (Okuwaki, 2004 ) ), reducing CO2 emissions by reducing emissions from incineration and the demand for fossil fuels. In Brazil, charcoal is used as an alternative to coke in blast furnaces. While recent data are not available, use of charcoal declined in the late 1990 s, as merchant coke became cheaper ( Kim and Worrell, 2002a [SRC] ). The use of hydrogen to reduce iron ore is a longer-term technology discussed in Section 7.12 . CCS is another longer-term technology that might be applicable to steel making (see section 7.3.7 ).
7.4.2 Non-ferrous metals
The commercially relevant non-ferrous metals and specific and total CO2 emissions from electrode and reductant use are shown in Table 7.6 . Annual production of these metals ranges from approximately 30 Mt for aluminium to a few hundred kilotonnes for metals and alloys of less commercial importance. Production volumes are fairly low compared to some of the world’s key industrial materials like cement, steel, or paper. However, primary production of some of these metals from ore can be far more energy intensive. In addition, the production of these metals can result in the emission of high-GWP GHGs, for example PFCs in aluminium or SF6 in magnesium, which can add significantly to CO2-eq emissions.
Generally, the following production steps need to be considered: mining, ore refining and enrichment, primary smelting, secondary smelting, metal refining, rolling and casting. For most non-ferrous metals, primary smelting is the most energy-intensive step, but significant levels of emissions of fluorinated GHGs have been reported from the refining and casting steps.
Table 7.6: Emission factors and estimated global emissions from electrode use and reductant use for various non-ferrous metals
| CO2 emissions (tCO2/t product) | Global CO2 emissions (ktCO2) | |
|---|---|---|
| Primary aluminium | 1.55 | 44,700 |
| Ferrosilicon | 2.92 | 10,500 |
| Ferrochromium | 1.63 | 9,500 |
| Silicomanganese | 1.66 | 5,800 |
| Calcium carbide | 1.10 | 4,475 |
| Magnesium | 0.05 | 4,000 |
| Silicon metal | 4.85 | 3,500 |
| Lead | 0.64 | 3,270 |
| Zinc | 0.43 | 3,175 |
| Others | 6,000 | |
| Total | 91,000 | |
| Note: Indirect emissions and non-CO2 greenhouse-gas emissions are not included. Source: Sjardin, 2003. | ||
7.4.2.1 Aluminium
Global primary aluminium production was 29.9 Mt in 2004 ( IAI, 2006b [NPR, MoS] ) and has grown an average of 5% per year over the last ten years. Production is expected to grow by 3% per year for the next ten years. Recycled aluminium production was approximately 14 Mt in 2004 and is also expected to double by 2020 (Marchek, 2006 ).
Primary aluminium metal (Al) is produced by the electrolytic reduction of alumina (Al2O3) in a highly energy-intensive process. In addition to the CO2 emissions associated with electricity generation, the process itself is GHG-intensive. It involves a reaction between Al2O3 and a carbon anode: 2 Al2O3 + 3 C = 4 Al + 3 CO2. In the electrolysis cell, Al2O3 is dissolved in molten cryolite (Na3AlF6). If the flow of Al2O3 to the anode is lower than required, cryolite will react with the anode to form PFCs, CF4 and C2F6 ( IAI, 2001 [NPR] ). CF4 has a GWP [8] of 6500 and C2F6, which accounts for about 10% of the mix, has a GWP of 9200 ( IPCC, 1995 [NPR] ). These emissions can be significantly reduced by careful attention to operating procedures and more use of computer-control. Even larger reductions in emissions can be achieved by upgrading older cell technology (for example., Vertical Stud Södeberg or Side Worked Prebake) by addition of point feeders to better control alumina feeding. The cost of such a retrofit can be recovered through the improved productivity. Use of the newer technologies, which require a major retrofit, can cost up to 27 US$/tCO2-eq (99 US$/tC-eq) ( US EPA, 2006a [NPR] ).
Members of the International Aluminium Institute (IAI), responsible for more than 70% of the world’s primary aluminium production, have committed to an 80% reduction in PFC emissions intensity for the industry as a whole, and to a 10% reduction in smelting energy intensity by 2010 compared to 1990 for IAI member companies. IAI data ( IAI, 2006a [NPR] ) shows a reduction in CF4 emissions intensity from 0.60 to 0.16 kg/t Al, and a reduction in C2F6 emissions intensity from 0.058 to 0.016 kg/t Al between 1990 and 2004, with best available technology having a median emission rate of only 0.05 kg CF4/t in 2004 . Overall, PFC emissions from the electrolysis process dropped from 4.4 to 1.2 tCO2-eq/t (1.2 to 0.3 tC-eq/t) Al metal produced. IAI data ( IAI, 2006b [NPR, MoS] ) show a 6% reduction in smelting energy use between 1990 and 2004 .
Benchmarking has been used to identify opportunities for emission reductions. The steps taken to control these emissions have been mainly low or no-cost, and have commonly been connected to smelter retrofit, conversion, or replacements ( Harnisch et al., 1998 [NPR, SRC] ; IEA GHG 2000 [NPR] ). However, much of the 30% of production from non-IAI members still uses older technology ( EDGAR, 2005 [NPR, MoS] ).
SF6 (GWP = 23,900 ( IPCC, 1995 [NPR] )) has been used for stirring and degassing of molten aluminium in secondary smelters and foundries ( Linde, 2005 [NPR] ). The process is not very common because of cost and technical problems ( UBA, 2004 [NPR] ). Current level of use is unknown, but is believed to be much smaller than SF6 used in magnesium production.
The main potentials for additional CO2-eq emission reductions are a further penetration of state-of-the-art, point feed, prebake smelter technology and process control plus an increase of recycling rates for old-scrap ( IEA GHG, 2001 [NPR] ). Research is proceeding on development of an inert anode that would eliminate anode-related CO2 and PFC emissions from Al smelting. A commercially viable design is expected by 2020 (The Aluminium Association, 2003 ). However, IEA 2006a [NPR] ) notes that the ultimate technical feasibility of inert anodes has yet to be proven, despite 25 years of research.
7.4.2.2 Magnesium
Magnesium, produced in low volumes, is very energy intensive. Its growth rate has been high due to increasing use of this lightweight metal in the transport industry. SF6 is quite commonly used as cover gas for casting the primary metal into ingots and for die casting magnesium. Estimates of global SF6 emissions from these sources in 2000 range from about 9 MtCO2-eq (2.4 MtC-eq) ( US EPA, 2006a [NPR] ), to about 20 MtCO2-eq (5.5 MtC-eq) ( EDGAR, 2005 [NPR, MoS] ). The later value is about equal to energy related emissions from the production of magnesium. Harnisch and Schwarz 2003 [NPR, SRC] ) found that the majority of these emissions can be abated for <1.2 US$/tCO2-eq (<4.4 US$/tC-eq) by using SO2, the traditional cover gas, which is toxic and corrosive, or using more advanced fluorinated cover gases with low GWPs. US EPA 2006a [NPR] ) report similar results. Significant parts of the global magnesium industry located in Russia and China still use SO2 as a cover gas. The International Magnesium Association, which represented about half of global magnesium production in 2002, has committed its member companies to phasing out SF6 use by 2011 ( US EPA, 2006a [NPR] ).
Table 7.7: Greenhouse-gas emission from production of various non-ferrous metals
| Metal | Global emissions (MtCO2-eq/yr) | Source and year |
|---|---|---|
| Aluminium | ||
| CO2 - Mining and refining | 109 | IEA GHG, 2000 for 1995 |
| CO2 - Electrodes | 48 | IAI, 2006b for 2004 |
| PFC - Emissions | 69 | EDGAR, 2005 for 2000 |
| CO2 - Electricity | 300 | IEA GHG, 2001 for 1995 |
| Magnesium | ||
| CO2 - Electrode and cell-feed | 4 | Sjardin, 2003 for 1995 |
| SF6 - Production and casting | 9 | US EPA, 2006b for 2000 |
| CO2 - Electricity | Unknown | |
| CO2 - Other steps in the production process | Unknown | |
| All other non-ferrous metals | ||
| CO2 - Process | 40 | Sjardin, 2003 |
| CO2 - Electricity | Unknown | |
| CO2 - Other steps | Unknown | |
| All non-ferrous metals | Approximately 500 (lower bound) |
7.4.2.3 Total emissions and reduction potentials
Table 7.7 gives the lower bounds for key emission sources in the non-ferrous metal industry. Total annual GHG gas emissions from the non-ferrous metal industry were at least 500 MtCO2-eq (140 MtC-eq) in 2000 . The GHG abatement options for the production of non-ferrous metals other than aluminium are still fairly uncertain. In the past, these industries have been considered too small or too complex regarding raw materials, production technologies and product qualities, to be systematically assessed for reduction options.
7.4.3 Chemicals and fertilizers
The chemical industry is highly diverse, with thousands of companies producing tens of thousands of products in quantities varying from a few kilograms to thousand of tonnes. Because of this complexity, reliable data on GHG emissions is not available ( Worrell et al., 2000a [NPR, SRC] ). The majority of the CO2-eq direct emissions from the chemical industry are in the form of CO2, the largest sources being the production of ethylene and other petrochemicals, ammonia for nitrogen-based fertilizers, and chlorine. These emissions are from both energy use and from venting and incineration of byproducts. In addition, some chemical processes create other GHGs as byproducts, for example N2O from adipic acid, nitric acid and caprolactam manufacture; HFC-23 from HCFC-22 manufacture; and very small amounts of CH4 from the manufacture of silicon carbide and some petrochemicals. Pharmaceutical manufacture uses relatively little energy, most of which is used in the buildings that house industrial facilities ( Galitsky and Worrell, 2004 [NPR, SRC] ).
The chemical industry makes use of many of the sector-wide technologies described in Section 7.3 . Much of the petro-chemical industry is co-located with petroleum refining, creating many opportunities for process integration and cogeneration of heat and electricity. Both industries make use of the energy in byproducts that would otherwise be vented or flared, contributing to GHG emissions. Galitsky and Worrell 2004 [NPR, SRC] ) identify separations, chemical synthesis and process heating as the major energy consumers in the chemical industry, and list examples of technology advances that could reduce energy consumption in each area, for example improved membranes for separations, more selective catalysts for synthesis and greater process integration to reduce process heating requirements. Longer-term, biological processing offers the potential of lower energy routes to chemical products (See Section 7.12.1 ).
7.4.3.1 Ethylene
Ethylene, which is used in the production of plastics and many other products, is produced by steam cracking hydrocarbon feedstocks, from ethane to gas oil. Hydrogen, methane, propylene and heavier hydrocarbons are produced as byproducts. The heavier the feedstock, the more and heavier the byproducts, and the more energy consumed per tonne of ethylene produced ( Worrell et al., 2000a [NPR, SRC] Ren et al. 2006 [NPR, ARC] ) report that steam cracking for olefin production is the most energy consuming process in the chemicals industry, accounting for emissions of about 180 MtCO2/yr (49MtC/yr), but that significant reductions are possible. Cracking consumes about 65% of the total energy used in ethylene production, but use of state-of-the-art technologies (e.g., improved furnace and cracking tube materials and cogeneration using furnace exhaust) could save up to about 20% of total energy. The remainder of the energy is used for separation of the ethylene product, typically by low-temperature distillation and compression. Up to 15% total energy can be saved by improved separation and compression techniques (e.g., absorption technologies for separation). Catalytic cracking also offers the potential for reduced energy use, with a savings of up to 20% of total energy. This savings is not additional to the energy savings for improved steam cracking ( Ren et al., 2006 [NPR, ARC] ). Processes have been developed for converting methane in natural gas to olefins as an alternative to steam cracking. However, Ren et al. 2005 [NPR, ARC] ) conclude that the most efficient of these processes uses more than twice as much primary energy as state-of-the-art steam cracking of naphtha.
7.4.3.2 Fertilizer manufacture
Swaminathan and Sukalac 2004 [NPR] ) report that the fertilizer industry uses about 1.2% of world energy consumption and is responsible for about the same share of global GHG emissions. More than 90% of this energy is used in the production of ammonia (NH3). However, as the result of energy efficiency improvements, modern ammonia plants are designed to use about half the energy per tonne of product than those designed in 1960 s, (see Figure 7.2 ), with design energy consumption dropping from over 60 GJ/t NH3 in the 1960 s to 28 GJ/t NH3 in the latest design plants, approaching the thermodynamic limit of about 19 GJ/t NH3, and limiting scope for further efficiency increases. Benchmarking data indicate that the best-in-class performance of operating plants ranges from 28.0 to 29.3 GJ/t NH3 ( Chaudhary, 2001 [NPR, MoS] ; PSI, 2004 [NPR] ).
Figure 7.2: Design energy consumption trends in world ammonia plants
Sources: Chaudhary, 2001 [NPR, MoS] ; PSI, 2004 [NPR] .
The newest plants tend to have the best energy performance, and many of them are located in developing countries, which now account for 57% of nitrogen fertilizer production ( IFA, 2004 [NPR] ). Individual differences in energy performance are mostly determined by feedstock (natural gas compared with heavier hydrocarbons) and the age and size of the ammonia plant ( PSI, 2004 [NPR] , Phylipsen et al., 2002 [Ambiguous] ). National and regional averages are strongly influenced by whether the sector has undergone restructuring, which tends to drive less efficient producers out of the market ( Sukalac, 2005 [NPR] ). Ammonia plants that use natural gas as a feed-stock have an energy efficiency advantage over plants that use heavier feedstocks and a high percentage of global ammonia capacity already is based on natural gas. China is an exception in that 67% of its ammonia production is based on coal ( CESP, 2004 [NPR] ) and small-scale plants account for 90% of the coal-based production. The average energy intensity of Chinese coal-based production is about 53 GJ/t, compared with a global average of 41.4 GJ/t ( Giehlen, 2006 [NPR] ).
Retrofit of old plants is feasible and offers a potential for improved efficiency. Verduijn and de Wit 2001 [NPR] ) concluded that the energy efficiency of large single train ammonia plants, the bulk of existing capacity, could be improved at reasonable cost to levels approaching newly designed plants, provided that the upgrading is accompanied by an increase in capacity. Significant reductions of CO2 emissions, below those achieved by state-of-the-art ammonia plants, could be achieved by using low-carbon or carbon-free hydrogen, which could be obtained through the application of CCS technology (see Section 7.3.7 ), biomass gasification, or electrolysis of water using electricity from nuclear or renewables. About half the ammonia produced for fertilizer is reacted with CO2 to form urea (UNIDO and IFDC, 1998 [NotFound] ), but the CO2 is released when the fertilizer is applied. However, this use of CO2 reduces the potential for applying CCS technology.
7.4.3.3 Chlorine manufacture
The TAR ( IPCC, 2001a [NPR] ) reported on the growing use of more energy-efficient membrane electrolysis cells for chlorine production. There have been no significant developments affecting GHG emissions from chlorine production since the TAR.
Global N2O emissions from caprolactam production in 2000 were estimated at 10 to 15 MtCO2-eq (2.7 to 4.1 MtC) ( EDGAR, 2005 [NPR, MoS] IPCC 2006 [NPR] ) indicates that these emissions can be controlled to a high degree by non-specific catalytic reduction.
7.4.3.4 N2O emissions from adipic acid, nitric acid and caprolactam manufacture
N2O emissions from nitric and adipic acid plants account for about 5% of anthropogenic N2O emissions. Due to significant investment in control technologies by industry in North America, Japan and the EU, worldwide emissions of N2O (GWP = 310 ( IPCC, 1995 [NPR] )) from adipic and nitric acid production decreased by 30%, from 223 MtCO2-eq (61 MtC-eq) in 1990 to 154 MtCO2-eq (42 MtC-eq) in 2000 ( US EPA 2006b [NPR] ). Some of the reduction was due to the installation of NO control technology to meet regulatory requirements. By 2020, global emission from the manufacture of adipic acid and from the manufacture of nitric acid are projected to grow to 177 MtCO2-eq (48 MtC-eq). Developed nations account for approximately 55% of emissions in both 2000 and 2020 ( US EPA, 2006b [NPR] ). Experience in the USA, Japan and the EU shows that thermal destruction can eliminate 96% of the N2O emitted from an adipic acid plant. Catalytic reduction can eliminate 89% of the N2O emitted from a typical nitric plant in a developed country ( US EPA, 2006a [NPR] ). Mitigation potential at nitric acid plants can range from 70% to almost 100% depending on the catalyst and plant operating conditions ( US EPA, 2001 [NPR] , Continental Engineering BV, 2001 ). Costs range from 2.0 to 5.8 US$/tCO2-eq (7.3 to 21.2 US$/tC-eq) ( 2000 US$) using a 20% discount rate and a 40% corporate tax rate, and a maximum mitigation potential of 174 MtCO2-eq (44 MtC-eq) is projected in 2030 .
7.4.3.5 HFC-23 emissions from HCFC-22 manufacture
On average, 2.3% HFC-23 (GWP = 11,700 ( IPCC, 1995 [NPR] )) is produced as a byproduct of HCFC-22 manufacture. The EDGAR database estimated 2000 emissions at 78 MtCO2-eq (21 MtC-eq) ( EDGAR, 2005 [NPR, MoS] ), while the US EPA estimated 96 MtCO2-eq (26 MtC-eq) ( US EPA, 2006a [NPR] ). HCFC-22 has been used as a refrigerant, but under the Montreal Protocol its consumption is scheduled to end by 2020 in developed countries and over a longer period in developing countries. However, production of HCFC-22 for use as a feedstock in the manufacture of fluoropolymers, plastics and HFCs is expected to grow, leading to increasing emissions through 2015 in the business-as-usual case. Data on production rates and control technologies are contained in the IPCC Special Report on Safeguarding the Ozone Layer and the Global Climate System (IPCC/TEAP, 2005 ). Capture and destruction by thermal oxidation is a highly effective option for reducing HFC-23 emissions at a cost of less than 0.20 to 0.35 US$/tCO2-eq (0.75 to 1.20 US$/tC-eq) (IPCC/TEAP, 2005, US EPA, 2006a [NPR] ).
Refineries use hydrogen to remove sulphur and other impurities from products, and to process heavy hydrocarbons into lighter components for use in gasoline and distillate fuels. The hydrogen is supplied from reformer gas, a hydrogen-rich byproduct of catalytic reforming, and a process for upgrading gasoline components. If this source is insufficient for the refinery’s needs, hydrogen is manufactured by gasification of fossil fuels. US refineries use about 4% of their energy input to manufacture hydrogen ( Worrell and Galitsky, 2005 [NPR, SRC] ). Hydrogen production produces a CO2-rich stream, which is a candidate for CCS (see Section 7.3.7 ).
7.4.4 Petroleum refining
As of the beginning of 2004, there were 735 refineries in 128 countries with a total crude oil distillation capacity of 82.3 million barrels per day. The U.S (20.5%), EU-25 (16.4%), Russia (6.6%), Japan (5.7%) and China (5.5%) had the largest shares of this capacity ( EIA, 2005 [NPR, MoS] ). Petroleum industry operations consume up to 15 to 20% of the energy in crude oil, or 5 to 7% of world primary energy, with refineries consuming most of that energy ( Eidt, 2004 [NPR] ). Comparison of energy or CO2 intensities among countries is not practical because refining energy use is a complex function of crude and product slates and processing equipment. Simple metrics (e.g., energy consumed/barrel refined) do not account for that complexity. The shifts towards heavier crude and lower sulphur products will increase refinery energy use and CO2 emissions. One study indicated that the combination of heavier crude and a 10 ppm maximum gasoline and diesel sulphur content would increase European refinery CO2 emissions by about 6% ( CONCAWE, 2005 [NPR] ).
Worrell and Galitsky 2005 [NPR, SRC] ), based on a survey of US refinery operations, found that most petroleum refineries can economically improve energy efficiency by 10–20%, and provided a list of over 100 potential energy saving steps. Key items included: use of cogeneration, improved heat integration, combustion optimization, control of compressed air and steam leaks and use of efficient electrical devices. The petroleum industry has had long-standing energy efficiency programmes for refineries and the chemical plants with which they are often integrated. These efforts have yielded significant results. Exxon Mobil reported over 35% reduction in energy use in its refineries and chemical plants from 1974 to 1999, and in 2000 instituted a programme whose goal was a further 15% reduction, which would reduce emissions by an additional 12 MtCO2/yr. ( Eidt, 2004 [NPR] ). Chevron ( 2005 ) reported a 24% reduction in its index of energy use between 1992 and 2004 . Shell ( 2005 ) reported energy efficiency improvements of 3 to 7% at its refineries and chemical plants. Efficiency improvements are expected to continue as technology improves and energy prices rise.
Refineries typically use a wide variety of gaseous and liquid byproducts as fuel. Byproducts that are not used as fuel are flared. Reducing the amount of material flared will increase refinery energy efficiency and decrease CO2 emissions, and has become an objective for refinery management worldwide, though flare reduction projects are often undertaken to reduce local environmental impacts Munn 2004 [NPR] ). No estimate of the incremental reduction in CO2 emissions is available.
7.4.5 Minerals
7.4.5.1 Cement
Cement is produced in nearly all countries. Cement consumption is closely related to construction activity and to general economic activity. Global cement production grew from 594 Mt in 1970 to 2200 Mt in 2005, with the vast majority of the growth occurring in developing countries. In 2004 developed countries produced 570 Mt (27% of world production) and developing countries 1560 Mt (73%) ( USGS, 2005 [NPR] ). China has almost half the world’s cement capacity, manufacturing an estimated 1000 Mt in 2005 (47% of global production), followed by India with a production of 130 Mt in 2005 ( USGS, 2006 [NPR] ). Global cement consumption is growing at about 2.5%/yr.
The production of clinker, the principal component of cement, emits CO2 from the calcination of limestone. Cement production is also highly energy-intensive. The major energy uses are fuel for the production of clinker and electricity for grinding raw materials and the finished cement. Coal dominates in clinker making. Based on average emission intensities, total emissions in 2003 are estimated at 1587 MtCO2 (432 MtC) to 1697 MtCO2 (462 MtC), or about 5% of global CO2 emissions, half from process emissions and half from direct energy use. Global average CO2 emission per tonne cement production is estimated by Worrell et al. 2001b [SRC] ) at 814 kg (222 kg C), while Humphreys and Mahasenan 2002 [NPR] ) estimated 870 kg (264 kg C). CO2 emission/t cement vary by region from a low of 700 kg (190 kg C) in Western Europe and 730 kg (200 kg C) in Japan and South Korea, to a high of 900, 930, and 935 kg (245, 253 and 255 kg C) in China, India and the United States ( Humphreys and Mahasenan, 2002 [NPR] ; Worrell et al., 2001b [SRC] ). The differences in emission intensity are due (in order of contribution) to differences in the clinker content of the cement produced, energy efficiency, carbon intensity of the clinker fuel and carbon intensity of power generation ( Kim and Worrell, 2002b [SRC] ).
Emission intensities have decreased by approximately 0.9%/yr since 1990 in Canada, 0.3%/yr ( 1970 – 1999 ) in the USA, and 1%/yr in Mexico ( Nyboer and Tu, 2003 [NPR] ; Worrell and Galitsky, 2004 [NPR, SRC] ;( Sheinbaum and Ozawa, 1998 ) ). A reduction in energy intensity in India since 1995 – 1996 has led to a reduction in emissions from the industry despite the increase in output ( Dasgupta and Roy, 2001 [MoS, SRC] ). Analysis of CO2 emission trends in four major cement-producing countries showed that energy efficiency improvement and reduction of clinker content in cement were the main factors contributing to emission reduction, while the carbon intensity of fuel mix in all countries increased slightly.
Both energy-related and process CO2 emissions can be reduced. The combined technical potential of these opportunities is estimated at 30% globally, varying between 20 and 50% for different regions ( Humphreys and Mahasenan, 2002 [NPR] ; Kim and Worrell, 2002b [SRC] ). Energy efficiency improvement has historically been the main contributor to emission reduction. Benchmarking and other studies have demonstrated a technical potential for up to 40% improvement in energy efficiency ( Kim and Worrell, 2002b [SRC] ; Worrell et al., 1995 [NPR, SRC] ). Countries with a high potential still use outdated technologies, like the wet process clinker kiln. Studies for the USA identified 30 opportunities in every production step in the cement-making process and estimated the economic potential for energy efficiency improvement in the US cement industry at 11%, reducing emissions by 5% ( Worrell et al., 2000b [SRC] ; Worrell and Galitsky, 2004 [NPR, SRC] ). The cement industry is capital intensive and equipment has a long lifetime, limiting the economic potential in the short term. The clinker kiln is an ideal candidate for the use of a wide variety of fuels, including waste-derived fuels, such as tyres, plastics, biomass, municipal solid wastes and sewage sludge (see Section 7.3.2 ). Section 7.3.7 discusses the potential for applying CCS in the cement industry.
Standard Portland cement contains 95% clinker. Clinker production is responsible for the process emissions and most of the energy-related emissions. The use of blended cement, in which clinker is replaced by alternative cementitious materials, for example blast furnace slag, fly ash from coal-fired power stations, and natural pozzolanes, results in lower CO2 emissions ( (Josa et al., 2004 ) Humphreys and Mahasenan 2002 [NPR] and Worrell et al. 1995 [NPR, SRC] ) estimate the potential for reduction of CO2 emissions at more than 7%. Current use of blended cement is relatively high in continental Europe and low in the USA and UK. Alternatives for limestone-based cement are also being investigated ( (Gartner, 2004; ) Humphreys and Mahasenan, 2002 [NPR] ). Geopolymers have been applied in niche markets, but have yet to be proven economical for large-scale application.
7.4.5.2 Lime
Generally lime refers both to high-calcium and dolomitic forms containing magnesium. Lime is produced by burning limestone or dolomite in small-scale vertical or large-scale rotary kilns. While in most industrialized countries the industry is concentrated in a small number of larger corporations, in most developing countries lime kilns are small operations using local technology. Even in industrialized countries like Greece there are independent small-scale vertical kilns in operation. Pulp and sugar mills may have captive lime production to internally regenerate lime. Lime is mainly used in a small number of industries (especially steel, but also chemicals, paper and sugar), mining, as well as for flue gas desulphurization. There are no detailed statistics on global lime production, however Miller 2003 [NPR] ) estimated global production at 120 Mt, excluding regenerated lime. The largest producers are China, the USA, Russia, Germany, Mexico and Brazil.
Process CO2 emissions from the calcination of limestone and dolomite are a function of the amounts of calcium carbonate, magnesium carbonate and impurity in the feedstock, and the degree of calcination. Theoretical process emissions are 785 kg CO2/t (214 kgC/t) calcium oxide and 1092 kg CO2/t (298 kgC/t) magnesium oxide produced. Energy use emissions are a function of the efficiency of the process, the fuel used, and indirect emissions from the electric power consumed in the process. In efficient lime kilns about 60% of the emissions are due to de-carbonisation of the raw materials. No estimates of global CO2 emissions due to lime production are available. In Europe process emissions are estimated at 750 kg CO2/t (205 kgC/t) lime ( IPPC, 2001 [NPR] ). For some applications, lime is re-carbonated, mitigating part of the emissions generated in the lime industry. Regeneration of lime in pulp and sugar mills does not necessarily lead to additional CO2 emissions, as the CO2 is from biomass sources ( Miner and Upton, 2002 [MoS, SRC] ). Emissions from fuel use vary with the kiln type, energy efficiency and fuel mix. Energy use is 3.6 to 7.5 GJ/t lime in the EU ( IPPC, 2001 [NPR] ), 7.2 GJ/t in Canada ( CIEEDAC, 2004 [NPR] ) and for lime kilns in US pulp mills ( Miner and Upton, 2002 [MoS, SRC] ), and up to 13.2 GJ/t for small vertical kilns in Thailand ( (Dankers, 1995 ) ). In Europe, fuel-related emissions are estimated at 0.2 to 0.45 tCO2/t (0.05 to 0.12 tC/t) lime ( IPPC, 2001 [NPR] ). Electricity use for lime production is 40 to 140 kWh/t lime, depending on the type of kiln and the required fineness of the lime ( IPPC, 2001 [NPR] ).
Emission reductions are possible by use of more efficient kilns ( (Dankers, 1995; ) IPPC, 2001 [NPR] ) and through improved management of existing kilns, using similar techniques to the cement industry (see Section 7.4.5.1 ). Switching to low-fossil carbon fuels can further reduce CO2 emissions. The use of solar energy has been investigated for small-scale installations ( Meier et al., 2004 [NPR] ). It may also be possible to reduce lime consumption in some processes, for example the sugar industry ( (Vaccari et al., 2005 ) ).
7.4.5.3 Glass
Glass is produced by melting raw materials (mainly silica, soda ash and limestone), and often cullet (recycled glass), in glass furnaces of different sizes and technologies. Typical furnace designs include: cross-fired or end-fired with regenerative air preheat, recuperative heat recovery and fuel-oxygen firing (EU-BREF Glass, 2001 [NPR] ). The industry is capital intensive, furnaces have a lifetime of up to 12 years and there are a limited number of technology providers. Natural gas and fuel oil are the main fuels used by the glass industry. Reliable international statistics on glass production are not available. The global glass industry is dominated by the production of container glass and flat glass. According to industry estimates the global production of container glass was 57 Mt in 2001 ( ISO, 2004 [NPR] ); production of flat glass was 38 Mt in 2004 (Pilkington, 2005 ). The production volumes of special glass, domestic glass, mineral wool and glass fibres are each smaller by roughly an order of magnitude.
Beerkens and van Limpt 2001 [NPR] ) report the energy intensity of continuous glass furnaces in Europe and the USA as 4 to 10 GJ/t of container glass and 5 to 8.5 GJ/t of flat glass, depending on the size and technology of the furnace and the share of cullet used. The energy consumption for batch production is higher, typically 12.5 to 30 GJ/t of product (Römpp, 1995 ). Assuming an average energy use of 7 GJ/t of product, half from natural gas and half from fuel oil, yields an emission factor of 450 kg energy related CO2/t of product. Globally, energy used in the production of container and flat glass results in emissions of approximately 40 to 50 MtCO2 (11 to 14 MtC) per year. Emissions from the decarbonisation of soda ash and limestone can contribute up to 200 kg CO2/t (55 kgC/t) of product depending on the composition of the glass and the amount of cullet used (EU-BREF Glass, 2001 [NPR] ).
The mid-term emission potential for energy efficiency improvements is less than half of what corresponds to the range of efficiencies reported by Beerkens and van Limpt 2001 [NPR] ), which also reflect differences in product quality and furnace age. The global potential for emissions reduction from fuel switching is unknown. The main mitigation options in the industry include: improved process control, increased use (up to 100%) of cullet (Kirk-Othmer, 2005 ), increased furnace size, use of regenerative heating, oxy-fuel technology, batch and cullet pre-heating, reduction of reject rates ( Beerkens and van Limpt, 2001 [NPR] ), use of natural gas instead of fuel oil, and CO2 capture for large oxy-fuel furnaces. High caloric value biogas could be used to reduce net CO2 emissions, but potential new break-through technologies are not in sight.
7.4.5.4 Ceramics
The range of commercial ceramics products is large and includes bricks, roof, wall and floor tiles, refractory ceramics, sanitary ware, tableware and cookware and other products. In terms of volume, the production of bricks and tiles dominate. The main raw materials used in the brick industry include clay and kaolin. Production technologies and respective energy efficiencies vary tremendously from large industrial operations to cottage and artisan production, which are still very common in many developing countries. The main fuels used in modern industrial kilns are natural gas and fuel oil. Specific energy consumption varies considerably for different products and kiln designs. The EU-BREF Ceramics 2005 [NPR] ) reported specific energy consumptions for modern industrial brick production of 1.4 to 2.4 GJ/t of product.
Small-scale kilns – used mainly for brick production – are often used in developing countries. Wood, agricultural residues and coal ( FAO, 1993 [NPR] ) are the main fuels used, with specific energy consumptions of 0.8 to 2.8 GJ/t of brick for the small- to medium sized kilns, and 2 to 8 GJ/t of brick for the very small-scale kilns used by cottage industries and artisans ( FAO, 1993 [NPR] ). Producers also utilize the energy contained in the organic fraction of clay and shale as well as in pore forming agents (e.g., sawdust) added to the clay in the production process. CO2 emissions from the calcination of carbonates contained in clay and shale typically contribute 20 to 50% of total emissions. The current choices of building materials and kiln technologies are closely related to local traditions, climate, and the costs of labour, capital, energy and transport, as well as the availability of alternative fuels, raw materials and construction materials.
Reliable international statistics on the production of ceramics products are not available. Consumption of bricks, tiles and other ceramic products in tonnes per capita per year is estimated at 1.2 in China ( Naiwei, 2004 [NPR] ); 0.4 in the EU (EU-BREF Ceramics, 2005 [NPR] ), 0.1 in the USA ( USGS, 2005 [NPR] ), and 0.25, 0.12, and 0.05 for Pakistan, India and Bangladesh ( FAO, 1993 [NPR] ). This suggests that the global production of ceramic products exceeds 2 Gt/yr, leading to the emission of more than 400 MtCO2 (110 MtC) per year from energy use and calcination of carbonates. Additional research to better understand the emission profile and mitigation options for the industry is needed.
GHG mitigation options include the use of more efficient kiln design and operating practices, fuel switching from coal to fuel oil, natural gas and biomass, and partial substitution of clay and shale by alternative raw materials such as fly ash. Mitigation options could also include the use of alternative building materials such as wood or bricks made from lime and sand. However, emissions over the whole life cycle of the products including their impact on the energy performance of the building need to be considered.
7.4.6 Pulp and paper
The pulp and paper industry is a highly diverse and increasing global industry. In 2003, developing countries produced 26% of paper and paperboard and 29% of global wood products; 31% of paper and paperboard output was traded internationally ( FAOSTAT, 2006 [NPR] ). Direct emissions from the pulp, paper, paperboard and wood products industries are estimated to be 264 MtCO2/yr (72 MtC/yr) ( Miner and Lucier, 2004 [NPR, SRC] ). The industry’s indirect emissions from purchased electricity are less certain, but are estimated to be 130 to 180 MtCO2/yr (35 to 50 MtC/yr) ( WBCSD, 2005 [NPR] ).
7.4.6.1 Mitigation options
Use of biomass fuels: The pulp and paper industry is more reliant on biomass fuels than any other industry. In developed countries biomass provides 64% of the fuels used by wood products facilities and 49% of the fuel used by pulp, paper and paperboard mills ( WBCSD, 2005 [NPR] ). Most of the biomass fuel used in the pulp and paper industry is spent pulping liquor, which contains dissolved lignin and other materials from the wood that are not used in paper production. The primary biomass fuel in the wood-products sector is manufacturing residuals that are not suitable for use as byproducts.
Use of combined heat and power: In 2002, the pulp and paper industry used cogeneration to produce 40% of its electricity requirements in the USA ( US DOE, 2002 [NPR] ) and over 30% in the EU ( CEPI, 2001 [NPR] ), and that use continues to grow.
Black liquor gasification: Black liquor is the residue from chemical processing to produce wood pulp for papermaking. It contains a significant amount of biomass and is currently being burned as a biomass fuel. R&D is underway on gasification of this material to increase the efficiency of energy recovery. Gasification could also create the potential to produce synfuels and apply CCS technology. IEA 2006a [NPR] ) estimates a 10 to 30 MtCO2 (2.7 to 8.1 MtC) mitigation potential for this technology in 2030 . While gasification would increase the energy efficiency of pulp and paper plants, the industry as a whole would not become a net exporter of biomass energy ( Farahani et al., 2004 [SRC] ).
Recycling: Recovery rates for waste paper (defined as the percentage of domestic consumption that is collected for reuse) in developed countries are typically at least 50% and are over 65% in Japan and parts of Europe ( WBCSD, 2005 [NPR] ). Globally, the utilization rate (defined as the fraction of fibre feedstock supplied by recovered fibre) was about 44% in 2004 ( IEA, 2006a [NPR] ). The impact of this recycling is complex, affecting the emissions profile of paper plants, forests and landfills. A number of studies examine the impacts of recycling on life-cycle GHG emissions ( (Pickens et al., 2002, ) ( Bystrom and Lonnstedt, 1997 ) ). These and other studies vary in terms of boundary conditions and assumptions about end-of-life management, and none attempt to examine potential indirect impacts of recycling on market-based decisions to leave land in forest rather than convert it to other uses. Although most (but not all) of these studies find that paper recycling reduces life-cycle emissions of GHG compared to other means of managing used paper, the analyses are dependent on study boundary conditions and site-specific factors and it is not yet possible to develop reliable estimates of the global mitigation potential related to recycling. However, both the USA ( US EPA, 2002 EU [NPR] EU (EC, 2004 [NPR] ) identify paper recycling as a GHG emissions reduction option.
7.4.6.2 Emission reduction potential
Because of increased use of biomass and energy efficiency improvements, the GHG emissions from the pulp and paper industry have been reduced over time. Since 1990, CO2 emission intensity of the European paper industry has decreased by approximately 25% ( WBCSD, 2005 [NPR] ), the Australian pulp and paper industry about 20% (A3P, 2006 ), and the Canadian pulp and paper industry over 40% (FPAC, n.d.). Fossil fuel use by the US pulp and paper industry declined by more than 50% between 1972 and 2002 ( AF & PA, 2004 [NPR] ). However, despite these improvements, Martin et al. 2000 [NPR, SRC] ) found a technical potential for GHG reduction of 25% and a cost-effective potential of 14% through widespread adoption of 45 energy-saving technologies and measures in the US pulp and paper industry. ( Möllersten et al. 2003 ) ) found that CO2 emissions from the Swedish pulp and paper industry could be reduced by 0.5 to 5.0 MtCO2/yr (0.14 to 1.4 MtC/yr) at negative cost using commercially available technologies, primarily by generating more biomass-based electricity to displace carbon-intensive electricity from the grid. The large variation in the results reflected varying assumptions about the carbon intensity of displaced electricity and the impacts of ‘industrial valuation’ compared with ‘societal valuation’ of capital. Inter-country comparisons of energy-intensity in the mid- 1990 s suggest that fuel consumption by the pulp and paper industry could be reduced by 20% or more in a number of countries by adopting best practices ( Farla et al., 1997 [ARC] ).
Corn refining, including wet corn milling, has been the fastest growing market for US agriculture over the past twenty years ( CRA, 2002 [NPR] ). Further growth is projected as a result of the demand for ethanol as an automotive fuel. Corn wet milling is the most energy-intensive food industry, using 15% of total US food industry energy ( EIA, 2002 [NPR] ). Over 100 technologies and measures for improving energy efficiency of corn wet milling have been identified ( Galitsky et al., 2003 [NPR, SRC] ).
7.4.7 Food
Most food industry products are major commercial commodities, particularly for developing countries, and are quite energy-intensive. The most important products from a climate perspective are sugar, palm oil, starch and corn refining, since these can be a source of fuel products. The sugar cane industry produces 1.2 Gt sugar/yr. ( Banda, 2002 [NPR] ) from about 1670 mills, mostly located in tropical developing countries ( Sims, 2002 [NPR, ARC] ). Edible oils are another significant product, the exports of which support many developing country economies. Malaysia, the world’s largest producer and exporter of palm oil, has 3.5 Mha under palm oil production ( UNDP, 2002 [NPR] ), whilst Sri Lanka, the world’s fourth largest producer of coconut oils, has 0.4 Mha under cultivation ( (Kumar et al., 2003 ) ).
7.4.7.1 Production processes, emissions and emission intensities
The main production processes for the food industry are almost identical, involving preparatory stages including crushing, processing/refining, drying and packaging. Most produce process residuals, which typically go to waste. Food production requires electricity, process steam and thermal energy, which in most cases are produced from fossil fuels. The major GHG emissions from the food industry are CO2 from fossil fuel combustion in boilers and furnaces, CH4 (GWP=21 ( IPCC, 1995 [NPR] )) and N2O (GWP = 310 ( IPCC, 1995 [NPR] )) from waste water systems.
The largest source of food industry emissions is CH4 from waste water treatment, which could be recovered for energy generation. For example, the Malaysian palm oil industry emits an estimated 5.17 MtCO2-eq (1.4 MtC-eq) from open-ponding systems that could generate 2.25 GWh of electricity while significantly reducing GHG emissions ( Yeoh, 2004 [NPR] ). Emissions from the Thai starch industry ( Cohen, 2001 [NPR] ) are estimated at 370 ktCO2-eq/yr (101 ktC-eq/yr), 88% were from waste water treatment, 8% from combustion of fuel oil and 4% from grid electricity. Although individual food industry factory emissions are low, their cumulative effect is significant in view of the large numbers of factories in both developed and developing countries. Typical energy intensities estimated at about 11 GJ/t for edible oils, 5 GJ/t for sugar and 10 GJ/t for canning operations ( UNIDO, 2002 [NPR] ).
7.4.7.2 Mitigation opportunities
The most important mitigation opportunities to reduce food industry GHG emissions in the near- and medium-term include technology and processes related to good housekeeping and improved management, improvements in both cross-cutting systems (e.g., boilers, steam and hot water distribution, pumps, compressors and fans) and process-specific technologies, improved process controls, more efficient process designs and process integration ( Galitsky et al., 2001 [SRC] ), cogeneration to produce electricity for own use and export ( Cornland, 2001 [NPR, SRC] ), and anaerobic digestion of residues to produce biogas for electricity generation and/or process steam ( Yeoh, 2004 [NPR] ). These technologies were discussed in Section 7.3 , but some specific food industry applications are presented below.
In Brazil, electricity sales to the grid from bagasse cogene-ration reached 1.6 TWh in 2005 from an installed capacity of 400 MW. This capacity is expected to increase to 1000 MW with implementation of a government-induced voluntary industry programme ( (Moreira, 2006 ) ). In India, the sugar industry has diversified into cogeneration of power and production of fuel ethanol. Cogeneration began in 1993 – 1994, and as of 2004 reached 680 MW. Full industry potential is estimated at 3500 MW. In 2001, India instituted a mixed fuel programme requiring use of a 5% ethanol blend, which will create an annual demand for 500 M litres of ethanol ( Balasubramaniam, 2005 [NPR] ).
Application of traditional boilers with improved combustion and CEST (Condensing Extraction Steam Turbines) in the southern African sugar industry could produce surpluses of 135 MW for irrigation purposes and 1620 MW for export to the national grid ( Yamba and Matsika, 2003 [NPR, SRC] ) in 2010 Sims 2002 [NPR, ARC] ) found that if all 31 of Australia’s existing sugar mills were converted to CEST technology, they could generate 20 TWh/yr of electricity and reduce emissions by 16 MtCO2/yr (4.4 MtC/yr), assuming they replaced coal-fired electricity generation. Gasifying the biomass and using it in combined cycle gas turbine could double the CO2 savings ( Cornland, 2001 [NPR, SRC] ). Proposed CDM projects in the Malaysian palm oil industry ( UNDP, 2002 [NPR] ), and the Thai starch industry ( Cohen, 2001 [NPR] ) demonstrated that use of advanced anaerobic methane reactors to produce electricity would yield a GHG emission reduction of 56 to 325 ktCO2-eq/yr (15 to 90 MtC-eq/yr). Application of improved energy management practices in the coconut industry ( (Kumar et al., 2003 ) ) and bakery industry ( (Kannan and Boy, 2003 ) ) showed significant saving of 40 to 60 % in energy consumption for the former and a modest saving of 6.5% for the latter. In the long term, use of residue biomass generated from the food industry in state-of-the-art Biomass Integrated Gasifier Combined Cycle (BIG/CC) technologies, could double electricity generation and GHG savings compared to CEST technology ( Yamba and Matsika, 2003 [NPR, SRC] ; Cornland et al., 2001 [NPR, SRC] ).
Virtually all countries have environmental regulations of varied stringency, which require installations including the food industry to limit final effluent BOD (Biochemical Oxygen Demand) in the waste water before discharge into waterways. Such measures are compelling industries to use more efficient waste water treatment systems. The recently introduced EU-directive requiring Best Available Techniques (BAT) as a condition for environmental permits in the fruit and vegetable processing industry ( (Dersden et al., 2002 ) ) will compel EU industry in this sector to introduce improved waste water purification processes thereby reducing fugitive emissions due to anaerobic reactions.
7.4.8 Other industries
This section covers a selection of other industries with significant emissions of high GWP gases. While some analyses include all emissions of these gases in the industrial sector, this chapter will consider only those which actually occur in the industrial sector. Thus, HFC and PFC emissions from use of automotive and residential air conditioning are covered in Chapter 5 , section 5.2.1 and Chapter 6 , section 6.8.4 respectively.
The manufacture of semiconductors, liquid crystal display and photovoltaic cells can result in the emissions of PFCs, SF6, NF3 and HFC-23 ( IPCC, 2006 [NPR] ). The technology available to reduce these emissions from semiconductor manufacturing, and the World Semiconductor Council (WSC) commitment to reduce PFC emissions by at least 10% by 2010 from 1995 -levels are discussed in the TAR ( IPCC, 2001a [NPR] US EPA 2006a [NPR] ) reports that emission levels from semiconductor manufacture were about 30 MtCO2-eq (7 MtC-eq) in 2000, and that significant growth in emissions will occur unless the WSC commitment is implemented globally and strengthened after 2010 US EPA 2006a [NPR] ) estimates that this 10% reduction could occur cost-effectively through replacement of C2F6 by C3F8 (which has a lower GWP), NF3 remote cleaning of the chemical vapour deposition chamber, or capturing and recycling of SF6. Emissions from the production of liquid crystal displays and photovoltaic cells, mainly located in Asia, Europe and the USA, are growing rapidly and mitigation options need further research.
SF6 emissions in 2000 from the production of medium and high voltage electrical transmission and distribution equipment were estimated at about 10 MtCO2-eq (2.8 MtC-eq) ( IEA GHG, 2001 [NPR] ). These emissions, mainly located in Europe and Japan, are estimated to have declined, despite a 60% growth in production between 1995 and 2003, mainly due to targeted training of staff and improved gas handling and test procedures at production sites. Emissions of SF6 at the end-of-life of electrical equipment are growing in relevance, and US EPA 2006b [NPR] ) estimates total SF6 emissions from production, use and disposal of electrical equipment at 27 MtCO2 in 2000 growing to 66 MtCO2 in 2020, if no mitigation actions are taken. Emissions from disposal of electrical equipment could be reduced by implementation of a comprehensive recovery system, addressing all entities involved in handling and dismantling this equipment ( Wartmann and Harnisch, 2005 [NPR, SRC] ).
A third group of industries that emits hydrofluorocarbons (HFCs) includes those manufacturing rigid foams, refrigeration and air conditioning equipment and aerosol cans, as well as industries using fluorinated compounds as solvents or for cleaning purposes. This group of industries previously used ozone-depleting substances (ODS), which are subject to declining production and use quotas defined under the Montreal Protocol. As part of the phase out of ODS, many of them have switched to HFCs as replacements, or intend to do so in the future. Mitigation options include improved containment, training of staff, improved recycling at the end-of-life, the use of very low GWP alternatives, and the application of not-in-kind technologies. A detailed discussion of use patterns, emission projections and mitigation options for these applications can be found in IEA GHG 2001 [NPR] ), IPCC/TEAP ( 2005 ) and more recent US EPA reports ( 2006a, b).
IEA GHG 2001 [NPR] ) estimated that global fugitive emissions from the production of HFCs will rise from 2 MtCO2-eq (0.6 MtC-eq) in 1996 to 8 MtCO2-eq (2.2 MtC-eq) by 2010 . Solvent and cleaning uses of HFCs and PFCs are commonly emissive despite containment and recycling measures. IEA GHG 2001 [NPR] ) forecast that these emissions would increase to up to 20 MtCO2-eq/yr (5.5 MtC-eq/yr) by 2020 . However other analyses suggest a more moderate growth in emissions from solvent applications to about 5 MtCO2-eq/yr (1.4 MtC-eq/yr) by 2020 (IPCC/TEAP, 2005 ).
7.4.9 Inter-industry options
Some options for reducing GHG emissions involve more than one industry, and may increase energy use in one industry to achieve a greater reduction in energy use in another industry or for the end-use consumer. For example, the use of granulated slag in Portland cement may increase energy use in the steel industry, but can reduce both energy consumption and CO2 emissions during cement production by about 40%. Depending on the concrete application, slag content can be as high as 60% of the cement, replacing an equivalent amount of clinker ( Cornish and Kerkhoff, 2004 [NPR] ). Lightweight materials (high-tensile steel, aluminium, magnesium, plastics and composites) often require more energy to produce than the heavier materials they replace, but their use in vehicles will reduce transport sector energy use, leading to an overall reduction in global energy consumption. Life-cycle calculations ( IAI, 2000 [NPR] ) indicate that the CO2 emission reductions in vehicles resulting from the weight reduction achieved by using aluminium more than offsets the GHG emissions from producing the aluminium.
Co-siting of industries can achieve GHG mitigation by allowing the use of byproducts as useful input and by integrating energy systems. In Kalundborg (Denmark) various industries (e.g., cement and pharmaceuticals production and a CHP plant) form an eco-industrial park that serves as an example of the integration of energy and material flows ( (Heeres et al., 2004 ) ). Heat-cascading systems, where waste heat from one industry is used by another, are a promising cross-industry option for saving energy. Based on the Second Law of Thermodynamics, ( Grothcurth et al. 1989 ) ) estimated up to 60% theoretical energy saving potential from heat cascading systems. However, Matsuhashi et al. 2000 [NPR, SRC] ) found the practical potential of these systems was limited to approximately 5% energy saving. Actual potential will depend on site-specific conditions.
7.5 Short- and medium-term mitigation potential and cost
Limited information is available on mitigation potential and cost [9] in industry, but it is sufficient to develop a global estimate for the industrial sector. Available studies vary widely with respect to system boundaries, baseline, time period, subsectors included, completeness of mitigation measures included, and economic factors (e.g., costs and discount rates). In many cases study assumptions are not specified, making it impossible to adjust the studies to a common basis, or to quantify overall uncertainty. A full discussion of the basis for evaluating costs in this report appears in Chapter 2 .5.
Table 7.8 presents an assessment of the industry-specific literature. Mitigation potential and cost for industrial CO2 emissions were estimated as follows:
(1) Price et al. 2006 [NPR, SRC] )s estimates for 2030 production rate by industry and geographic area for the SRES A1 and B2 scenarios ( IPCC, 2000b [NPR] ) were used.
(2) Literature estimates of mitigation potential were used, where available. In other cases, mitigation potential was estimated by assuming that current best practice could be achieved by all plants in 2030 .
(3) Literature estimates of mitigation cost were used, where available. When literature values were not available, expert judgment (informed by the available literature and data) was used to assign costs to mitigation technology.
Cost estimates are reported as 2030 mitigation potential below a given cost level. In most cases it was not possible to develop a marginal abatement cost curve that would allow estimation of mitigation potential as a function of cost. Estimates have not been made for some smaller industries (e.g., glass) and for the food industry. One or more of the critical inputs needed for these estimates were missing.
Table 7.8 should be interpreted with care. It is based on a limited number of studies sometimes only one study per industry and implicitly assumes that current trends will continue until 2030 . Key uncertainties in the projections include: the rate of technology development and diffusion, the cost of future technology, future energy and carbon prices, the level of industrial activity in 2030, and policy driver, both climate and non-climate. The use of two scenarios, A1B and B2, is an attempt to bracket the range of these uncertainties.
Table 7.8 projects 2030 mitigation potential for the industrial sector at a cost of <100 US$/tCO2-eq (<370 US$/tC-eq) of 3.0 to 6.3 GtCO2-eq/yr (0.8 to 1.7 GtC-eq/yr) under the A1B scenario, and 2.0 to 5.1 GtCO2-eq/yr (0.6 to 1.4 GtC-eq/yr) under the B2 scenario. The largest mitigation potentials are found in the steel, cement, and pulp and paper industries and in the control of non-CO2 gases. Much of that potential is available at <50 US$/tCO2-eq (<180 US$/tC-eq). Application of CCS technology offers a large additional potential, albeit at higher cost (low agreement, little evidence).
Some data are available on industrial sector mitigation potential and cost by country or region. However, an attempt to build-up a global estimate from this data was unsuccessful. Information was lacking for the former Soviet Union, Africa, Latin America and parts of Asia.
7.5.1 Electricity savings
Electricity savings are of particular interest, since they feedback into the mitigation potential calculation for the energy sector and because of the potential for double counting of the emissions reductions. Section 7.3.2 indicates that in the EU and USA electric motor driven systems account for about 65% of industrial energy use, and that efficient systems could reduce this use by 30%. About one-third of the savings potential was assumed to be realized in the baseline, resulting in a net mitigation potential of 13% of industrial electricity use. This mitigation potential was included in the estimates of mitigation potential for energy-intensive industries presented in Table 7.8 . However, it is also necessary to consider the potential for electricity savings from non-energy-intensive industries, which are large consumers of electricity.
The estimation procedure used to develop these numbers was as follows: Because data could not be found on other countries/regions, US data ( EIA, 2002 [NPR] ) on electricity use as a fraction of total energy use by industry and on the fraction of electricity use consumed by motor driven systems was taken as representative of global patterns. Based on de Keulenaer et al. 2004 [NPR] ) and Xenergy ( 1998 ), a 30% mitigation potential was assumed. Emission factors to convert electricity savings into CO2 reductions were derived from IEA data ( IEA, 2004 [NPR] ). The emission reduction potential from non-energy-intensive industries were calculated by subtracting the savings from energy-intensive industries from total industrial emissions reduction potential. Using the B2 baseline, 49% of total electricity savings are found in industries other than those identified in Table 7.8 .
Table 7.8: Mitigation potential and cost in 2030
| 2030 production (Mt)a | GHG intensity (tCO2-eq/t prod.) | Mitigation potential (%) | Cost range (US$) | Mitigation potential (MtCO2-eq/yr) | ||||
|---|---|---|---|---|---|---|---|---|
| Product | Areab | A1 | B2 | A1 | B2 | |||
| CO2 emissions from processes and energy use | ||||||||
| Steelc,d | Global | 1,163 | 1,121 | 1.6-3.8 | 15-40 | 20-50 | 430-1,500 | 420-1,500 |
| OECD | 370 | 326 | 1.6-2.0 | 15-40 | 20-50 | 90-300 | 80-260 | |
| EIT | 162 | 173 | 20.-3.8 | 25-40 | 20-50 | 80-240 | 85-260 | |
| Dev. Nat. | 639 | 623 | 1.6-3.8 | 25-40 | 20-50 | 260-970 | 250-940 | |
| Primary | Global | 39 | 37 | 8.4 | 15-25 | <100 | 53-82 | 49-75 |
| aluminiume,f | OECD | 12 | 11 | 8.5 | 15-25 | <100 | 16-25 | 15-22 |
| EIT | 9 | 6 | 8.6 | 15-25 | <100 | 12-19 | 8-13 | |
| Dev. Nat. | 19 | 20 | 8.3 | 15-25 | <100 | 25-38 | 26-40 | |
| Cementg,h,i | Global | 6,517 | 5,251 | 0.73-0.99 | 11-40 | <50 | 720-2,100 | 480-1,700 |
| OECD | 600 | 555 | 0.73-0.99 | 11-40 | <50 | 65-180 | 50-160 | |
| EIT | 362 | 181 | 0.81-0.89 | 11-40 | <50 | 40-120 | 20-60 | |
| Dev. Nat. | 5,555 | 4,515 | 0.82-0.93 | 11-40 | <50 | 610-1,800 | 410-1,500 | |
| Ethylenej | Global | 329 | 218 | 1.33 | 20 | <20 | 85 | 58 |
| OECD | 139 | 148 | 1.33 | 20 | <20 | 35 | 40 | |
| EIT | 19 | 11 | 1.33 | 20 | <20 | 5 | 3 | |
| Dev. Nat. | 170 | 59 | 1.33 | 20 | <20 | 45 | 15 | |
| Ammoniak,l | Global | 218 | 202 | 1.6-2.7 | 25 | <20 | 110 | 100 |
| OECD | 23 | 20 | 1.6-2.7 | 25 | <20 | 11 | 10 | |
| EIT | 21 | 23 | 1.6-2.7 | 25 | <20 | 10 | 12 | |
| Dev. Nat. | 175 | 159 | 1.6-2.7 | 25 | <20 | 87 | 80 | |
| Petroleum | Global | 4,691 | 4,508 | 0.32-0.64 | 10-20 | Half <20 | 150-300 | 140-280 |
| refiningm | OECD | 2,198 | 2,095 | 0.32-0.64 | 10-20 | Half <50 | 70-140 | 67-130 |
| EIT | 384 | 381 | 0.32-0.64 | 10-20 | 12-24 | 12-24 | ||
| Dev. Nat. | 2,108 | 2,031 | 0.32-0.64 | 10-20 | 68-140 | 65-130 | ||
| Pulp and papern | Global | 1,321 | 920 | 0.22-1.40 | 5-40 | <20 | 49-420 | 37-300 |
| OECD | 695 | 551 | 0.22-1.40 | 5-40 | <20 | 28-220 | 22-180 | |
| EIT | 65 | 39 | 0.22-1.40 | 5-40 | <20 | 3-21 | 2-13 | |
| Dev. Nat. | 561 | 330 | 0.22-1.40 | 5-40 | <20 | 18-180 | 13-110 | |
| 2030 production (Mt)a | CCS Potential (tCO2/t) | Mitigation potential (%) | Cost range (US$) | Mitigation potential (MtCO2-eq) | ||||
|---|---|---|---|---|---|---|---|---|
| Product | Areab | A1 | B2 | A1 | B2 | |||
| Carbon Capture and Storage | ||||||||
| Ammoniao,p | Global | 218 | 202 | 0.5 | about 100 | <50 | 150 | 140 |
| OECD | 23 | 20 | 0.5 | about 100 | <50 | 15 | 13 | |
| EIT | 21 | 23 | 0.5 | about 100 | <50 | 14 | 16 | |
| Dev. Nat. | 175 | 159 | 0.5 | about 100 | <50 | 120 | 110 | |
| Petroleum | Global | 4,691 | 4,508 | 0.032-0.064 | about 50 | <50 | 75-150 | 72-150 |
| Refiningm,p,q | OECD | 2,198 | 2,095 | 0.032-0.064 | about 50 | <50 | 35-70 | 34-70 |
| EIT | 384 | 381 | 0.032-0.064 | about 50 | <50 | 6-12 | 6-12 | |
| Dev. Nat. | 2,108 | 2,031 | 0.032-0.064 | about 50 | <50 | 34-70 | 32-65 | |
| Cementr | Global | 6,517 | 5,251 | 0.65-0.89 | about 6 | <100 | 250-350 | 200-280 |
| OECD | 600 | 555 | 0.65-0.80 | about 6 | <100 | 23-32 | 22-27 | |
| EIT | 362 | 181 | 0.73-0.80 | about 6 | <100 | 16-17 | 8-9 | |
| Dev. Nat. | 5,555 | 4,515 | 0.74-0.84 | about 6 | <100 | 210-300 | 170-240 | |
| Iron and Steel | Global | 1,163 | 1,121 | 0.32-0.76 | about 20 | <50 | 70-180 | 70-170 |
| OECD | 370 | 326 | 0.32-0.40 | about 20 | <50 | 24-30 | 21-26 | |
| EIT | 162 | 173 | 0.40-0.76 | about 20 | <50 | 13-25 | 14-26 | |
| Dev. Nat. | 639 | 623 | 0.32-0.76 | about 20 | <50 | 33-120 | 35-120 | |
| Non-CO2 gasesr | ||||||||
| Global | 668 | 37% <0US$ | 380 | |||||
| OECD | 305 | 53% <20US$ | 160 | |||||
| EIT | 53 | 55% <50US$ | 29 | |||||
| Dev. Nat. | 310 | 57%<100US$ | 190 | |||||
| Other industries, electricity conservations | ||||||||
| Global | 25% <20 | 1,100-1,300 | 410-540 | |||||
| OECD | 25% <50 | 140-210 | 65-140 | |||||
| EIT | 50% <100 | 340-350 | 71-85 | |||||
| Dev. Nat. | d | 640-700 | 280-320 | |||||
| Sumt,u,v,w | Global | 3,000-6,300 | 2,000-5,100 | |||||
| OECD | 580-1,300 | 470-1,100 | ||||||
| EIT | 540-830 | 250-510 | ||||||
| Dev. Nat. | 2,000-4,300 | 1,300-3,400 | ||||||
7.5.2 Non-CO2 gases
Table 7.9 shows mitigation potential for non-CO2 gases in 2030 based on a global study conducted by the US EPA 2006a [NPR] ,b), which projected emission and mitigation costs to 2020 . Emissions in 2030 were projected by linear extrapolation by region using 2010 and 2020 data. Mitigation costs were assumed to be constant between 2020 and 2030, and interpolated from US EPA data, which used different cost categories. The analysis uses US EPAs technical adoption scenario, which assumes that industry will continue meeting its voluntary commitments. The SRES A1B and B2 scenarios used as the base case for the rest of this chapter do not include sufficient detail on non-CO2 gases to allow a comparison of the two approaches. IPCC/TEAP ( 2005 ) contains significantly different estimates of 2015 baseline emissions for HFCs and PFCs in some sectors compared to Table 7.9 . We note that these emissions are reported by end-use, not by the sectoral approach used in this report, and that insufficient information is provided to extrapolate to 2030 . Caprolactam projections were not found in the literature. They were estimated based on historical data from a variety of industry sources. Mitigation costs and potentials were estimated by applying costs and potential from nitric acid production.
Table 7.9: Global mitigation potential in 2030 for non-CO2 gases
| Source | 2030 Baseline emissions (MtCO2-eq) | Mitigation potential by cost category (US$) | ||||
|---|---|---|---|---|---|---|
| <0 | <20 | <50 | <100 | |||
| N2O from adipic and nitric acid production | 190 | 158 | 158 | 158 | 174 | |
| N2O from caprolactam production | 20 | 16 | 16 | 16 | 16 | |
| PFC from aluminium production | 51 | 1.6 | 7.6 | 8.2 | 8.2 | |
| PFC and SF6 from semiconductor manufacture | 20 | 9.6 | 9.6 | 10 | 10 | |
| SF6 from use of electrical equipment (excluding manufacture) | 74 | 32 | 39 | 39 | 39 | |
| SF6 from magnesium production | 9.3 | 9.2 | 9.2 | 9.2 | 9.2 | |
| HFC-23 from HCFC-22 production | 106 | 0 | 86 | 86 | 86 | |
| ODSa substitutes: aerosols | 88 | 27 | 27 | 27 | 27 | |
| ODS substitutes: industrial refrigeration and cooling | 80 | 3.5 | 3.5 | 3.5 | 3.5 | |
| ODS substitutes: fire extinguishing | 27 | 0 | 0 | 6.3 | 6.7 | |
| ODS substitutes: solvents | 4.0 | 1.2 | 2.0 | 2.0 | 2.0 | |
| Total: | Global | 668 | 249 | 357 | 364 | 380 |
| OECDb | 305 | 135 | 154 | 157 | 158 | |
| Economies in Transition | 53 | 27 | 28 | 29 | 29 | |
| Developing Nations | 309 | 87 | 182 | 187 | 187 | |
| a ODS = Ozone-Depleting Substances b Regional information given in references. Source: Extrapolated from US EPA 2006a,b. | ||||||
7.5.3 Summary and comparison with other studies
Using the SRES B2 as a baseline (see Section 11.3.1 ), Table 7.10 summarizes the mitigation potential for the different cost categories. To avoid double counting, the total mitigation potential as given in Table 7.8 has been corrected for changes in emission factors of the transformation sectors to arrive at the figures included in Table 7.10 (see also Chapter 11 , table 11.3 ).
Table 7.10: Estimated economic potentials for GHG mitigation in industry in 2030 for different cost categories using the SRES B2 baseline
| Mitigation option | Region | Economic potential <100 US$/tCO2-eq | Economic potential in different cost categories | ||||
|---|---|---|---|---|---|---|---|
| Cost category (US$/tCO2-eq) | <0 | 0-20 | 20-50 | 50-100 | |||
| Cost category (US$/tC-eq) | <0 | 0-73 | 73-183 | 183-367 | |||
| Low | High | ||||||
| (MtCO2-eq) | |||||||
| Electricity savings | OECD | 300 | 70 | 70 | 150 | ||
| EIT | 80 | 20 | 20 | 40 | |||
| Non-OECD/EIT | 450 | 100 | 100 | 250 | |||
| Other savings, including non-CO2 GHG | OECD | 350 | 900 | 300 | 250 | 50 | |
| EIT | 200 | 450 | 80 | 250 | 20 | ||
| Non-OECD/EIT | 1,200 | 3,300 | 500 | 1,700 | 80 | ||
| Total | OECD | 600 | 1,200 | 350 | 350 | 200 | |
| EIT | 250 | 550 | 100 | 250 | 60 | ||
| Non-OECD/EIT | 1,600 | 3,800 | 600 | 1,800 | 300 | ||
| Global | 2,500 | 5,500 | 1,100 | 2,400 | 550 | ||
Two recent studies provide bottom-up, global estimates of GHG mitigation potential in the industrial sector in 2030 IEA 2006a [NPR] ) used its Energy Technology Perspectives Model (ETP), which belongs to the MARKAL family of bottom-up modelling tools, to estimate mitigation potential for CO2 from energy use in the industrial sector to be 5.4 Gt/yr (1.5 GtC/yr) in 2050 . IEAs base case was an extrapolation of its World Energy Outlook 2005 Reference Scenario, which projected energy use to 2030 . IEA provides ranges for mitigation potential in 2030 for nine groups of technologies totalling about 2.5 to 3.0 GtCO2/yr (0.68 to 0.82 GtC/yr). Mitigation cost is estimated at <25 US$/tCO2 (<92 US$/tC) ( 2004 US$). While IEAs estimate of mitigation potential is in the range found in this assessment, their estimate of mitigation cost is significantly lower.
ABARE ( Matysek et al., 2006 [NPR, SRC] ) used its general equilibrium model of the world economy (GTEM) to estimate the emission reduction potential associated with widespread adoption of advanced technologies in five key industries: iron and steel, cement, aluminium, pulp and paper, and mining. In the most optimistic ABARE scenario, industrial sector emissions across all gases are reduced by an average of about 1.54 GtCO2-eq/yr) (0.42GtC-eq/yr) over the 2001 to 2050 time frame and 2.8 GtCO2-eq/yr (0.77 GtC-eq/yr) over the 2030 - 2050 time frame, relative to the GTEM reference case, which assumes energy efficiency improvements and continuation of current or announced future government policy. The ABARE carbon dioxide only industry mitigation potential for the period 2030 2050 of approximately 1.94 GtCO2-eq/yr (0.53GtC/yr) falls below the range developed in this assessment. This outcome is the likely result of differences in the modelling approaches used ABAREs GTEM model is a top down model whereas the mitigation potentials in this assessment are developed using detailed bottom-up methodologies. ABARE did not estimate the cost of these reductions.
The TAR ( IPCC, 2001a [NPR] ) developed a bottom up estimate of mitigation potential in 2020 for the industrial sector of 1.4 to 1.6 GtC (5.1 to 5.9 GtCO2) based an SRES B2 scenario baseline and on the evaluation of specific technologies. Extrapolating the TAR estimate to 2030 would give values above the upper end of the range developed in this assessment. The newer studies used in this assessment take industry-specific conditions into account, which reduces the risk of double counting.
7.6 Barriers to industrial GHG mitigation
Full use of mitigation options is not being made in either industrialized or developing nations (high agreement, much evidence). In many areas of the world, GHG mitigation is neither demanded nor rewarded by the market or government. In these areas, companies will invest in GHG mitigation to the extent that other factors provide a return for their investments. This return can be economic, for example energy-efficiency improvements that show an economic payout. Nicholson 2004 [NPR] ) reported that the projects BP undertook to lower its CO2 emissions by 10% increased shareholder value by US$ 650 million. Alternatively, the return can be in terms of achievement of a larger corporate goal, for example DuPont’s commitment to cut its GHG emission by two-thirds as part of a larger commitment to sustainable growth ( Holliday, 2001 [NPR] ).
Even though a broad range of cost-effective GHG mitigation technologies exist, a variety of economic barriers prevent their full realisation in both developed and developing countries. Policies and measures must overcome the effective costs of capital ( Toman, 2003 [NPR, MoS] ). Industry needs a stable, transparent policy regime addressing both economic and environmental concerns to reduce the costs of capital.
The slow rate of capital stock turnover in many of the industries covered in this chapter is a barrier to mitigation ( Worrell and Biermans, 2005 [SRC] ). Excess capacity, as exists in some industries, can further slow capital stock turnover. Policies that encourage capital stock turnover, such as Japan’s programme to subsidize the installation of new high performance furnaces ( WEC, 2001 [NPR] ), will increase GHG mitigation. Companies must also take into consideration the risks involved with adopting a new technology, the payback period of a technology, the appropriate discount rate and transaction costs. Newer, relatively expensive technologies often have longer payback periods and represent a greater risk. Reliability is a key concern of industry, making new technologies less attractive ( Rosenberg, 1999 [NPR] ). Discount rates vary substantially across industries and little information exists on transaction costs of mitigation options ( US EPA, 2003 [NPR, MoS] ).
Resource constraints are also a significant barrier to mitigation. Unless legally mandated, GHG mitigation will have to compete for financial and technical resources against projects to achieve other company goals. Financial constraints can hinder diffusion of technologies within firms ( (Canepa and Stoneman, 2004 ) ). Projects to increase capacity or bring new products to the market typically have priority, especially in developing countries, where markets are growing rapidly and where a large portion of industrial capacity is in SMEs. Energy efficiency and other GHG mitigation technologies can provide attractive rates of return, but they tend to increase initial capital costs, which can be a barrier in locations where capital is limited. If the technology involved is new to the market in question, even if it is well-demonstrated elsewhere, the problem of raising capital may be further exacerbated ( Shashank, 2004 [NPR] ). Provision of funding for demonstration of the technology can overcome this barrier ( CPCB, 2005 [NPR] ).
The rate of technology transfer is another factor limiting the adoption of mitigation technologies. As documented in the IPCC Special Report Methodological and Technological Issues in Technology Transfer ( IPCC, 2000c [NPR] ), lack of an enabling environment is a barrier to technology transfer in some countries. Even when an enabling environment is present, the ability of industrial organizations to access and absorb information on technologies is limited. Access to information tends to be more of a problem in developing nations, but all companies, even the largest, have limited technical resources to interpret and translate the available information. The success of programmes such as US DOE’s Industrial Technologies Programs (ITP) and of the voluntary information sharing programmes discussed in Section 7.9.2 is evidence of the pervasiveness of this barrier.
McKane et al. 2005 [NPR] ) provide a case study of the interaction of some of these elements in their analysis of the barriers to the adoption of energy-efficient electric motors and motor-systems. These include: (1) industrial markets that focus on components, not systems; (2) energy efficiency not being a core mission for most industries, which results in a lack of internal support systems for mitigation goals; and (3) lack of technical skills to optimize the systems to the specific application – one size does not fit all. They found industrial energy efficiency standards a useful tool in overcoming these barriers.
7.7 Sustainable Development (SD) implications of industrial GHG mitigation
Although there is no universally accepted, practical definition of SD, the concept has evolved as the integration of economic, social and environmental aims ( IPCC, 2000a [NPR] ; Munasinghe, 2002 [ARC] ). Companies worldwide adopted Triple Bottom Line (financial, environmental and social responsibility) reporting in the late 1990 ’s. The Global Reporting Initiative (GRI, n.d.), a multi-stakeholder process, has enabled business organizations to account for and better explain their contributions to sustainable development. Companies are also reporting under Sigma Guidelines (The Sigma Project, 2003a ), and AA 1000 (The Sigma Project, 2003b ) and SA 8000 ( SAI, 2001 [NPR] ) procedures. Many companies are trying to demonstrate that their operations minimize water use and carbon emissions and produce zero solid waste ( ITC, 2006 [NPR] ). SD consequences can be observed or monitored through various indicators grouped under the three major categories. (See Section 12.1.1 and 12.1.3 for more detail).
However, the SD consequences of mitigation options are not automatic. GHG mitigation, per se, has little impact on four of the SD indicators: poverty reduction, empowerment/gender, water pollution and solid waste. The literature indicates that supplementing mitigation options with appropriate national macroeconomic policies, and with social and local waste reduction strategies at the company level (Tata Steel, Ltd., 2005 [NotFound] ; BEE, 2006 [NPR] ), has achieved some sustainability goals. Economy-wide impact studies ( Sathaye et al., 2005 [NPR, SRC] ; Phadke et al., 2005 [NPR, ARC] ) show that in developing countries, like India, adoption of efficient electricity technology can lead to higher employment and income generation. However, the lack of empirical studies leads to much uncertainty about the SD implications of many mitigation strategies, including use of renewables, fuel switching, feedstock and product changes, control of non-CO2 gases, and CCS. For example, fuel switching can have a positive effect on local air pollution and company profitability, but its impacts on employment are uncertain and will depend on inter-input substitution opportunities.
GHG emissions mitigation policies induce increased innovation that can reduce the energy and capital intensity of industry. However, this could come at the expense of other, even more valuable, productivity-enhancing investments or learning-by-doing efforts ( Goulder and Schneider, 1999 [PoC, JoC] ). If policies are successful in stimulating economic activity, they are also likely to stimulate increased energy use. GHG emissions would increase unless policies decreased the carbon-intensity of economic activity by more than the increase in activity. Due to energy efficiency improvements and fuel switching in OECD countries ( Schipper et al., 2000 [NPR] ;( Liskas et al., 2000 ) ), as well as in developing countries like India ( Dasgupta and Roy, 2001 [MoS, SRC] ), China ( Zhang, 2003 [NPR] ), Korea ( (Choi and Ang, 2001; ) ( Chang, 2003 ) ), Bangladesh (Bain, 2005 ), and Mexico ( (Aguayo and Gallagher, 2005 ) ), energy and carbon intensity have decreased, for the industry sector in general and for energy-intensive industries in particular. In Mexico, deindustrialization also played a role. For OECD countries, structural change has also played an important role in emissions reduction. However, overall economic activity has increased more rapidly, resulting in higher total carbon emissions.
SMEs have played a part in advancing the SD agenda, for example as part of coordinated supply chain or industrial park initiatives, or by participating in research and innovation in sustainable goods and services ( (Dutta et al., 2004 ) ). US DOE’s Industrial Assessment Centers (IACs) are an example of how SMEs can be provided with financial and technical support to assess and identify energy and cost-saving opportunities and training to improve human capital ( US DOE, 2003 [NPR] ).
7.8 Interaction of mitigation technologies with vulnerability and adaptation
Industry’s vulnerability to extreme weather events arises from site characteristics, for example coastal areas or flood-prone river basins (high agreement, much evidence). Because of their financial and technical resources, large industrial organizations typically have a significant adaptive capacity for addressing vulnerability to weather extremes. SMEs typically have fewer financial and technical resources and therefore less adaptive capacity. The food processing industry, which relies on agricultural resources that are vulnerable to extreme weather conditions like floods or droughts, is engaging in dialogue with its supply chain to reduce GHGs emissions. Companies are also attempting to reduce vulnerability through product diversification ( (Kolk and Pinkse, 2005 ) ).
Linkages between adaptation and mitigation in the industrial sector are limited. In areas dependent on hydropower, mitigation options that reduce industrial electricity demand will help in adapting to climate variability or change that affects water supply ( Subak et al., 2000 [JoC] ). Many mitigation options (e.g., energy efficiency, heat and power recovery, recycling) are not vulnerable to climate change and therefore create no additional adaptation link. Others, such as fuel switching can be vulnerable to climate change under certain circumstances. As the 2005 Atlantic hurricane season demonstrated, the oil and gas infrastructure is vulnerable to weather extremes. Use of solar or biomass energy will be vulnerable to both weather extremes and climate change. Adaptation, the construction of more weather resistant facilities and provision of back-up energy supplies could reduce this vulnerability.
7.9 Effectiveness of and experience with policies
As noted in the TAR ( IPCC, 2001b [NPR] ), industrial enterprises of all sizes are vulnerable to changes in government policy and consumer preferences. While the specifics of government climate policies will vary greatly, all will have one of two fundamental objectives: constraining GHG emissions or adapting to existing or projected climate change. And while consumers may become more sensitive to the GHG impacts of the products and services they use, it is almost certain that they will continue to seek the traditional qualities of low-cost, reliability, etc. The challenge to industry will be to continue to provide the goods and services on which society depends in a GHG-constrained world. Industry can respond to the potential for increased government regulation or changes in consumer preferences in two ways: by mitigating its own GHG emissions or by developing new, lower GHG emission products and services. To the extent that industry does this before required by either regulation or the market, it is demonstrating the type of anticipatory, or planned, adaptation advocated in the TAR ( IPCC, 2001b [NPR] ).
7.9.1 Kyoto mechanisms (CDM and JI)
The Clean Development Mechanism (CDM) was created under the Kyoto Protocol to allow Annex I countries to obtain GHG emission reduction credits for projects that reduced GHG emission in non-Annex I countries, provided that those projects contributed to the sustainable development of the host country ( UNFCCC, 1997 [NPR] ). As of November 2006, over 400 projects had been registered, with another 900 in some phase of the approval process. Total emission reduction potential of both approved and proposed projects is nearly 1.5 GtCO2 (410 MtC). The majority of these projects are in the energy sector; as of November 2006, only about 6% of approved CDM projects were in the industrial sector (UNFCCC, CDM, n.d.). The concept of Joint Implementation (JI), GHG-emissions reduction projects carried out jointly by Annex I countries or business from Annex I countries, is mentioned in the UNFCCC, but amplified in the Kyoto Protocol. However, since the Kyoto Protocol does not allow JI credits to be transferred before 2008, progress on JI implementation has been slow. Both CDM and JI build on experience gained in the pilot-phase Activities Implemented Jointly (AIJ) programme created by the UNFCCC in 1995 ( UNFCCC, 1995 [NPR] ). A fuller discussion of CDM, JI and AIJ appears in Section 13.3.3 .
7.9.1.1 Regional differences
Project-based mechanisms are still in their early stages of implementation, but significant differences have emerged in the ability of developing countries to take advantage of them. This is particularly true of Africa, which, as of November 2006, lagged behind other regions in their implementation. Only two of fifty AIJ projects were in Africa. None of the twenty projects recently approved under The Netherlands carbon purchase programme, CERUPT, were in Africa (CDM for Sustainable Africa, 2004 ), and only 3% of the registered CDM projects were in Africa (UNFCCC, CDM, n.d.).
Yamba and Matsika 2004 [NPR, SRC] ) identified financial, policy, technical and legal barriers inhibiting participation in the CDM in sub-Saharan Africa. Financial barriers pose the greatest challenges: low market value of carbon credits, high CDM transaction costs and lack of financial resources discourage industry participation. Policy barriers include limited awareness of the benefits of CDM and the project approval process in government and the private sector, non-ratification of the Kyoto Protocol, and failure to establish the Designated National Authorities required by CDM. Technical barriers include limited awareness of the availability of energy-saving and other appropriate technologies for potential CDM projects. Legal barriers include limited awareness in government and the private sector of the Kyoto Protocol, and the legal requirements for development of CDM projects. Limited human resources for the development of CDM projects, and CDM’s requirements on additionality are additional constraints. Other countries, for example Brazil, China and India ( Silayan, 2005 [NPR] ), have more capacity for the development of CDM projects. The Government of India ( GOI, 2004 [NPR] ) has identified energy efficiency in the steel industry as one of the priorities for Indian CDM projects.
7.9.2 Voluntary GHG programmes and agreements
7.9.2.1 Government-initiated GHG programmes and voluntary agreements
Government-initiated GHG programmes and agreements that focus on energy-efficiency improvement, reduction of energy-related GHG emissions and reduction of non-CO2 GHG emissions are found in many countries. Voluntary Agreements are defined as formal agreements that are essentially contracts between government and industry that include negotiated targets with time schedules and commitments on the part of all participating parties ( IEA, 1997 [NPR] ). Voluntary agreements for energy efficiency improvement and reduction of energy-related GHG emissions by industry have been implemented in industrialized countries since the early 1990 s. These agreements fall into three categories: completely voluntary; voluntary with the threat of future taxes or regulation if shown to be ineffective; and voluntary, but associated with an energy or carbon tax ( Price, 2005 [NPR, SRC] ). Agreements that include explicit targets, and exert pressure on industry to meet those targets, are the most effective ( UNFCCC, 2002 [NPR] ). An essential part of voluntary agreements is government support, including the programme elements such as information-sharing, energy and GHG emissions management, financial assistance, awards and recognition, standards and target-setting ( APERC, 2003 [NPR] ; CLASP, 2005 [NPR] ; Galitsky et al., 2004 [Ambiguous] ; WEC, 2004 [NPR] ). Voluntary agreements typically cover a period of five to ten years, so that strategic energy-efficiency investments can be planned and implemented. There are also voluntary agreements covering process emissions in Australia, Bahrain, Brazil, Canada, France, Germany, Japan, the Netherlands, New Zealand, Norway, the UK and the USA ( Bartos, 2001 [NPR, MoS] ; EFCTC, 2000 [NPR] ; US EPA, 1999 [NPR] ).
Independent assessments find that experience with voluntary agreements has been mixed, with some of the earlier programmes, such as the French Voluntary Agreements on CO2 Reductions and Finland’s Action Programme for Industrial Energy Conservation, appearing to have been poorly designed, failing to meet targets, or only achieving business-as-usual savings ( Bossoken, 1999 [NPR] ; Chidiak, 2000 [NPR] ;( Chidiak, 2002; ) Hansen and Larsen, 1999 [NPR] ; OECD, 2002 [NPR] ; Starzer, 2000 [NPR] ). Recently, a number of voluntary agreement programmes have been modified and strengthened, while additional countries, including some newly industrialized and developing countries, are adopting such agreements in efforts to increase the efficiency of their industrial sectors ( Price, 2005 [NPR, SRC] ). Such strengthened programmes include the French Association des Enterprises por la Réduction de l’Effet de Serre (AERES) agreements, Finland’s Agreement on the Promotion of Energy Conservation in Industry, and the German Agreement on Climate Protection ( AERES, 2005 [NPR] ; IEA, 2004 [NPR] ; RWI, 2004 [NPR] ). The more successful programmes are typically those that have either an implicit threat of future taxes or regulations, or those that work in conjunction with an energy or carbon tax, such as the Dutch Long-Term Agreements, the Danish Agreement on Industrial Energy Efficiency and the UK Climate Change Agreements (see Box 13.2 ). Such programmes can provide energy savings beyond business-as-usual ( Bjørner and Jensen, 2002 [JoC] ; Future Energy Solutions, 2004; Future Energy Solutions, 2005 ) and are cost-effective ( Phylipsen and Blok, 2002 [NPR, ARC] ). The Long-Term Agreements, for example, stimulated between 27% and 44% (17 to 28 PJ/yr) of the observed energy savings, which was a 50% increase over historical autonomous energy efficiency rates in the Netherlands prior to the agreements ( Kerssemeeckers, 2002 [NPR] ; Rietbergen et al., 2002 [ARC] ). The UK Climate Change Agreements saved 3.5 to 9.8 MtCO2 (1.0 to 2.7 MtC) over the baseline during the first target period ( 2000 – 2002 ) and 5.1 to 8.9 MtCO2 (1.4 to 2.4 MtC) during the second target period ( 2002 – 2004 ) depending upon whether the adjusted steel sector target is accounted for (Future Energy Solutions, 2005 ).
In addition to the energy and carbon savings, these agreements have important longer-term impacts ( Delmas and Terlaak, 2000 [NPR] ; Dowd et al., 2001 [NPR] ) including:
- Changing attitudes towards and awareness of energy efficiency;
- Reducing barriers to innovation and technology adoption;
- Creating market transformations to establish greater potential for sustainable energy-efficiency investments;
- Promoting positive dynamic interactions between different actors involved in technology research and development, deployment, and market development, and
- Facilitating cooperative arrangements that provide learning mechanisms within an industry.
The most effective agreements are those that set realistic targets, include sufficient government support, often as part of a larger environmental policy package, and include a real threat of increased government regulation or energy/GHG taxes if targets are not achieved ( Bjørner and Jensen, 2002 [JoC] ; Price, 2005 [NPR, SRC] ) (medium agreement, much evidence).
7.9.2.2 Company or industry-initiated voluntary actions
Many companies participate in GHG emissions reporting programmes as well as take voluntary actions to reduce energy use or GHG emissions through individual corporate programmes, non-governmental organization (NGO) programmes and industry association initiatives. Some of these companies report their GHG emission in annual environmental or sustainable development reports, or in their Corporate Annual Report. Beginning in the late 1990 s, a number of individual companies initiated in-house energy or GHG emissions management programmes and made GHG emissions reduction commitments ( Margolick and Russell, 2001 [NPR] ; PCA, 2002 [NPR] ).
Questions have been raised as to whether such initiatives, which operate outside regulatory or legal frameworks, often without standardized monitoring and reporting procedures, just delay the implementation of government-initiated programmes without delivering real emissions reductions ( OECD, 2002 [NPR] ). Early programmes appear to have produced little benefit. For example, an evaluation of the Germany industry’s self-defined global-warming declaration found that achievements in the first reporting period appeared to be equivalent to business-as-usual trends ( Jochem and Eichhammer, 1999 [NPR, ARC] ; Ramesohl and Kristof, 2001 [MoS] ). However, more recent efforts appear to have yielded positive results ( RWI, 2004 [NPR] ). Examples of targets and the actual reductions achieved include:
- DuPont’s reduction of GHG emissions by over 72% while holding energy use constant, surpassing its pledge to reduce GHG emissions by 65% by 2010 and hold energy use constant compared to a 1990 baseline (DuPont, 2002; McFarland, 2005);
- BP’s target to reduce GHG emissions by 10% in 2010 compared to a 1990 baseline which was reached in 2001 (BP, 2003; BP, 2005), and
- United Technologies Corporation’s goal to reduce energy and water consumption by 25% as a percentage of sales by the year 2007 using a 1997 baseline that was exceeded by achieving a 27% energy reduction and 34% water use reduction through 2002 (Rainey and Patilis, 2000; UTC, 2003).
Often these corporate commitments are formalized through GHG reporting programmes or registries such as the World Economic Forum Greenhouse Gas Register where 13 multinational companies disclose the amount of GHGs their worldwide operations produce ( WEF, 2005 [NPR] ) and through NGO programmes such as the Pew Center on Global Climate Change’s Business Environmental Leadership Council (Pew Center on Global Climate Change, 2005 ), the World Wildlife Fund’s Climate Savers Program (WWF, n.d.), as well as programmes of the Chicago Climate Exchange ( CCX, 2005 [NPR] ).
Industrial trade associations provide another platform for organizing and implementing GHG mitigation programmes:
- The International Aluminium Institute initiated the Aluminium for Future Generations sustainability programme in 2003, which established nine sustainable development voluntary objectives (increased to 12 in 2006), 22 performance indicators, and a programme to provide technical services to member companies (IAI, 2004). Performance to date against GHG mitigation objectives was discussed in Section 7.4.2.1.
- The World Semiconductor Council (WSC), comprised of semiconductor industry associations of the United States, Japan, Europe, Republic of Korea and Chinese Taipei, established a target of reducing PFC emissions by at least 10% below the 1995 baseline level by 2010 (Bartos, 2001).
- The World Business Council for Sustainable Development (WBCSD) started the Cement Sustainability Initiative in 1999 with ten large cement companies and it has now grown to 16 (WBCSD, 2005). The Initiative conducts research related to actions that can be undertaken by cement companies to reduce GHG emissions (Battelle Institute/WBCSD, 2002) and outlines specific member company actions (WBCSD, 2002). As of 2004, 94% of the 619 kilns of CSI member companies had developed CO2 inventories and three had established emissions reduction targets (WBCSD, 2005).
- By 2003, the Japanese chemical industry had reduced its CO2 emissions intensity by 9% compared with 1990-levels (Nippon Keidanren, 2004), but due to increased production, overall CO2 emissions were up by 10.5%.
- The European Chemical Industry Council established a Voluntary Energy Efficiency Programme (VEEP) with a commitment to improve energy efficiency by 20% between 1990 and 2005, provided that no additional energy taxes are introduced (CEFIC, 2002).
In 2003, the members of the International Iron and Steel Institute, representing 38% of global steel production, committed to voluntary reductions in energy and GHG emission intensities. In most countries this programme is too new to provide meaningful results ( IISI, 2006 [NPR, MoS] ). However, as part of a larger voluntary programme in Japan, Japanese steelmakers committed to a voluntary action programme to mitigate climate change with the goal of a 10% reduction in energy consumption in 2010 against 1990 . In fiscal year 2003, this programme resulted in a 6.4% reduction in CO2 intensity emissions against 1990, through improvement of blast furnaces, upgrade of oxygen production plants, installation of regenerative burners and other steps (Nippon Keidanren, 2004 [NPR] ).
7.9.3 Financial instruments: taxes, subsidies and access to capital
To date there is limited experience with taxing industrial GHG emissions. France instituted an eco-tax on a range of activities, including N2O emission from the production of nitric, adipic and glyoxalic acids. The tax rate is modest (37 US$ ( 2000 ) per tonne N2O, or 1.5 US$/tCO2-eq (5.5 US$/tC-eq), but it provides a supplementary incentive for emissions reductions. The UK Climate Change Levy applies to industry only and is levied on all non-household use of coal (0.15 UK pence/kWh or 0.003 US$/kWh), gas (0.15 UK pence/kWh), electricity (0.43 UK pence/kWh or 0.0085 US$/kWh) and non-transport LPG (0.07 UK pence/kWh or 0.0014 US$/kWh). Industry includes agriculture and the public sector. Fuels used for electricity generation or non-energy uses, waste-derived fuels, renewable energy, including quality CHP, which uses specified fuels and meets minimum efficiency standards, are exempt from the tax. The UK Government also provided an 80% discount from the levy for those energy-intensive sectors that agreed to challenging targets for improving their energy efficiency. Climate change agreements have now been concluded with almost all eligible sectors (UK DEFRA, 2006 [NPR] ).
In 1999, Germany introduced an eco-tax on the consumption of electricity, gasoline, fuel oil and natural gas. Revenues are recycled to subsidize the public pension system. The tax rate for electricity consumed by industrial consumers is € 0.012/kWh. Very large consumers are exempt to maintain their competitiveness. The impact of this eco-tax on CO2 emissions is still under discussion (Green Budget Germany, 2004 ).
Tax reductions are frequently used to stimulate energy savings in industry. Some examples include:
- In the Netherlands, the Energy Investment Deduction (Energie Investeringsaftrek, EIA) stimulates investments in low-energy capital equipment and renewable energy by means of tax deductions (deduction of the fiscal profit of 55% of the investment) (IEA, 2005).
- In France, investments in energy efficiency are stimulated through lease credits. In addition to financing equipment, these credits can also finance associated costs such as construction, land and transport (IEA, 2005).
- The UK’s Enhanced Capital Allowance Scheme allows businesses to write off the entire cost of energy-savings technologies specified in the ‘Energy Technology List’ during the year they make the investment (HM Revenue & Customs, n.d.).
- Australia requires companies receiving more than AU$ 3 million (US$ 2.5 million) of fuel credits to be members of its Greenhouse Challenge Plus programme (Australian Greenhouse Office, n.d.).
- Under Singapore’s Income Tax Act, companies that invest in qualifying energy-efficient equipment can write-off the capital expenditure in one year instead of three. (NEEC, 2005).
- In the Republic of Korea, a 5% income tax credit is available for energy-efficiency investments (UNESCAP, 2000).
- Romania has a programme where imported energy-efficient technologies are exempt from customs taxes and the share of company income directed for energy efficiency investments is exempt from income tax (CEEBICNet Market Research, 2004).
- In Mexico, the Ministry of Energy has linked its energy efficiency programmes with Energy Service Companies (ESCOs). These are engineering and financing specialised enterprises that provide integrated energy services with a wide range and flexibility of technologies to the industrial and service sectors (NREL, 2006).
Subsidies are used to stimulate investment in energy-saving measures by reducing investment cost. Subsidies to the industrial sector include: grants, favourable loans and fiscal incentives, such as reduced taxes on energy-efficient equipments, accelerated depreciation, tax credits and tax deductions. Many developed and developing countries have financial schemes to promote industrial energy savings. A WEC survey ( WEC, 2004 [NPR] ) showed that 28 countries, most in Europe, provide grants or subsidies for industrial energy efficiency projects. Subsides can be fixed amounts, a percentage of the investment (with a ceiling), or be proportional to the amount of energy saved. In Japan, the New Energy and Technology Development Organization (NEDO) pays up to one-third of the cost of each new high performance furnace. NEDO estimates that the project will save 5% of Japan’s final energy consumption by 2010 ( WEC, 2001 [NPR] ). The Korean Energy Management Corporation (KEMCO) provides, long-term, low interest loans to certified companies ( IEA, 2005 [NPR] ).
Evaluations show that subsidies for industry may lead to energy savings and corresponding GHG emission reductions and can create a larger market for energy efficient technologies ( De Beer et al., 2000b [NPR, ARC] ; WEC, 2001 [NPR] ). Whether the benefits to society outweigh the cost of these programmes, or whether other instruments would have been more cost-effective, has to be evaluated on a case-by-case basis. A drawback to subsidies is that they are often used by investors who would have made the investment without the incentive. Possible approaches for improving their cost-effectiveness include restricting schemes to specific target groups and/or techniques (selected list of equipment, only innovative technologies, etc.), or using a direct criterion of cost-effectiveness.
Investors in developing countries tend to have a weak capital basis. Development and finance institutions therefore often play a critical role in implementing energy efficiency and emission mitigation policies. Their role often goes beyond the provision of project finance and may directly influence technology choice and the direction of innovation ( (George and Prabhu, 2003 ) ). The retreat of national development banks in some developing countries (as a result of both financial liberalisation and financial crises in national governments) may hinder the widespread adoption of mitigation technologies because of lack of financial mechanisms to handle the associated risk.
7.9.4 Regional and national GHG emissions trading programmes
Several established or evolving national, regional or sectoral CO2 emissions trading systems exist, for example in the EU, the UK, Norway, Denmark, New South Wales (Australia), Canada and several US States. The International Emissions Trading Association ( IETA, 2005 [NPR] ) provides an overview of systems. This section focuses on issues relevant to the industrial sector. A more in-depth discussion of emission trading can be found in Section 13.2.1 .
The results of an assessment of the first two years of the UK scheme ( NERA, 2004 [NPR] ) show that reduction of non-CO2 GHG emissions from industrial sources provided the least cost options. It also found that the heterogeneity of industrial emitters may require a tiered approach for the participation of small, medium-sized and large emitters, that is in respect to monitoring and verification, and described the impacts of individual industrial emitters gaining dominating market power on allowance prices.
In January 2005, the European Union Greenhouse Gas Emission Trading Scheme (EU ETS) was launched as the world’s largest multi-country, multi-sector GHG emission trading scheme ( EC, 2005 [NPR] ). A number of assessments have analysed current and projected likely future impacts of the EU-ETS on the industrial sector in the EU ( IEA, 2005 [NPR] ; Egenhofer et al., 2005 [NPR] ). Recurring themes with specific relevance to industry include: allocation approaches based on benchmarking, grand-fathering and auctioning; electricity price increases leading to so-called ‘windfall profits’ in the utility sector; competitiveness of energy-intensive industries; specific provisions for new entrants, closures, capacity expansions, and organic growth; and compliance costs for small emitters. The further refinement of these trading systems could be informed by evidence which suggests that in some important aspects participants from industrial sectors face a significantly different situation from those in the electricity sector (Carbon Trust, 2006 [NPR] ):
- The range of products from industry sectors is generally more diverse (e.g., in the paper, glass or ceramics industry) making it difficult to define sector specific best practice values to be used for the allocation of allowances (see discussion in DTI (2005)).
- While grid connections limit electricity to regional or national markets, many industrial products are globally traded commodities, constrained only by transport costs. This increasingly applies as value per mass or volume goes up, that is from bulk ceramics products and cement, to petrochemicals, to base metals, making the impacts of trading schemes on international competitiveness a matter of varying concern for the different subsectors.
- Only a few industrial sectors (e.g., steel and refineries) are prepared to actively participate in the early phase of trading schemes, leading to reduced liquidity and higher allowance prices, suggesting that specific instruments are needed to increase industrial involvement in trading.
- Responses to carbon emission price in industry tend to be slower because of the more limited technology portfolio and absence of short term fuel switching possibilities, making predictable allocation mechanisms and stable price signals a more important issue for industry.
The EU Commission recently published its findings and recommendations based on the first year of trading under the EU-ETS ( EC, 2006a [NPR] ). An EU High Level Group on Competitiveness, Energy and the Environment has been formed to review the impacts of the EU-ETS on industry ( EU-HLG, 2006 [NPR] ). Issues highlighted in these EU processes include the need for the allocation of credits to be more harmonized across the EU, the need to increase certainty for investors, that is through long-term clarity on allocations, extension of the scheme to other sectors and alleviation of high participation costs for small installations. Industrial sectors sources considered for inclusion in the EU-ETS include CO2 emissions from ammonia production, N2O emissions from nitric and adipic acid production and PFC emission from aluminium production ( EC, 2006b [NPR] ).
7.9.5 Regulation of non-CO2 gases
The first regulations on non-CO2 GHGs are emerging in Europe. A new EU regulation (EC 842/ 2006 ) on fluorinated gases includes prohibition of the use of SF6 in magnesium die casting. The regulation contains a review clause that could lead to further use restrictions. National legislation is in place in Austria, Denmark, Luxembourg, Sweden and Switzerland that limits the use of HFCs in refrigeration equipment, foams and solvents. During the review of permits for large emitters under the EU’s Integrated Pollution Prevention and Control (IPPC) Directive (EC, 96/61) a number of facilities have been required to implement best available control technologies for N2O and fluorinated gases ( EC, 2006c [NPR] ).
Government is expected to lower financial risk and promote the investment through technology policy, which includes diverse options: budget allocations for R&D on innovative technologies, subsidy or legislation to stimulate specific environmental technologies, or regulation to suppress unsustainable technologies. See for example the US DOE’s solicitation for industrial R&D projects (US DOE, n.d.-a) and the Government of India’s Central Pollution Control Board Programmes on development and deployment of energy efficient technologies ( CPCB, 2005 [NPR] ).
7.9.6 Energy and technology policies
The IEA’s World Energy Outlook 2006 ( IEA, 2006c [NPR] ) provides an up-to-date estimate of the impacts of energy policies on the industrial sector [10] . The IEA compares two scenarios, a Reference Scenario, which assumes continuation of policies currently in place, and an Alternate Policy Scenario, which projects the cumulative impact of the more than 1400 energy policies being considered by governments worldwide, many of which affect the industrial sector. The Alternate Policy Scenario assumes faster deployment of commercially demonstrated technology, but not technologies that are still to be commercially demonstrated, including CCS and advanced biofuels.
Global industrial energy demand in 2030 in the IEA’s Alternate Policy Scenario is 9% (14 EJ) lower than in the Reference Scenario. Industrial sector CO2 emissions are 12% (0.9 GtCO2) lower. Estimated investment to achieve these savings is US$ 362 billion ( 2005 US$), US$ 195 billion of which is in electrical equipment. The savings in electricity costs are about three times the investment in electrical equipment. The IEA 2006c [NPR] ) does not provide information on the value of the fuel savings in industry, but clearly it is larger than the investment.
7.9.7 Sustainable Development policies
Appropriate sustainable development policies focusing on energy efficiency, dematerialization and use of renewables can support GHG mitigation objectives. For example, the policy options selected by the Commission on Sustainable Development 13th session to provide a supportive environment for new business formation and the development of small enterprises, included:
- Reduce information barriers for energy efficiency technology for industries;
- Build capacity for industry associations, and
- Stimulate technological innovation and change to reduce dependency on imported fuels, to improve local air pollution and to generate local employment (CSD, 2005).
Individual countries are also trying to achieve these objectives. Most policies are stated in general terms, but their implementation would have to include the industrial sector.
The EU’s strategy for sustainable development highlights addressing climate change through the reduction of energy use in all sectors and the control of non-CO2 GHGs ( EC, 2001 [NPR] ). The UK’s sustainable development policy incorporates the UK’s emissions trading and climate levy policies for the control of CO2 emissions from industry (UK DEFRA, emissions from industry (UK DEFRA,). As part of its sustainable development policy, Sweden is emphasizing energy efficiency and a long-term goal of obtaining all energy from renewable sources ( OECD, 2002 [NPR] ). China faces a significant challenge in achieving its sustainable development goals, because from 2002 to 2004 its primary energy use grew faster than its GDP, with over two-thirds of that increase coming from coal. In 2005 the Chinese government emphasized that rapid growth must be sustainable and announced the goal of reducing energy consumption per unit of GDP by 20% between 2005 and 2010 ( Naughton, 2005 [NPR] ). India has launched a series of reforms aimed at achieving industrial sector sustainable development. The 2001 Energy Conservation Act mandated a Bureau of Energy Efficiency charged with ensuring efficient use of energy and use of renewables ( GOI, 2004 [NPR] ). The Indian Industry Programme for Energy Conservation includes both mandatory and voluntary efforts, with greater emphasis on voluntary approaches ( BEE, 2006 [NPR] ).
These countries are trying to improve resources use efficiency, waste management, water and air pollution reduction, and enhance use of renewables, while providing health benefits and improved services to communities. Many developed ( Sutton, 1998 [NPR] ) and developing countries (Jindal Steel and Power, Ltd., 2006 [NPR] ; ITC, 2006 [NPR] ) encourage companies to help achieve these goals thought dematerialization, habitat restoration, recycling, and commitment to corporate social responsibility.
7.9.8 Air quality policies
Section 4.5.2 contains a more general discussion of the relationships between air quality policies and GHG mitigation. In general air quality and climate change are treated as separate issues in national and international policies, even though most practices and technologies that will reduce GHG emissions will also cause a net reduction of emissions of air pollutants. However, air pollutant reduction measures do not always reduce GHG emissions, as many require the use of additional energy (STAPPA/ALAPCO, 1999 ). Examples of policies dealing with air pollution and GHG emissions in an integrated fashion include: (1) the EU IPPC Directive (96/61/EC), which lays down a framework requiring Member States to issue operating permits for certain industrial installations, and (2) the Dutch plan for a NOx emission trading system, which will be implemented through the same legal and administrational infrastructure as the European CO2 emission trading system ( Dekkers, 2003 [NPR] ).
Mineral wastes, such as fly-ash or blast-furnace slag can have several competing alternative uses in the waste, construction and industrial sectors. The production of cement, brick and stone-wool provides energy saving uses for these materials in industry. For secondary fuels and raw materials, life-cycle assessment can help to quantify the net effects of these policies on emission across the affected parts of the economy ( Smith et al., 2001 [NPR] ). The interactions between climate policies and waste policies can be complex, sometimes leading to unexpected results because of major changes of industry practices and material flows induced by minor price differences.
7.9.9 Waste management policies
Waste management policies can reduce industrial sector GHG emissions by reducing energy use through the re-use of products (e.g., of refillable bottles) and the use of recycled materials in industrial production processes. Recycled materials significantly reduce the specific energy consumption of the production of paper, glass, steel, aluminium and magnesium. The amount, quality and price of recycled materials are largely determined by waste management policies. These policies can also influence the design of products – including the choice of materials, with its implications for production levels and emissions. Prominent examples can be found in the packaging sector, for example the use of cardboard rather than plastic for outer sales packages, or PET instead of conventional materials in the beverage industry. Vertical and horizontal integration of business provides synergies in the use of raw materials and reuse of wastes. The paper and paper boards wastes generated in cigarette packaging and printing are used as raw materials in paper and paper board units ( ITC, 2006 [NPR] ).
Another important influence of waste policies on industrial GHG emissions is their influence on the availability of secondary ‘waste’ fuels and raw materials for industrial use. For example, the ‘EU Landfill Directive’ ( EU-OJ, 1999 [NPR] ), which limits the maximum organic content of wastes acceptable for landfills, resulted in the restructuring of the European waste sector currently taking place. It makes available substantial amounts of waste containing significant biomass fractions. Typically there is competition between the different uses for these wastes: dedicated incineration in the waste sector, co-combustion in power plants, or combustion in industrial processes, for example cement kilns. In order to provide additional inexpensive disposal routes, several countries have set incentives to promote the use of various wastes in industrial processes in direct competition with dedicated incineration. Emissions trading systems or project-based mechanisms like CDM/JI can provide additional economic incentives to expand the use of secondary fuels or biomass as substitutes for fossil fuels. The impact of switching from a fossil fuel to a secondary fuel on the energy efficiency of the process itself is frequently negative, but is often compensated by energy savings in other parts of the economy.
7.10 Co-benefits of industrial GHG mitigation
The TAR explained that ‘co-benefits are the benefits from policy options implemented for various reasons at the same time, acknowledging that most policies resulting in GHG mitigation also have other, often at least equally important, rationales’ ( IPCC, 2001a [NPR] ). Significant co-benefits arise from reduction of emissions, especially local air pollutants. These are discussed in Section 11.8.1 . Here we focus on co-benefits of industrial GHG mitigation options that arise due to reduced emissions and waste (which in turn reduce environmental compliance and waste disposal costs), increased production and product quality, improved maintenance and operating costs, an improved working environment, and other benefits such as decreased liability, improved public image and worker morale, and delaying or reducing capital expenditures (see Table 7.11 ( (Pye and McKane, 2000; ) Worrell et al., 2003 [SRC] ).
Table 7.11: Co-benefits of greenhouse-gas mitigation or energy-efficiency programmes of selected countries
| Category of Co-benefit | Examples |
|---|---|
| Health | Reduced medical/hospital visits, reduced lost working days, reduced acute and chronic respiratory symptoms, reduced asthma attacks, increased life expectancy. |
| Emissions | Reduction of dust, CO, CO2, NOx and SOx; reduced environmental compliance costs. |
| Waste | Reduced use of primary materials; reduction of waste water, hazardous waste, waste materials; reduced waste disposal costs; use of waste fuels, heat and gas. |
| Production | Increased yield; improved product quality or purity; improved equipment performance and capacity utilization; reduced process cycle times; increased production reliability; increased customer satisfaction. |
| Operation and maintenance | Reduced wear on equipment; increased facility reliability; reduced need for engineering controls; lower cooling requirements; lower labour requirements. |
| Working environment | Improved lighting, temperature control and air quality; reduced noise levels; reduced need for personal protective equipment; increased worker safety. |
| Other | Decreased liability; improved public image; delayed or reduced capital expenditures; creation of additional space; improved worker morale. |
A review of forty-one industrial motor system optimization projects implemented between 1995 and 2001 found that twenty-two resulted in reduced maintenance requirements on the motor systems, fourteen showed improvements in productivity in the form of production increases or better product quality, eight reported lower emissions or reduction in purchases of products such as treatment chemicals, six projects forestalled equipment purchases, and others reported increases in production or decreases in product reject rates ( Lung et al., 2003 [NPR] ). Motor system optimization projects in China are seen as an activity that can reduce operating costs, increase system reliability and contribute to the economic viability of Chinese industrial enterprises faced with increased competition ( McKane et al., 2003 [NPR] ).
A review of 54 emerging energy-efficient technologies, produced or implemented in the USA, EU, Japan and other industrialized countries for the industrial sector, found that 20 of the technologies had environmental benefits in the areas of ‘reduction of wastes’ and ‘emissions of criteria air pollutants’. The use of such environmentally friendly technologies is often most compelling when it enables the expansion of incremental production capacity without requiring additional environmental permits. In addition, 35 of the technologies had productivity or product quality benefits ( Martin et al., 2000 [NPR, SRC] ).
Quantification of the co-benefits of industrial technologies is often done on a case-by-case basis. One evaluation identified 52 case studies from projects in the USA, the Netherlands, UK, New Zealand, Canada, Norway and Nigeria that monetized non-energy savings. These case studies had an average simple payback time of 4.2 years based on energy savings alone. Addition of the quantified co-benefits reduced the simple payback time to 1.9 years ( Worrell et al., 2003 [SRC] ). Inclusion of quantified co-benefits in an energy-conservation supply curve for the US iron and steel industry doubled the potential for cost-effective savings ( Worrell et al., 2001a [SRC] ; 2003 ).
Not all co-benefits are easily quantifiable in financial terms (e.g., increased safety or employee satisfaction), there are variations in regulatory regimes vis-à-vis specific emissions and the value of their reduction and there is a lack of time series and plant-level data on co-benefits. Also, there is a need to assess net co-benefits, as negative impacts that may be associated with some technologies, such as increased risk, increased training requirements and production losses during technology installation ( Worrell et al., 2003 [SRC] ).
7.11 Technology Research, Development, Deployment and Diffusion (RDD&D)
Most industrial processes use at least 50% more than the theoretical minimum energy requirement determined by the laws of thermodynamics, suggesting a large potential for energy-efficiency improvement and GHG emission mitigation ( IEA, 2006a [NPR] ). However, RDD&D is required to capture these potential efficiency gains and achieve significant GHG emission reductions. Studies have demonstrated that new technologies are being developed and entering the market continuously, and that new technologies offer further potential for efficiency improvement and cost reduction ( Worrell et al., 2002 [SRC] ).
While this chapter has tended to discuss technologies only in terms of their GHG emission mitigation potential and cost, it is important to realize that successful technologies must also meet a host of other performance criteria, including cost competitiveness, safety, and regulatory requirements; as well as winning consumer acceptance. (These topics are discussed in more detail in Section 7.11.2 .) While some technology is marketed as energy-efficient, other benefits may drive the development and diffusion of the technology, as evidenced by a case study of impulse drying in the paper industry, in which the driver was productivity ( Luiten and Blok, 2004 [ARC] ). This is understandable given that energy cost is just one of the drivers for technology development. Innovation and the technology transfer process are discussed in Section 2.8.2.
Technology RDD&D is carried out by both governments (public sector) and companies (private sector). Ideally, the roles of the public and private sectors will be complementary. Flannery 2001 [NPR, ARC] ) argued that it is appropriate for governments to identify the fundamental barriers to technology and find solutions that improve performance, including environmental, cost and safety performance, and perhaps customer acceptability; but that the private sector should bear the risk and capture the rewards of commercializing technology. Case studies of specific successful energy-efficient technologies, including shoe press in papermaking ( Luiten and Blok, 2003a [ARC] ) and strip casting in the steel industry ( Luiten and Blok, 2003b [ARC] ), have shown that a better understanding of the technology and the development process is essential in the design of effective government support of technology development. Government can also play an important role in cultivating ‘champions’ for technology development, and by ‘anchoring’ energy and climate as important continuous drivers for technology development ( Luiten and Blok, 2003a [ARC] ).
While GHG mitigation is not the only objective of energy R&D, IEA studies show a mismatch between R&D spending and the contribution of technologies to reduction of CO2 emissions. In its analysis of its Accelerated Technology scenarios, IEA 2006a [NPR] ) found that end-use energy efficiency, much of it in the industrial sector, contributed most to mitigation of CO2 emissions from energy use. It accounted for 39–53% of the projected reduction, except in the scenario that deemphasized these technologies. However, IEA countries spent only 17% of their public energy R&D budgets on energy-efficiency ( IEA, 2005 [NPR] ).
Many studies have indicated that the technology required to reduce GHG emissions and eventually stabilize their atmospheric concentrations is not currently available ( Jacoby, 1998 [NPR] ; Hoffert et al., 2002 [JoC, ARC] ; Edmonds et al., 2003 [NPR, ARC] ) (medium agreement, medium evidence). While these studies concentrated on energy supply options, they also indicate that significant improvements in end-use energy efficiency will be necessary. Much of the necessary research and development is being carried out in public-private partnerships, for example the US Department of Energy’s Industrial Technologies Program (US DOE, n.d.-b).
7.11.1 Public sector
A more complete discussion of public sector policies is presented in Section 7.9 and in Chapter 13 . While government use many policies to spur RDD&D in general, this section focuses specifically on programmes aimed at improving energy efficiency and reducing GHG emissions.
7.11.1.1 Domestic policies
Governments are often more willing than companies to fund higher-risk technology research and development. This willingness is articulated in the US Department of Energy’s Industrial Technologies Program role statement: ‘The programme’s primary role is to invest in high-risk, high-value research and development that will reduce industrial energy requirements while stimulating economic productivity and growth’ (US DOE, n.d.-a). The Institute for Environment and Sustainability of the EU’s Joint Research Centre has a similar mission, albeit focusing on renewable energy (Joint Research Centre, n.d.a), as does the programme of the Japanese government’s New Energy and Industrial Technology Development Organization (NEDO, n.d.).
Selection of technology is a crucial step in any technology adoption. Governments can play an important role in technology diffusion by disseminating information about new technologies and by providing an environment that encourages the implementation of energy-efficient technologies. For example, energy audit programmes, provide more targeted information than simple advertising. Audits by the US Department of Energy’s Industrial Assessment Center program in SMEs resulted in implementation of about 42% of the suggested measures ( Muller and Barnish, 1998 [NPR] ). Programmes or policies that promote or require reporting and benchmarking of energy consumption can have a similar function. These programmes have been implemented in many countries, including Canada, Denmark, Germany, the Netherlands, Norway, the UK and the USA ( Sun and Williamson, 1999 [NPR] ), and in specific industrial sectors such as the petroleum refining, ethylene and aluminium industries. (See Section 7.3.1 ).
Many of the voluntary programmes discussed in Section 7.9.2 include information exchange activities to promote technology diffusion at the national level and across sectors. For 2004, the US Industrial Technologies Program claimed cumulative energy savings of approximately 5 EJ as the result of diffusion of more than 90 technologies across the US industrial sector ( US DOE, 2006 [NPR] ). EU programmes, for example Lights of the Future and the Motor Challenge Programme (Joint Research Centre, n.d.b), have similar objectives, as do programmes in other regions.
A wide array of policies has been used and tested in the industrial sector in industrialized countries, with varying success rates ( Galitsky et al., 2004 [Ambiguous] ; WEC, 2004 [NPR] ). No single instrument will reduce all the barriers to technology diffusion; an integrated policy accounting for the characteristics of technologies, stakeholders and regions addressed is needed.
Evenson 2002 [NPR, MoS] ) suggests that the presence of a domestic research and development programme in a developing country increase the county’s ability to adapt and adopt new technologies. Preliminary analysis seems to suggest that newly industrialized countries are becoming more active in the generation of scientific and technical knowledge, although there is no accurate information on the role of technology development and investments in scientific knowledge in developing countries ( (Amsden and Mourshed, 1997 ) ).
7.11.1.2 Foreign or international policies
Industrial RDD&D programmes assume that technologies are easily adapted across regions with little innovation. This is not always the case. While many industrial facilities in developing nations are new and include the latest technology, as in industrialized countries, many older, inefficient facilities remain. The problem is exacerbated by the presence of large numbers of small-scale, much less energy-efficient plants in some developing nations; for example the iron and steel, cement and pulp and paper industries in China, and in the iron and steel industry in India ( IEA, 2006a [NPR] ). This creates a huge demand for technology transfer to developing countries to achieve energy efficiency and emissions reductions.
Internationally, there are a growing number of bilateral technology RDD&D programmes to address the slow and potentially sporadic diffusion of technology across borders. A December, 2004 US Department of State Fact Sheet lists 20 bilateral agreements with both developed and developing nations (US Dept. of State, 2004 ), many of which include RDD&D.
Multilaterally, the UNFCCC has resulted in the creation of two technology diffusion efforts, the Climate Technology Initiative (CTI) and the UNFCCC Secretariat’s TT:CLEAR technology transfer database. CTI was established in 1995 by 23 IEA/OECD member countries and the European Commission, and as of 2003 has been recognized as an IEA Implementing Agreement. Its focus is the identification of climate technology needs in developing countries and countries with economies-in-transition, and filling those needs with training, information dissemination and other support activities ( CTI, 2005 [NPR] ). TT:CLEAR is a more passive technology diffusion mechanism that depends on users accessing the database and finding the information they need ( UNFCCC, 2004 [NPR] ). Additionally, in 2001, the UNFCCC established an Expert Group on Technology Transfer (EGTT) ( UNFCCC, 2001 [NPR] ). EGTT has promoted a number of activities including workshops on enabling environments and innovative financing for technology transfer. Ultimately, the Kyoto Protocol’s CDM and JI should act as powerful tools for the diffusion of GHG mitigation technology.
IEA implementing agreements, for example the Industrial Energy Related Technology and Systems Agreement (IEA-IETS, n.d.), also provide a multilateral basis for technology transfer. While still in the planning stage, it is hoped that the newly established Asia-Pacific Partnership on Clean Development and Climate will play a key role in technology transfer to China, India and Korea (APP, n.d.)
7.11.2 Private sector
In September, 2004, the IPCC convened an expert meeting on industrial technology development, transfer and diffusion. One of the objectives of the meeting was to identify the key drivers of these processes in the private sector ( IPCC, 2005a [NPR] ). Among the key drivers for private sector involvement in the technology process discussed at the meeting were:
- Maintaining competitive advantage in open markets;
- Consumer acceptance in response to environmental stewardship;
- Country-specific characteristics: economic and political as well as its natural resource endowment;
- Scale of facilities, which affects the type of technology that can be deployed;
- Intellectual property rights (IPR): protection of IPR is critical to achieving competitive advantage through technology.
- Regulatory framework, including: government incentives; government policies on GHG emissions reduction, energy security and economic development; rule of law; and investment certainty.
The meeting concluded that each of these drivers could either be stimulants or barriers to the technology process, depending on their level, for example a high level of protection for IPR would stimulate the deployment of innovative technology in a specific country while a low level would be a barrier. However, it was also recognized that these drivers were only indicators and that actual decisions had to consider interactions between the drivers, as well as non-technology factors.
7.12 Long-term outlook, system transitions, decision-making and inertia
7.12.1 Longer-term mitigation options
Many technologies offer long-term potential for mitigating industrial GHG emissions, but interest has focused in three areas:
- Advanced biological processing, in which chemicals are produced by biological reactions that require lower energy input;
- Use of hydrogen for metal smelting, in fuel cells for electricity production, and as a fuel – provided the hydrogen is produced via a low or zero-carbon process – and;
- Nanotechnology, which could provided the basis for more efficient catalysts for chemical processing and for more effective conversion of low-temperature heat into electricity (Hillhouse and Touminen, 2001).
While some applications of these technologies could enter the marketplace by 2030, their widespread application, and impact on GHG emissions, is not expected until post- 2030 .
7.12.2 System transitions, inertia and decision-making
Given the complexity of the industrial sector, the changes required to achieve low GHG emissions cannot be characterized in terms of a single system transition. For example, development of an inert electrode for aluminium smelting would significantly lower GHG emissions from this process, but would have no impact on emissions from other industries.
Inertia in the industrial sector is characterized by capital stock turnover rate. As discussed in Section 7.6 , the capital stock in many industries has lifetimes measured in decades. While opportunities exist for retrofitting some capital stock, basic changes in technology occur only when the capital stock is installed or replaced. This inertia is often referred to as ‘technology lock-in’, a concept first proposed by Arthur 1988 [NPR, MoS] IEA 2006a [NPR] ) discusses the potential effects of technology lock-in in electric power generation, where much of the capital stock in developed nations will be replaced, and much of the capital stock in developing nations will be installed, in the next few decades. Installation of lower-cost, but less efficient technology will then impact GHG emission for decades thereafter. The same concerns and impacts apply in the industrial sector.
Industrial companies are hierarchical organizations and have well-established decision-making processes. In large companies, these processes have formal methods for incorporating technical and economic information, as well as regulatory requirements, consumer preferences and stakeholder inputs. Procedures in SMEs are often informal, but all successful enterprises have to address the same set of inputs.
7.13 Key uncertainties and gaps in knowledge
Gaps in knowledge are defined as missing information that could be developed by research. Uncertainties are missing information that cannot be developed through research. Key uncertainties in the projection of mitigation potential and cost in 2030 are:
- The rate of technology development and diffusion;
- The cost of future technology;
- Future energy and carbon prices;
- The level of industry activity in 2030; and
- Policy drivers, both climate and non-climate.
Key gaps in knowledge are: base case energy intensity for specific industries, especially in transition economies; co-benefits, SD implications of mitigation options and consumer preferences.
REFERENCES
AERES, 2005: Rapport 2005. Association des Entreprises pour la Réduction de l’Effet de Serre Nanterre, France, <http://www.aeres-asso.org/doc/28-06-06%20AERES_RA%20FR.pdf>, accessed 31/05/07. [NPR]
AF&PA, 2004: AF&PA Environmental, Health and Safety Verification Program: Year 2002 Report. American Forest and Paper Association, Washington, D.C. [NPR]
Aguayo, F. and K. Gallagher, 2005: Economic reform, energy and development: The case of Mexican manufacturing. Energy Policy, 33 (7), pp. 829-837. Clean
Amsden, A.H. and M. Mourshed, 1997: Scientific publications, patents and technological capabilities in Late-Industrializing Countries. Technology Analysis & Strategic Management, 3, pp. 343-359. Clean
Anderson, S. and R. Newell, 2004: Prospects for carbon capture and storage technologies. Annual Review of Environmental Resources, 29, pp. 109-42. Clean
ANSI, 2005: A management system for energy. ANSI/MSE 2000:2005, American National Standards Institute Washington, D.C., American National Standards Institute. [NPR]
APEC, 2002: Profiles in SMEs and SME Issues, 1990-2000. Asia-Pacific Economic Cooperation, Singapore, World Scientific Publishing. [NPR]
APERC, 2003: Energy efficiency programmes in developing and transitional APEC economies. Asia Pacific Energy Research Centre, Tokyo. [NPR]
API, 2005: 3rd API/DOE Conference on voluntary actions by the oil and gas industry to address climate change. September 29-30, 2004, American Petroleum Institute Arlington, Virginia. <www.api.org/ehs/climate/response/upload/3rd_ConferenceSynopsis.pdf>, accessed 31/05/07. [NPR]
APP, n.d.: Asia pacific partnership on clean development and climate. Asia Pacific Partnership, <www.asiapacificpartnership.org>, accessed 01/06/07. [NPR]
A3P, 2006: Performance, people and prosperity: A3P Sustainability Action Plan. Australian Plantation Products and Paper Industry Council, <http://www.a3p.asn.au/assets/pdf/PerformancePeopleProsperity_actionplan.pdf>, accessed 01/06/07. [NPR]
AREED, 2000: Open for business. African Rural Energy Enterprise Development, <http://www.areed.org/status/Open_for_Business.pdf>, accessed 01/06/07. [NPR]
Arthur, B.: 1988: Self-reinforcing mechanisms in economics. In The Economy as an Evolving Complex System. P.W. Anderson and K.J. Arrow (eds.), New York: Addison-Wesley. [NPR, MoS]
Aunan, K., J. Fang, H. Vennemo, K. Oye, and H.M. Seop, 2004: Co-benefits of climate policy: Lessons learned from a study in Shanxi, China. Energy Policy, 32, pp. 567-581. Clean
Australian Greenhouse Office, n.d.: Fuel tax credits and greenhouse challenge plus membership. <www.greenhouse.gov.au/challenge/members/fueltaxcredits.html>, accessed 31/05/07. [NPR]
Bailey, O. and E. Worrell, 2005: Clean energy technologies, a preliminary inventory of the potential for electricity generation. Lawrence Berkeley National Laboratory, Berkeley, California (LBNL-57451). [NPR, SRC]
Bain, 2005: An analysis of energy consumption in Bangladesh. PhD. Thesis. Jadavpur University, Kolkata, India. [NPR]
Bakaya-Kyahurwa, E., 2004: Energy efficiency and energy awareness in Botswana; ESI, Power Journal of Africa, Issue 2, 2004, pp. 36-40. Clean
Balasubramaniam, S.V., 2005: Sugar: New dimensions of growth. In Survey of Indian Industry, 2005. The Hindu, Chennai, India. [NPR]
Banda, A., 2002: Electricity production from sugar industries in Africa: A case study of South Africa. M.Sc thesis, University of Cape Town, South Africa. [NPR]
Barats, C., 2005: Managing greenhouse gas risk: A roadmap for moving forward. Presented at Risk and Insurance Management Society Annual Meeting. Philadelphia, PA, April 20, 2005. [NPR]
Bartos, S.C., 2001: Semiconductor industry leadership in global climate protection. Future Fab International 10, <www.future-fab.com/documents.asp?d_ID=1144>, accessed 31/05/07. [NPR, MoS]
Battelle Institute/WBCSD, 2002: Toward a sustainable cement industry. World Business Council on Sustainable Development, <www.wbcsd.org/web/publications/batelle-full.pdf>, accessed 31/05/07. [NPR]
BEE, 2003: Energy management policy - Guidelines for energy-intensive industry of India. Bureau of Energy Efficiency Delhi, Government of India. Ministry of Power. [NPR]
BEE, 2006: Implementation: Case studies from industries. Bureau of Energy Efficiency, <www.bee-india.nic.in/Implementation/Designated%20Consumers.htm>, accessed 31/05/07. [NPR]
Beerkens, R.G.C. and J. van Limpt, 2001: Energy efficient benchmarking of glass furnaces; 62nd conference on glass problems. University of Illinois, October 16-17, 2001. [NPR]
Bergmeier, M., 2003: The history of waste energy recovery in Germany since 1920. Energy, 28, pp. 1359-1374. Clean
Berntsson, T., P-A Franke, and A. Åsblad, 1997. Process heating in the low and medium temperature range. IEA/Caddet, Sittard, The Netherlands. [NPR]
Birat, J.P., 2005: The UCLOS Program (Ultra Low CO2 Steelmaking). <http://cordis.europa.eu/estep/events-infostp_en.html>, accessed 31/05/07. [NPR]
Bjørner, T.B. and H.H. Jensen, 2002: Energy taxes, voluntary agreements and investment subsidies - A micro panel analysis of the effect on Danish industrial companies’ energy demand. Resource and Energy Economics, 24, pp. 229-249. [JoC]
Bossoken, E., 1999: Case study: A comparison between France and The Netherlands of the Voluntary Agreements Policy. Study carried out in the frame of the MURE project, financed by the SAVE programme, European Commission, Directorate General for Energy (DG 17). [NPR]
Boyd, G.A., 2005. A method for measuring the efficiency gap between average and best practice energy use: The ENERGY STAR Industrial Performance Indicator. Journal of Industrial Ecology, 3, pp. 51-66. Clean
BP, 2003: Defining our path: Sustainability Report 2003. London: BP. <www.bp.com/liveassets/bp_internet/globalbp/STAGING/global_assets/downloads/B/BP_Sustainability_Report_2003.pdf>, accessed 31/05/07. [NPR]
BP, 2005: Making energy more: Sustainability Report 2005. London: BP. <www.bp.com/liveassets/bp_internet/globalbp/STAGING/global_assets/downloads/S/bp_sustainability_report_2.pdf>, accessed 31/05/07. [NPR]
Bystrom, S. and L. Lonnstedt, 1997: Paper recycling, environmental and economic impacts. Resource Conservation and Recycling, 21, pp. 109-127. Clean
Caffal, C., 1995. Energy management in industry. Centre for the Analysis and Dissemination of Demonstrated Energy Technologies (CADDET). Analysis Series 17. Sittard, The Netherlands. [NPR]
Canepa, A. and P. Stoneman, 2004: Financing constraints in the inter firm diffusion of new process technologies. Journal of Technology Transfer, 30, pp. 159-169. Clean
Carbon Trust, 2006: Allocation and competitiveness in the EU emissions trading scheme: Options for Phase II and beyond. London, June, 2006. <www.carbontrust.co.uk/climatechange/policy/allocation_competitiveness.htm>, accessed 31/05/07. [NPR]
CCX, 2005: Members of the Chicago Climate Exchange ® (CCX®). <http://www.chicagoclimatex.com/about/members.html>, accessed 31/05/07. [NPR]
CDM for Sustainable Africa, 2004: Synergy: CDM for sustainable Africa - capacity building for Clean Development Mechanisms in Sub-Saharan Africa - final project report. <http://www.rgesd-sustcomm.org/CDM_AFRICA/PDF/Final_Report-CDM_for_Sustainable_Africa_Total.pdf >, accessed 31/05/07. [NPR]
CEEBICNet Market Research, 2004: Energy Efficiency in Romania. <www.mac.doc.gov/ceebic/country/Romania/EnergyEfficiency.htm>, accessed 31/05/07. [NPR]
CEEEZ, 2003: Multi-energy efficiency through use of automatic load control for selected industries. Centre for Energy, Environment and Engineering, Zambia Ltd.Project Idea Note (PIN) submitted to 500 ppm, Frankfurt, Germany. [NPR]
CEFIC, 2002: Managing energy: Voluntary energy efficiency program (VEEP). <http://www.cefic.org/Templates/shwStory.asp?NID=471&HID=149>, accessed 31/05/07. [NPR]
CEPI, 2001: Potential energy efficiency improvements for the European Pulp and Paper Industry, Confederation of European Paper Industries, Brussels, March 2001. [NPR]
CESP, 2004: China’s sustainable energy scenarios in 2020. China Environmental Science Press. Beijing. [NPR]
CFI, 2003: Energy benchmarking: Canadian potash production facilities. Canadian Fertilizer Institute, Ottawa, Canada, Canadian Industry Program for Energy Conservation. [NPR]
Chang, H.J., 2003: New horizons for Korean energy industry - shifting paradigms and challenges ahead. Energy Policy, 31, pp. 1073-1084. Clean
Chaudhary, T.R., 2001: Technological measures of improving productivity: Opportunities and constraints. Presented at the Fertilizer Association of India Seminar “Fertiliser and Agriculture Future Directions,” New Delhi, India, 6-8 December 2001. [NPR, MoS]
Chaudhuri, S. and K. Gupta, 2003: Inflow of foreign capital, environmental pollution, and welfare in the presence of an informal sector. Dept. of Economics, University of Calcutta, Kolkata, India. [NPR]
Chevron, 2005: Corporate Responsibility Report 2005. <www.chevron.com/cr_report/2005/downloads.asp>, accessed 31/05/07. [NPR]
Chidiak, M., 2000: Voluntary agreements - Implementation and efficiency. The French Country Study: Case Studies in the Sectors of Packaging Glass and Aluminium. Paris: CERNA, Centre d’économie industrielle, Ecole Nationale Supérieure des Mines de Paris. [NPR]
Chidiak, M., 2002: Lessons from the French experience with voluntary agreements for greenhouse gas reduction. Journal of Cleaner Production, 10, pp. 121-128. Clean
Choi, K.H. and B.W. Ang, 2001: A time-series analysis of energy-related carbon emissions in Korea. Energy Policy, 29, pp. 1155-1161. Clean
CIEEDAC, 2004: Development of energy intensity indicators for Canadian industry 1990-2002. Canadian Industrial Energy End-Use Data and Analysis Center Burnaby, BC, Canada. [NPR]
CLASP, 2005: S&L Worldwide Summary. Collaborative Labelling and Appliance Standards Program, <www.clasponline.org/main.php>, accessed 31/05/07. [NPR]
Cohen, A.T, 2001: Prospects for biogas harvesting at Sungunn Wongse Industries (Thailand). Report of a feasibility and technical evaluation. [NPR]
CONCAWE, 2005: The impact of reducing sulphur to 10 ppm max in European automotive fuels: An Update. Report 8/05. Conservation of Clean Air and Water Europe (CON-CAWE), Brussels, 53 pp. [NPR]
Continental Engineering BV, 2001: High temperature catalytic reduction of nitrous oxide emissions from nitric acid production plants. Report funded by Dutch Ministry of Housing Spatial Planning and Environment, SenterNovem Project No. 375001/0080, CE Ref. J-4586-02. [NPR]
Cornish, A.T. and B. Kerkhoff, 2004: European standards and specifications in cements. In Innovations in Portland Cement Manufacture. J.I. Bhatty, F.M. Miller and S.H. Kosmatka (eds.), Portland Cement Association, Skokie, IL, USA, pp. 1209-1218. [NPR]
Cornland, D., F. Johnson, F.D. Yamba, E.W. Chidumayo, M.N. Morales, O. Kalumiana, and S.B. Mtonga-Chidumayo, 2001: Sugar cane resources for sustainable development, Stockholm Environment Institute: ISBN: 91 88714713. [NPR, SRC]
Cordi, A. and L. Lombardi, 2004: End life tyres: Alternative final disposal process compared by LCA. Energy, 29, pp. 2089-2108. Clean
CPCB, 2005: Annual Action Plan 2005-2006. Central Pollution Control Board of India <www.cpcb.nic.in/actionplan/plan2005-ch3.htm>, accessed 31/05/07. [NPR]
CRA, 2002: Information on corn wet milling. Corn Refiners Association <www.corn.org/whatiscornrefining.htm>, accessed 31/05/07. [NPR]
CSD, 2005: Report on the thirteenth session (30 April 2004 and 11-22 April 2005). Commission on Sustainable Development UN ECOSOC, Official Records, 2005, Supplement No. 9. [NPR]
CTI, 2005: Climate Technology Initiative. <www.climatetech.net>, accessed 31/05/07. [NPR]
Dankers, A.P.H., 1995: The energy situation in Thai lime industries. Energy for Sustainable Development, 1, pp. 36-40. Clean
Das, T.K. and J. Roy, 1994: Cost benefit analysis of the solar water heating system in Khardah Jute Mill and solar cookers under operation in various parts of Calcutta. Report submitted to West Bengal Renewable Energy Development Agency. [NPR, SRC]
Dasgupta, M and J. Roy, 2000: Manufacturing energy use in India: A decomposition analysis. Asian Journal of Energy and Environment, 1, pp. 223-247. Clean
Dasgupta, M. and J. Roy, 2001: Estimation and analysis of carbon dioxide emissions from energy intensive manufacturing industries in India. International Journal of Energy, Environment and Economics, 11, pp. 165-179. [MoS, SRC]
De Beer, J.G., 1998: Potential for industrial energy-efficiency improvement in the long term. PhD Dissertation, Utrecht University, Utrecht, The Netherlands. [NPR]
De Beer, J.G., E. Worrell, and K. Blok, 1998: Future technologies for energy efficient iron and steelmaking. Annual Review of Energy and Environment, 23, pp. 123-205. [MoS, SRC]
De Beer, J.G., J. Harnisch and M. Kerssemeeckers, 2000a: Greenhouse gas emissions from iron and steel production. IEA Greenhouse Gas R&D Programme, Cheltenham, UK. [NPR, SRC]
De Beer, J.G., M.M.M. Kerssemeeckers, R.F.T. Aalbers, H.R.J. Vollebergh, J. Ossokina, H.L.F de Groot, P. Mulder, and K. Blok, 2000b: Effectiveness of energy subsidies: Research into the effectiveness of energy subsidies and fiscal facilities for the period 1988-1999. Ecofys, Utrecht, the Netherlands. [NPR, ARC]
De Beer, J.G., D. Phylipsen, and J. Bates, 2001: Economic evaluation of carbon dioxide and nitrous oxide emission reductions in industry in the EU. European Commission - DG Environment, Brussels, Belgium. [NPR]
De Keulenaer, H., R. Belmans, E. Blaustein, D. Chapman, A. De Almeid, and B. De Wachter, 2004: Energy efficient motor driven systems, <http://re.jrc.ec.europa.eu/energyefficiency/pdf/HEM_lo_all%20final.pdf>, accessed 01/06/07. [NPR]
Dekkers, C., 2003: Netherlands to launch legislation on NOx and CO2 emissions trading. Emissions Trader - Emissions Marketing Association, <www.emissions.org/publications/emissions_trader/0303>, accessed 31/05/07. [NPR]
Delmas, M. and A. Terlaak, 2000: Voluntary agreements for the environment: Innovation and transaction costs. CAVA Working Paper 00/02/13 February. [NPR]
Dersden, A., P. Vercaenst and P. Pijkmans 2002: Best available techniques (BAT) for the fruit and vegetable processing industry. Resources, Conservation and Recycling, 34, pp. 261-271. Clean
Dowd, J., K. Friedman, and G. Boyd, 2001: How well do voluntary agreements and programs perform at improving industrial energy efficiency. Proceedings of the 2001 ACEEE Summer Study on Energy Efficiency in Industry. Washington, D.C.: American Council for an Energy-Efficient Economy. [NPR]
DTI, 2005: EU Emissions trading scheme: Benchmark Research for Phase 2. Department of Trade and Industry (UK), Prepared by Entec UK Limited, and NERA Economic Consulting on behalf of the UK Department of Transport and Industry, July 2005. [NPR]
DuPont, 2002: Sustainable growth: 2002 Progress Report. Wilmington, DC: DuPont. <www1.dupont.com/dupontglobal/corp/documents/US/en_US/news/publications/dupprogress/2002USprogrept.pdf>, accessed 31/05/07. [NPR]
Dutta, S., P.K. Datta, S. Ghosal, A. Gosh and S.K. Sanyal, 2004: Low-cost pollution control devices for continuous cupolas. Indian Foundry Journal, 50, pp. 29-33. Clean
EC, 2001: A sustainable europe for a better world: A European Union strategy for sustainable development. European Commission, <http://europa.eu.int/eur-lex/en/com/cnc/2001/com2001_0264en01.pdf>, accessed 01/06/07. [NPR]
EC, 2004: Comprehensive Report 2002-2003 regarding the role of forest products for climate change mitigation. Enterprise DG Unit E.4, Forest-based industries. Brussels: European Commission. [NPR]
EC, 2005: The European Union Greenhouse Gas Emission Trading Scheme. European Commission, <http://europa.eu.int/comm/environment/climat/pdf/emission_trading2_en.pdf>, accessed 31/05/07. [NPR]
EC, 2006a: A report to the European Parliament and the Council considering the functioning of the EU Emissions Trading Scheme (ETS). European Commission, Brussels, 2006, <http://ec.europa.eu/environment/climat/emission/review_en.htm>, accessed 31/05/07. [NPR]
EC, 2006b: Inclusion of additional activities and gases into the EU - Emissions Trading Scheme, European Commission DG Environment, <http://ec.europa.eu/environment/climat/emission/review_en.htm>, accessed 01/06/07. [NPR]
EC, 2006c: Installations with reported emissions of more than 10% of EU wide total emissions according to EPER, European Commission, DG Environment, January 2006. <http://ec.europa.eu/environment/ippc/pdf/table_largest_emitters_jan_06.pdf>, accessed 31/05/07. [NPR]
EDGAR, 2005: EDGAR 2005 Fast Track Data Set. MNP - Netherlands Environmental Assessment Agency (MNP), <www.mnp.nl/edgar/model/v32ft2000edgar/>, accessed 31/05/07. [NPR, MoS]
Edmonds, J., J.J. Dooley, S.H. Kim, R. Cesar Izaurralde, N.J. Rosenberg, and G.M. Stokes, 2003: The potential role of biotechnology in addressing the long-term problem of climate change in the context of global energy and economic systems. In Greenhouse Gas Control Technologies: Proceedings of the Sixth International Conference on Greenhouse Gas Control Technologies, Kyoto, Japan, Elsevier Science, Oxford, UK, J. Gale and Y. Kaya (eds.), pp. 1427-1433, ISBN 0080442765. [NPR, ARC]
EFCTC, 2000: The United Nations Framework Convention on Climate Change - Fluorochemicals and the Kyoto Protocol. European Fluorocarbon Technical Committee, <www.cefic.be/sector/efctc/kyoto2000-02.htm>, accessed 31/05/07. [NPR]
Egenhofer, C., N. Fujiwara, and K. Giaoglou, 2005: Business consequences of the EU Emissions Trading Scheme. Brussels, Centre for European Policy Studies, February, 2005. [NPR]
EIA, 2002: Manufacturing consumption of energy 1998. Energy Information Agency Office of Energy Markets and End Use, Washington, D.C. [NPR]
EIA, 2005: Annual Energy Outlook 2005: With projections to 2025. Energy Information Agency, Washington, D.C., <www.eia.doe.gov/oiaf/aeo/pdf/0383(2005).pdf>, accessed 31/05/07. [NPR, MoS]
Eidt, B., 2004: Cogeneration opportunities - Global Energy Management System. Presented at the 3rd API/DOE Conference on Voluntary Actions by the Oil and Gas Industry to Address Climate Change, September 29-30, 2004, Arlington, Virginia, <www.api.org/ehs/climate/response/upload/3rd_ConferenceSynopsis.pdf>, accessed 31/05/07. [NPR]
Einstein, D., E. Worrell, and M. Khrushch, 2001: Steam systems in industry: Energy use and energy efficiency improvement potentials. Proceedings of the 2001 ACEEE Summer Study on Energy Efficiency in Industry - Volume 1, Tarrytown, NY, July 24-27th, 2001, pp. 535-548. [SRC]
ENDS-Directory, 2006: Getting help through environmental business support schemes. Judith Cowan, Business in the Environment, <http://www.endsdirectory.com/index.cfm?action=articles.view& articleID=bie>, accessed 31/05/07. [NPR]
EU-HLG, 2006: High Level group (HLG) on competitiveness, energy and the environment. <http://ec.europa.eu/enterprise/environment/hlg/hlg_en.htm>, accessed 31/05/07. [NPR]
EU-BREF Ceramics, 2005: Integrated pollution prevention and control (IPPC) reference document on best available technology in the ceramic manufacturing industry. <ftp://ftp.jrc.es/pub/eippcb/doc/cer_bref_1206.pdf>, accessed 31/05/07. [NPR]
EU-BREF Glass, 2001: Integrated Pollution Prevention and Control (IPPC) Reference Document on Best Available Technology in the Glass Manufacturing Industry. <ftp://ftp.jrc.es/pub/eippcb/doc/gls_bref_1201.pdf>, accessed 31/05/07. [NPR]
EU-OJ, 1999: Council Directive 1999/31/EC of 26 April 1999 on the landfill of waste. Official Journal L 182, 16/07/1999, Pages 0001-0019. [NPR]
Evenson, R.E., 2002: Induced adaptive invention/innovation convergence in developing countries. In Technological Change and the Environment, A. Grubler, N. Nackicenovic and W.D. Nordhaus (eds.), Resources for the Future, Washington, D.C. [NPR, MoS]
FAO, 1993: Status and development issues of the brick industry in Asia. Regional Wood Energy Development Programme in Asia, FAO: GCP/RAS/154/NET, Bangkok. [NPR]
FAO, 2006: Statistical database. <http://faostat.fao.org/default.aspx>, accessed 31/05/07. [NPR]
FAOSTAT, 2006: Forestry data. <http://faostat.fao.org/site/381/default.aspx>, accessed 31/05/07. [NPR]
Farahani, S., E. Worrell, and G. Bryntse, 2004: CO2-free paper? Resources and Conservation Recycling, 42, pp. 317-336. [SRC]
Farla, J., K. Blok, and L. Schipper, 1997: Energy efficiency developments in the paper and pulp industry. Energy Policy, 25, pp. 745-758. [ARC]
Flannery, B.P., 2001: An industry perspective on carbon management. Presented at Carbon Management: Implications for R&D in the Chemical Sciences and Technology, A Workshop Report to the Chemical Sciences Roundtable, (US) National Research Council. [NPR, ARC]
FPAC, n.d.: Environmental stewardship. Forest Products Association of Canada, <www.fpac.ca/en/sustainability/stewardship>, accessed 31/05/07. [NPR]
Future Energy Solutions, 2004: Climate Change Agreements: Results of the Second Target Period Assessment, Version 1.0., Didcot, UK: AEA Technology. www.defra.gov.uk/environment/ccl/pdf/cca_jul05.pdf [NPR]
Future Energy Solutions, 2005: Climate Change Agreements: Results of the second target period assessment, Version 1.2. Didcot, UK: AEA Technology. <www.defra.gov.uk/environment/ccl/pdf/cca_aug04.pdf>, accessed 31/05/07. [NPR]
Galitsky, C., N. Martin, and E. Worrell, 2001: Energy efficiency opportunities for United States breweries. MBAA Technical Quarterly, 4, pp. 189-198. [SRC]
Galitsky, C., E. Worrell, and M. Ruth, 2003: Energy efficiency improvement and cost saving opportunities for the corn wet milling industry: An ENERGY STAR Guide for Energy and Plant Manager. Berkeley, CA, Lawrence Berkeley National Laboratory (LBNL-52307). [NPR, SRC]
Galitsky, C., L. Price, and E. Worrell, 2004: Energy efficiency programs and policies in the industrial sector in industrialized countries. Berkeley, CA, Lawrence Berkeley National Laboratories (LBNL-54068). [NPR, SRC]
Galitsky, C. and E. Worrell, 2004: Profile of the chemical industry in California. Berkeley, CA, Lawrence Berkeley National Laboratory (LBNL-55668). [NPR, SRC]
Gartner, E., 2004: Industrially interesting approaches to “low-CO2” cements. Cement and Concrete Research, 34, pp. 1489-98. Clean
Giehlen, D.J., 2006: Energy efficiency and CO2 emissions reduction: Opportunities for the fertiliser industry. Paper presented at the 2006 IFA Technical Symposium. Vilnius, Lithuania, 25-28 April 2006. [NPR]
George, G. and G.N. Prabhu, 2003: Developmental financial institutions as technology policy instruments: Implications for innovation and entrepreneurship in emerging economies. Research Policy, 32, pp. 89-108. Clean
GOI, 2004: India’s initial national communication to the UN Framework Convention on Climate Change. Government of India Ministry of the Environment and Forest, New Delhi. [NPR]
GOI, 2005: Annual Report, 2004-2005 of the Ministry of Environment and Forest. Government of India, New Delhi. [NPR]
GOI, Various issues: Annual survey of industries. Central Statistical Organization, Government of India, Kolkata, India. [NPR]
Goulder, L.H. and S.H. Schneider, 1999: Induced technological change and the attractiveness of CO2 abatement policies. Resource and Energy Economics, 21, pp. 211-253. [PoC, JoC]
Government of Flanders, 2005: Energiebenchmarking in Vlaanderen. <www.benchmarking.be/en/home.html>, accessed 31/05/07. [NPR]
Green Budget Germany, 2004: Ecotax - Memorandum 2004. Förderverein Ökologische Steuerreform e.V., <www.czp.cuni.cz/ekoreforma/EDR/GBG_Memorandum_2004.pdf>, accessed 01/06/07. [NPR]
GRI, n.d.: A common framework for sustainability reporting. <www.globalreporting.org>, accessed 31/05/07. [NPR]
Gross, D., A. Gross, M. Schoenfeld, W. Simmons and B. Patrick, 2003: Jupiter oxygen combustion technology of coal and other fossil fuels in boilers and furnaces. Presented at the 28th International Technical Conference on Coal Utilization and Fuel Systems, Clearwater, FL, USA. [NPR]
Grothcurth, H.M., R. Kummel, and W.V. Goole, 1989: thermodynamic limits to energy optimization. Energy, 14, pp. 241-258. Clean
Gudbjerg, E., 2005: Energy Management: The Danish standard. Presented at the First European Energy Management Conference, Milan, Italy, 23-24 November 2005. [NPR]
Gupta, K., 2002: The urban informal sector and environmental pollution: A theoretical analysis. Working Paper No. 02-006. Center for Environment and Development Economics, University of York, York, UK. [NPR]
Hansen, K. and A. Larsen, 1999. Energy efficiency in industry through voluntary agreements - the implementation of five voluntary agreement schemes and an assessment. Copenhagen: AKF, Amternes og Kommunernes Forskningsinstitut. [NPR]
Harnisch, J., I. Sue Wing, H.D. Jacoby, and R.G. Prinn, 1998: Primary aluminium production: Climate policy, emissions and costs. MIT Joint Program on the Science and Policy of Global Change Report Series, Report No. 44, Massachusetts Institute of Technology, Cambridge, MA. [NPR, SRC]
Harnisch, J. and W. Schwarz, 2003: Costs and the impact on emissions of potential regulatory framework for reducing emissions of hydrofluorocarbons, perfluorocarbons and sulphur hexafluoride. On behalf of DG Environment of the European Commission, Brussels. <http://europa.eu.int/comm/environment/climat/pdf/ecofys_oekorecherchestudy.pdf>, accessed 31/05/07. [NPR, SRC]
Heeres, R.R.,et al., 2004: Eco-industrial park initiatives in the USA and the Netherlands: first lessons. Journal of Cleaner Production, 12(8-10), pp. 985-995. Clean
Heidelberg Cement, 2006: Building on sustainability, group sustainability report, 2004/2005. <www.heidelbergcement.com/global/en/company/sustainability/_channelposting.htm>, accessed 31/05/07. [NPR]
Hendriks, C.A., E. Worrell, L. Price, N. Martin, and L. Ozawa Meida, 1999: The reduction of greenhouse gas emissions from the cement industry. Cheltenham, UK, IEA Greenhouse Gas R&D Programme, Report PH3/7. [NPR, SRC]
Hidalgo, I., L. Szabo, J.C. Ciscar, and A. Soria, 2005: Technological prospects and CO2 emissions trading analysis in the iron and steel industry: a global model. Energy, 30, pp. 583-610. [MoS]
Hillhouse, H.W. and M.T. Touminen, 2001: Modelling the thermoelectric transport properties of nanowires embedded in oriented microporous and mesoporous films. Microporous and Mesoporous Materials, 47, pp. 39-50. [MoS]
HM Revenue & Customs, n.d.: ECA - 100% Enhanced capital allowances for energy-saving investments. <http://www.hmrc.gov.uk/capital_allowances/eca-guidance.htm>, accessed 01/06/07. [NPR]
Hoffert, M.I., K. Caldeira, G. Benford, C. Green, H. Herzog, A.K. Jain, H.S. Kheshgi, K.S. Lackner, J.S. Lewis, H.D. Lightfoot, W. Manheimer, J.C. Mankins, M.E. Mauel, L.J. Perkins, M.E. Schlesinger, T. Volk, and T.M.L. Wigley, 2002: Advanced Technology Paths to Global Climate Stability. Science, 298, pp. 981-987. [JoC, ARC]
Holliday, C., 2001: Sustainable growth, the DuPont Way. Harvard Business Review, September, 2001. Electronic Download R0108J. [NPR]
Houillon, G. and O. Jolliet, 2005: Life cycle assessment of processes for the treatment of wastewater urban sludge: Energy and global warming analysis. Journal of Cleaner Production. 13, pp. 287-99. Clean
Humphreys, K. and M. Mahasenan, 2002: Towards a sustainable cement industry - Substudy 8: Climate Change. World Business Council for Sustainable Development (WBCSD), Geneva, Switzerland. [NPR]
IAI, 2000: Life cycle results. International Aluminium Institute, <www.world-aluminium.org/environment/climate/lifecycle2.html>, accessed 31/05/07. [NPR]
IAI, 2001: Perfluorocarbon emissions reduction programme 1990-2000. International Aluminium Institute, <www.world-aluminium.org/iai/publications/documents/pfc2000.pdf>, accessed 31/05/07. [NPR]
IAI, 2003. Life cycle assessment of aluminium: Inventory data for the worldwide primary aluminium industry. International Aluminium Institute, London, 52 pp. [NPR]
IAI, 2004: The global sustainable development initiative. International Aluminium Institute London, 6 pp. [NPR]
IAI, 2006a: The International Aluminium Institute’s report on the Aluminium Industry’s global perfluorocarbon gas emissions reduction programme - Results of the 2004 anode effect survey. <www.world-aluminium.org/iai/publications/documents/pfc2004.pdf>, accessed 31/05/07. [NPR]
IAI, 2006b: Aluminium for future generations - sustainability update 2005. International Aluminium Institute, <www.world-aluminium.org/iai/publications/documents/update_2005.pdf>, accessed 31/05/07. [NPR, MoS]
IEA, 1997: Voluntary actions for energy-related CO2 abatement. OECD/International Energy Agency, Paris. [NPR]
IEA, 2003a: Energy balances of Non-OECD Countries: 2000-2001. Paris: IEA. [NPR]
IEA, 2003b: Energy Balances of OECD Countries: 2000-2001. International Energy Agency, Paris. [NPR]
IEA, 2004: Energy Policies of IEA Countries: Finland 2003 review. International Energy Agency, Paris [NPR]
IEA, 2005: R&D Database. International Energy Agency, <www.iea.org/dbtw-wpd/textbase/stats/rd.asp>, accessed 31/05/07. [NPR]
IEA, 2006a: Energy Technology Perspectives 2006: Scenarios and strategies to 2050. International Energy Agency, Paris, 484 pp. [NPR]
IEA, 2006b: Industrial motor systems energy efficiency: Towards a plan of action. International Energy Agency, Paris, 14 pp. [NPR]
IEA, 2006c: World Energy Outlook 2006. International Energy Agency, Paris, 596 pp. [NPR]
IEA GHG, 2000: Greenhouse gases from major industrial sources - IV. The aluminium industry; International Energy Agency Greenhouse Gas R&D Programme, Report Number PH3/23, ICF Kaiser, Cheltenham, UK, 88 pp. [NPR]
IEA GHG, 2001: Abatement of emissions of other greenhouse gases: Engineered Chemicals. IEA Greenhouse Gas R&D Programme, J. Harnisch, O. Stobbe, D. de Jager, Cheltenham, 83 pp. [NPR]
IEA-IETS, n.d.: Implementing Agreement. International Energy Agency - Industrial Energy-Related Technology and Systems, <www.iea-iets.org>, accessed 31/05/07. [NPR]
IEA-SolarPACES, n.d.: Task IV: SHIP - Solar heat for industrial processes. International Energy Agency - Concentrating Solar Power, <www.solarpaces.org/task_iv.htm>, accessed 31/05/07. [NPR]
IETA, 2005: Greenhouse Gas Market 2005: The rubber hits the road. International Emissions Trading Association, <www.ieta.org/ieta/www/pages/getfile.php?docID=1270>, accessed 31/05/07. [NPR]
IFA, 2004: IFADATA 2004. International Fertilizer Industry Association, <www.fertilizer.org/ifa/statistics/indicators/pocket_production.asp>, accessed 31/05/07. [NPR]
IFA, 2005: Summary report - World agriculture and fertilizer demand, global fertilizer supply and trade. International Fertilizer Industry Association 2005-2006. <www.fertilizer.org/ifa/publicat/PDF/2005_council_sevilla_ifa_summary.pdf>, accessed 31/05/07. [NPR]
IGEN/BEE, n.d.: Success stories. Indo-German Energy Program in cooperation with Bureau of Energy Efficiency under Ministry of Power, <www.energymanagertraining.com/new_success.php>, accessed 31/05/07. [NPR]
IISI, 2005: World steel in figures, 2005: International Iron and Steel Institute (IISI), Brussels. [NPR]
IISI, 2006: Steel: The foundation of a sustainable future - Sustainability report of the world steel industry 2005. International Iron and Steel Institute. <www.worldsteel.org/pictures/publicationfiles/SR2005.pdf>, accessed 31/05/07. [NPR, MoS]
IPCC, 1995: Climate Change 1995: The Science of Climate Change. Intergovernmental Panel on Climate Change (IPCC), Cambridge University Press, Cambridge, UK, 572 pp. [NPR]
IPCC, 2000a: Development, Equity and Sustainability. Cross Cutting Issues Guidance Papers. Intergovernmental Panel on Climate Change (IPCC), Geneva, pp. 69-113. [NPR]
IPCC, 2000b: Emission Scenarios. Special Report of the Intergovernmental Panel on Climate Change (IPCC) [N. Nakicenovic, et al.] Cambridge University Press, Cambridge, UK, 598 pp. [NPR]
IPCC, 2000c: Methodological and Technological Issues in Technology Transfer. Special Report of the Intergovernmental Panel on Climate Change (IPCC) [B. Metz, et al.] Cambridge University Press, Cambridge, UK, 466 pp. [NPR]
IPCC, 2001a: Climate Change 2001: Mitigation. Intergovernmental Panel on Climate Change (IPCC) [B. Metz, et al. (eds)]. Cambridge University Press, Cambridge, UK. [NPR]
IPCC, 2001b: Climate Change 2001: Impacts, Adaptation, and Vulnerability. Intergovernmental Panel on Climate Change (IPCC) [J.J. McCarthy, et al. (eds.), Cambridge University Press, Cambridge, UK. [NPR]
IPCC, 2005a: Meeting Report: IPCC Expert Meeting on Industrial Technology Development, Transfer and Diffusion. Intergovernmental Panel on Climate Change (IPCC), 21-23 Sept. 2004, Tokyo, 35 pp. [NPR]
IPCC, 2005b: Carbon Capture and Storage. Special Report of the Intergovernmental Panel on Climate Change (IPCC), Cambridge University Press, Cambridge, UK. [NPR]
IPCC, 2006: 2006 Inventory Guidelines. Intergovernmental Panel on Climate Change (IPCC), Cambridge University Press, Cambridge, UK. [NPR]
IPCC/TEAP, 2005: Safeguarding the Ozone Layer and the Global Climate System: Issues Related to Hydrofluorocarbons and Perfluorocarbons. Special Report of the Intergovernmental Panel on Climate Change/Technical and Economic Assessment Panel, Cambridge University Press, Cambridge, UK. [NPR]
IPPC, 2001: Reference document on best available techniques in the cement and lime manufacturing industries. European Commission, Brussels, Belgium. [NPR]
ISO, 1996: ISO 14001, Environmental Management Systems - Specification with guidance for use. International Standards Organization ISO, Geneva, 14 pp. [NPR]
ISO, 2004: Business Plan ISO/TC 63 Glass Containers, Geneva: International Standard Organization, 9 pp. [NPR]
ITC, 2006: For all our tomorrows. Sustainability Report 2005, <www.itcportal.com/sustainability_report_2005/default.html>, accessed 31/05/07. [NPR]
Jacoby, H.D., 1998: The uses and misuses of technology development as a component of climate policy. MIT Joint Program on the Science and Politics of Global Change, Report No, 43., <http://web.mit.edu/globalchange/www/rpt43.html>, accessed 31/05/07. [NPR]
Jain, A.K., 2005: Cement: strong growth performance, survey of Indian industry 2005. The Hindu, Chennai, India. [NPR]
Jindal Steel and Power, Ltd., 2006: Building a new India, 26th Annual Report 2004-2005. New Delhi, India. [NPR]
Jochem, E. and W. Eichhammer, 1999: Voluntary agreements as an instrument to substitute regulating and economic instruments. Lessons from the German Voluntary Agreements on CO2 Reduction. In Voluntary Approaches in Environmental Policy, C. Carraro and F. Lévêque (eds.), Kluwer Academic Publisher, Dordrecht, Boston, London. [NPR, ARC]
Joint Research Centre, n.d.a: Mission of the Joint Research Centre. <www.jrc.cec.eu.int>, accessed 31/05/07. [NPR]
Joint Research Centre, n.d.b.: End-use energy efficiency activities of the European Commission Joint Research Centre. <http://re.jrc.ec.europa.eu/energyefficiency/index.htm>, accessed 01/06/07. [NPR]
Josa, A., A. Aguado, A. Heino, E. Byars, and A. Cardin, 2004: Comparative analysis of available life-cycle inventories of cement in the EU. Cement and Concrete Research, 34, pp. 1313-1320. Clean
Jupiter Oxygen Corp., 2006: Supply-side energy efficiency and fossil fuel switch. Presented at Carbon Expo, May 11, 2006, Cologne, Germany, <www.jupiteroxygen.com/spotlight/uploads/050206_efficiency.pdf>, accessed 31/05/07. [NPR]
Kaltner, F.J., G.F.P Zepeda, I.A. Campos, and A.O.F. Mondi, 2005: Liquid biofuels for transportation in Brazil. <www.fbds.org.br/IMG/pdf/doc-116.pdf>, accessed 31/05/07. [NPR]
Kannan, R. and W. Boy, 2003: Energy management practices in SME- Case study of bakery in Germany. Energy Conversion and Management, 44, pp. 945-959. Clean
Kerssemeeckers, M., 2002: The Dutch long term voluntary agreements on energy efficiency improvement in industry. Utrecht, the Netherlands: Ecofys. [NPR]
Kim, Y. and E. Worrell, 2002a: International comparison of CO2 emissions trends in the iron and steel industry. Energy Policy, 30, pp. 827-838. [SRC]
Kim, Y. and E. Worrell, 2002b: CO2 emission trends in the cement industry: An international comparison. Mitigation and Adaptation Strategies for Global Change, 7, pp. 115-33. [SRC]
Kirk-Othmer, 2005: Recycling, Glass. In : C. Philip Ross, Kirk-Othmer, Encyclopaedia of Chemical Technology, John Wiley & Sons; Article Online Posting Date: May 13, 2005. Clean
Kolk, A. and J. Pinkse, 2005: Business response to climate change: Identifying emergent strategies. California Management Review, 47(3), pp. 6-20. Clean
Kumar, S., G. Senanakaye, C. Visvanathan, and B. Basu, 2003: Desiccated coconut industry of Sri-Lanka’s opportunities for energy efficiency and environmental protection. Energy Conversion and Management, 44(13), pp. 2205-2215. Clean
Laurin, A., J. Nyboer, C. Strickland, N. Rivers. M. Bennett, M. Jaccard, R. Murphy, and B. Sandownik, 2004: Strategic options for combined heat and power in Canada. Office of Energy Efficiency, Natural Resources Canada, Ottawa, ON, Canada. [NPR]
Lemar, P.L., 2001: The potential impact of policies to promote combined heat and power in US industry. Energy Policy, 29, pp. 1243-1254. Clean
Linde, 2005: Stirring and degassing. Linde Mix 14, <www.us.lindegas.com/international/web/lg/us/likelgus30.nsf/DocByAlias/nav_industry_alum_stir>, accessed 31/05/07. [NPR]
Liskas, K., G. Mavrotas, M. Mandaraka, and D. Diakoulaki, 2000: Decomposition of industrial CO2 emissions: The case of the European Union. Energy Economics, 22, pp. 383-394. Clean
Luiten, E. and K. Blok, 2003a: The success of a simple network in developing an innovative energy-efficient technology. Energy, 28, pp. 361-391. [ARC]
Luiten, E. and K. Blok, 2003b: Stimulating R&D of industrial energy-efficient technology; the effect of government intervention on the development of strip casting technology. Energy Policy, 31, pp. 1339-1356. [ARC]
Luiten, E. and K. Blok, 2004: Stimulating R&D of industrial energy-efficient technology. Policy Lessons - Impulse Technology. Energy Policy, 32, pp. 1087-1108. [ARC]
Lung, R.B., A. McKane, and M. Olzewski, 2003: Industrial Motor system optimization projects in the US: An impact study. Proceedings of the 2003 American Council for an Energy-Efficient Economy Summer Study on Energy Efficiency in Industry. Washington, D.C., ACEEE. [NPR]
Margolick, M. and D. Russell, 2001: Corporate greenhouse gas reduction targets. Washington, D.C.: Pew Center for Global Climate Change. [NPR]
Martcheck, K., 2006: Modelling more sustainable aluminium: Case study. International Journal of LCA, 11, pp. 34-37. [MoS]
Martin, N., E. Worrell, M. Ruth, M., L. Price, R.N. Elliott, A. Shipley and J. Thorne, 2000: Emerging energy-efficient industrial technologies. Washington D.C., American Council for an Energy-efficient economy and Berkeley, CA, Lawrence Berkeley National Laboratory (LBNL-46990). [NPR, SRC]
Matsuhashi, R., M. Shigyo, Y. Yoshida, and H. Ishitani, 2000: Life cycle analysis of systems utilizing waste heat to reduce CO2 emissions. Proceedings of International Conference on Electrical Engineering, pp. 613-616. [NPR, SRC]
Matysek, A., M. Ford, G. Jakeman, A. Gurney, and B.S. Fisher, 2006: Technology: Its role in economic development and climate change. ABARE Research Report 06.6, Canberra, Australia. <www.abare.gov.au/publications_html/climate/climate_06/cc_technology.pdf>, accessed 31/05/07. [NPR, SRC]
McFarland, M., 2005: Statement of Mack McFarland, Ph.D., Global Environmental Manager, DuPont Fluoroproducts, E.I. DuPont de Nemours and Company, Inc. before the Committee on Science, US House of Representatives, June 8, 2005, <www.house.gov/science/hearings/full05/june8/dupont.pdf>, accessed 31/05/07. [NPR, ARC]
McKane, A., G. Zhou, R. Williams, S. Nadel, and V. Tutterow, 2003: The China Motor Systems Energy Conservation Program: Establishing the foundation for systems energy efficiency. Berkeley, CA:, Lawrence Berkeley National Laboratory (LBNL-52772). [NPR]
McKane, A., W. Perry, A. Li, T. Li, and R. Williams, 2005: Creating a standard framework for sustainable industrial energy efficiency. Presented at Energy Efficiency in Motor Driven Systems (EEMODS 2005) Conference, Heidelberg, Germany, 5-8 September 2005. [NPR]
Meier, A., E. Bonaldi, G.M. Cella, W. Lipinski, D. Wullemin, and R. Palumbo, 2004: Design and experimental investigation of a horizontal rotary reactor for the solar thermal production of lime. Energy, 29, pp. 811-821. [NPR]
Miller, M.M., 2003: Lime. In 2003 Minerals Yearbook, United States Geological Survey. Reston, VA. [NPR]
Miner, R. and A. Lucier, 2004: A value chain assessment of the climate change and energy issues affecting the global forest products industry. <http://www.wbcsd.ch/web/projects/forestry/ncasi.pdf>, accessed 01/06/07. [NPR, SRC]
Miner, R. and B. Upton, 2002: Methods for estimating greenhouse gas emissions from lime kilns at Kraft pulp mills. Energy, 27, pp. 729-738. [MoS, SRC]
Möllersten, K., J. Yan, and M. Westermark, 2003: Potential and cost effectiveness of CO2 reductions through energy measures in Swedish pulp and paper mills. Energy, 28, pp. 691-710. Clean
Moreira, J., 2006: Global biomass energy potential. Journal of Mitigation and Adaptation Strategies for Global Change, 11, pp. 313-333. Clean
Morimoto, K., H.X. Nguyen, M. Chihara, T. Honda and R. Yamamoto, 2006: Proposals for classification and an environmental impact evaluation method for eco-services: Case study of municipal waste treatment in cement production. Journal of Life Cycle Assessment, Japan, 2, pp. 347-354. Clean
Mozorov, A. and G. Nikiforov, 2002: Energy development concept of OAO Magnitogorsk Iron and Steel Works. Steel Journal, 1, Moscow, Russia (original in Russian). Clean
Muller, M.R. and T.J. Barnish, 1998: Evaluation of the former EADC-Program. In Industrial Energy Efficiency Policies: Understanding Success and Failure, Martin et al.. (eds.), Proceedings of a workshop organized by the International Network for Energy Demand Analysis in the Industrial Sector. Utrecht, The Netherlands, June 11-12, 1998, (LBNL-42368). [NPR]
Munasinghe, M., 2002: Sustainomics transdisciplinary framework for making development more sustainable - application to energy issues. International Journal of Sustainable Development, 5, pp.125-82. [ARC]
Munn, A., 2004: Emission reduction at Engen refinery in South Durban. Paper presented at the 8th World Congress on Environmental Health, Durban, South Africa, 22-27 February 2004. [NPR]
Naiwei, S., 2004: The Chinese brick and tile industry - Steady development in 2004. Ziegelindustrie International, Pp. 46-49, 4/2004. [NPR]
Natural Resources Canada, n.d: Industrial Energy Innovators. <http://oee.nrcan.gc.ca/industrial/opportunities/innovator/index.cfm?attr=24>, accessed 31/05/07. [NPR]
Naughton, B. 2005: The new common economic program: China’s 11th five year plan and what it means. China Leadership Monitor No.16, <www.hoover.org/publications/clm/issues/2898936.html>, accessed 31/05/07. [NPR]
Navarro, P., G. Gregoric, O. Cobo, and A. Calandra, 2003: A new anode effect quenching procedure. Light Metals, 2003, pp. 479-486. Clean
NEDO, n.d.: Industrial technology research and development activities. New Energy and Industrial Technology Development Organization, <www.nedo.go.jp/english/activities/index.html>, accessed 31/05/07. [NPR]
NEEC, 2005: One-year accelerated depreciation allowance for energy efficient equipment and technology. National Energy Efficiency Committee (Singapore), < http://www.nccc.gov.sg/incentive/home.shtm >, accessed 01/06/07. [NPR]
NERA, 2004: Review of the first and second years of the UK emissions trading scheme. Prepared for UK Department for Environment, Food and Rural Affairs, D. Radov and P. Klevnäs, NERA, <http://www.defra.gov.uk/environment/climatechange/trading/uk/pdf/nera-commissionreport.pdf>, accessed 31/05/07. [NPR]
Nicholson, C., 2004: Protecting the environment - A business perspective. 4th Specialty Conference on Environmental Progress, Bahrain, <www.bp.com/genericarticle.do?categoryId=98&contentId=2017173>, accessed 31/05/07. [NPR]
Nippon Keidanren, 2004: Results of the fiscal 2004 follow-up to the Keidanren Voluntary Action Plan on the Environment - Section on Global Warming Measures, <http://www.keidanren.or.jp/english/policy/2004/091/index.html>, accessed 31/05/07. [NPR]
NRC, 2001: Energy Research at DOE: Was it worth it? Energy efficiency and fossil energy research 1978 to 100. National Research Council, Washington D.C, National Academy Press, < http://www.nap.edu/openbook/0309074487/html/>, accessed 01/06/07. [NPR]
NREL, 2006: Advancing Clean Energy Use in Mexico. National (US) Renewable Energy Laboratory, <www.nrel.gov/docs/fy05osti/38628.pdf>, accessed 31/05/07. [NPR]
Nyboer, J. and J.J. Tu, 2003: A review of energy consumption & related data: Canadian cement manufacturing industry, 1990 to 2001: Canadian Industry Energy End-use Data and Analysis centre, Simon Fraser University, Vancouver, BC. [NPR]
Ochs, T., P. Turner, D. Oryshchyn, B. Patrick, A. Gross, C. Summers, W. Simmons, and M. Schoenfeld, 2005: Proof of concept for integrating oxy-fuel combustion and the removal of all pollutants from a coal-fired flame. Presented at the 30th International Technical Conference on Coal Utilization and Fuel Systems, Clearwater, FL, USA. [NPR]
OECD, 2002: Voluntary approaches for environmental policy: Effectiveness, efficiency and use in policy mixes. Organization for Economic Cooperation and Development (OECD), Paris. [NPR]
Okazaki, T., M. Nakamura, and K. Kotani, 2004: Voluntary initiatives of Japan’s steel industry against global warming. Paper presented at IPCC Industrial Expert Meeting (ITDT) in Tokyo. [NPR, SRC]
Okuwaki, A., 2004: Feedstock recycling of plastics in Japan. Polymer Degradation and Stabilization, 85, pp. 981-988. Clean
Olivier, J.G.J. and J.A.H.W. Peters, 2005: CO2 from non-energy use of fuels: A global, regional and national perspective based on the IPCC Tier 1 approach. Resources, Conservation and Recycling, 45(3), pp. 210-225. [ARC]
PCA, 2002: Common elements among advanced greenhouse gas management programs: A discussion paper. New York, Partnership for Climate Action, <www.environmentaldefense.org/documents/1885_PCAbooklet.pdf>, accessed 31/05/07. [NPR]
Pew Center on Global Climate Change, 2005: Business Environmental Leadership Council (BELC). <www.pewclimate.org/companies_leading_the_way_belc>, accessed 31/05/07. [NPR]
Phadke, A., J. Sathaye, and S. Padmanabhan, 2005: Economic benefits of reducing Maharashtra’s electricity shortage through end-use efficiency improvement. LBNL-57053. Lawrence Berkeley National Laboratory, Berkeley, CA. [NPR, ARC]
Phylipsen, D., K. Blok, E. Worrell, and J. De Beer, 2002: Benchmarking the energy efficiency of Dutch industry: an assessment of the expected effect on energy consumption and CO2 emissions. Energy Policy, 30, pp. 663-679. [SRC]
Phylipsen, G.J.M. and K. Blok, 2002: The effectiveness of policies to reduce industrial greenhouse gas emissions. Paper for the AIXG Workshop on Policies to Reduce Greenhouse Gas Emissions in Industry - Successful Approaches and Lessons Learned, 2-3 December 2002, Berlin. [NPR, ARC]
Pickens, J.G., S.T.S. Yuen, and H. Hennings, 2002: Waste management options to reduce greenhouse gas emissions from paper in Australia. Atmospheric Environment, 36, pp. 741-752. Clean
Pilkington, 2005: Flat glass industry - Summary, Pilkington, plc. <www.pilkington.com/about+pilkington/flat+glass+industry/default.htm>, accessed 31/05/07. [NPR]
Price, L., 2005: Voluntary agreements for energy efficiency or ghg emissions reduction in industry: An assessment of programs around the world. Proceedings of the 2005 American Council for an Energy Efficient Economy Summer Study on Energy Efficiency in Industry, West Point, NY, USA, July, 2005. [NPR, SRC]
Price, L., J. Sinton, E. Worrell, D. Phylipsen, H. Xiulian, and L. Ji, 2002: Energy use and carbon dioxide emissions from steel production in China. Energy, 5, pp. 429-446. [SRC]
Price, L., S. de la Rue du Can, J. Sinton, E. Worrell, N. Zhou, J. Sathaye, and M. Levine, 2006: Sectoral trends in global energy use and greenhouse gas emissions. LBNL-56144, Lawrence Berkeley National Laboratory, Berkeley, CA. [NPR, SRC]
PSI, 2004: Energy efficiency and CO2 emissions benchmarking of IFA Ammonia Plants (2002-2003 Operating Period). Plant Surveys International, Inc General Edition. Commissioned by the International Fertilizer Industry Association (IFA). [NPR]
Pye, M. and A. McKane, 2000: Making a stronger case for industrial energy efficiency by quantifying non-energy benefits. Resources, Conservation and Recycling, 28, pp. 171-183. Clean
Rafiqul, I., C. Weber, B. Lehmann, and A. Voss, 2005: Energy efficiency improvements in ammonia plants: Perspectives and uncertainties. Energy, 30, pp. 2487-2504. Clean
Raina, S.J., 2002: Energy efficiency improvements in Indian Cement Industry. <www.energymanagertraining.com/cement/pdf/IIPEC_NCCBM-2.pdf>, accessed 31/05/07. [NPR]
Rainey, D. and P. Patilis, 2000: Developing an energy conservation program at United Technologies Corporation. Corporate Environmental Strategy, 7, pp. 333-342. Clean
Ramesohl, S. and K. Kristof, 2001: The declaration of German industry on global warming potential - A dynamic analysis of current performance and future prospects for development. Journal of Cleaner Production, 9, pp. 437-446. [MoS]
Ren, T., M. Patel, and K. Blok, 2005: Natural gas and steam cracking: Energy use and production cost. Published in the conference proceedings of the American Institute of Chemical Engineers Spring National Meeting, Atlanta, GA, USA, 10-14 April, 2005. [NPR, ARC]
Ren, T., M. Patel, and K. Blok, 2006: Olefins from conventional and heavy feedstocks: Energy use in steam cracking and alternative processes. Accepted for publication in Energy. Available online at <www.ScienceDirect.com>, accessed 31/05/07. [NPR, ARC]
Rietbergen, M.J., J.C.M. Farla, and K. Blok, 2002: Do agreements enhance energy efficiency improvement? Analyzing the Actual Outcome of the Long-Term Agreements on Industrial Energy Efficiency Improvement in The Netherlands. Journal of Cleaner Production, 10, pp. 153-163. [ARC]
Rock, M.T. and D.T. Angel, 2005: Industrial transformation in the developing world. Chapter 6: Win-Win environmental intensity or technique effects and technological learning: Evidence from Siam City Cement. New York, Oxford University Press, pp. 127-149. [NPR]
Römpp, 1995: Römpp Chemie Lexikon. J. Falbe and M. Regnitz (eds.), Stuttgart, Germany. [NPR]
Rosenberg, N., 1999: Inside the black box: Technology and economics. Cambridge University Press. [NPR]
RWI, 2004: Die Klimaschutzerklärung der Deutschen Wirtschaft, Rheinisch-Westfälisches Institut für Wirtschaftsforschung Essen, Germany: RWI <http://www.rwi-essen.de/pls/portal30/docs/FOLDER/PROJEKTE/CO2MONITORING/MONITORING_DATEIEN/MONITORINGBERICHT_2000-2002.PDF>, accessed 01/06/07. [NPR]
SAI, 2001: Social Accountability 8000. Social Accountability International, New York, SAI, 8 pp. [NPR]
Sathaye, J., J. Roy, R. Khaddaria, and S. Das, 2005: Reducing electricity deficit through energy efficiency in India: An evaluation of macroeconomic benefits. LBLN-59092. Berkeley, CA, Lawrence Berkeley National Laboratory. [NPR, SRC]
Schipper, L., F. Unander, and C. Marie-Lilliu, 2000: The IEA Energy Indicators: Analysing emissions on the road to Kyoto. Paris, France, IEA. [NPR]
Schleich, J., 2004: Do energy audits help reduce barriers to energy efficiency? An empirical analysis for Germany. International Journal of Energy Technology and Policy, 2, pp. 226-239. Clean
Shashank, J., 2004: Energy conservation in the industrial sector: A special report on energy conservation day. New Delhi, Economic Times. [NPR]
Sheinbaum, C. and L. Ozawa, 1998: Energy use and CO2 emissions for Mexico’s cement industry. Energy, 23, pp. 725-32. Clean
Shell, 2005: The Shell Sustainability Report, 2005. < www.shell.com/shellreport/>, accessed 31/05/07. [NPR]
Sims, R.E.H, 2002: Waste co-gen plant-rock point sugar. Renewable Energy World, (March-April, 2002), pp. 105-109. [NPR, ARC]
Sinton, J.E. and D.G. Fridley, 2000: What goes up: Recent trends in China’s Energy Consumption. Energy Policy, 28, pp. 671-687. Clean
Silayan, A., 2005: Equitable distribution of CDM projects among developing countries. HWWA-Report, Hamburg, Germany, Hamburg Institute of International Economics. [NPR]
Sjardin, M., 2003: CO2 emission factors for non-energy use in the non-ferrous metal, ferroalloys and inorganics industry. Copernicus Institute, Utrecht, pp. 63. [NPR]
Smith, A, K. Brown, S. Olilvie, K. Rushton, and J. Bates, 2001: Waste management options and climate change. AEA Technology for the European Commission, DG ENV. <http://europa.eu.int/comm/environment/waste/studies/pdf/climate_change.pdf>, accessed 31/05/07. [NPR]
Sobhanbabu, P.R.K., 2003: Sugar cogeneration for power challenges and opportunities. Cane-Cogen, India, Vol. 16. a quarterly newsletter in sugar and cogeneration. [NPR]
STAPPA/ALAPCO, 1999: Reducing greenhouse gases and air pollution, A menu of harmonized options. Final Report, State and Territorial Air Pollution Program Administrators/Association of Local Air Pollution Control Officials, Washington, pp. 119-163. [NPR]
Starzer, O., 2000: Negotiated agreements in industry: Successful ways of implementation. Vienna, EVA, Austrian Energy Agency. [NPR]
Subak, S., J.P. Palutikof, M.D. Agnew, S.J. Watson, C.G. Bentham, M.G.R. Cannell, M. Hulme, S. McNally, J.E. Thornes, D. Waughray, and J.C. Woods, 2000: The impact of the anomalous weather of 1995 on the UK Economy. Climatic Change, 44(1-2), pp. 1-26. [JoC]
Sukalac, K.E., 2005: Technology transfer to reduce climate change impacts from the fertilizer industry. Presented at UNFCCC COP-11 Side Event “Knowledge Transfer to Reduce Greenhouse Gas Emission: Lessons from the Fertilizer Industry. Montreal, Canada, 6 December 2005. [NPR]
Sun, C. and M. Williamson, 1999: Industrial energy use benchmarking. In Proceedings 1999 ACEEE Summer Study on Energy Efficiency in Industry, American Council for an Energy-Efficient Economy: Washington, D.C. [NPR]
Sutton, P., 1998: Sustainability: Which industries first? Green Innovations, Inc. <www.green-innovations.asn.au/wot-ind.htm>, accessed 31/05/07. [NPR]
Swamidass, P.M., 2003: Modelling the adoption rates of manufacturing technology innovations by small US manufacturers: a longitudinal investigation. Research Policy, 32(3), pp. 351-366. [MoS]
Swaminathan, B. and K.E. Sukalac, 2004: Technology transfer and mitigation of climate change: The fertilizer industry perspective. Presented at the IPCC Expert Meeting on Industrial Technology Development, Transfer and Diffusion, Tokyo, Japan, 21-23 Sept. 2004. [NPR]
Szklo, A.M., J.B. Soares, and M.T. Tolmasquim, 2004: Economic potential of natural gas-fired co-generation-analysis of Brazil’s chemical industry. Energy Policy, 32, pp. 1415-1428. Clean
Tanaka, K., K. Sasaki, and H. Kudoh, 2005: Estimation of CO2 reduction potentials through energy efficient technologies. Journal of Institute for Energy Economics in Japan, 31, pp. 61-81. [MoS, SRC]
Tanaka, K., R. Matsuhashi, N. Masahiro, and H. Kudo, 2006: CO2 reduction potential by energy efficient technology in energy intensive industry. <http://eneken.ieej.or.jp/en/data/pdf/324.pdf>, accessed 31/05/07. [NPR, SRC]
Tata Steel, Ltd, 2005: 3rd Social Audit. Mumbai, India. [NPR]
The Aluminum Association, 2003: Aluminum Industry Technology Roadmap. Washington, DC, 60 pp. [NPR]
The Sigma Project, 2003a: The Sigma guidelines: Putting sustainable development into practice - A guide for organizations. <www.projectsigma.co.uk/Guidelines/SigmaGuidelines.pdf>, accessed 31/05/07. [NPR]
The Sigma Project, 2003b: The Sigma Guidelines - Toolkit - The Sigma Guide to the AA1000 Assurance Standard. [NPR]
Toman, M., 2003: ‘Greening’ economic development activities for greenhouse gas mitigation. Resources for the Future, Issue Brief, RFF IB 03-02, <www.rff.org>, accessed 31/05/07. [NPR, MoS]
UBA, 2004: Fluorinated greenhouse gases in products and processes: Technical climate protection measures. Umweltbundesamt, <www.umweltbundesamt.de/produkte-e/fckw/index.htm>, accessed 31/05/07. Berlin, Germany, 249 pp. [NPR]
UK DEFRA, 2005: Securing the future: UK Government Sustainable Development Strategy. United Kingdom Department of the Environment, Food and Rural Affairs, http://www.sustainable-development.gov.uk/publications/uk-strategy/index.htm, accessed 31/05/07. [NPR, MoS]
UK DEFRA, 2006: Climate change agreements - The climate change levy; United Kingdom Department of the Environment, Food and Rural Affairs. <www.defra.gov.uk/environment/ccl/intro.htm>, accessed 31/05/07. [NPR]
UNDP, 2002: Oil Palm R&D: Malaysia. UN Development Programme. [NPR]
UNESCAP, 2000: Promotion of energy efficiency in industry and financing of investments. United Nations Economic and Social Commission for Asia and the Pacific, <www.unescap.org/esd/energy/publications/finance/index.html>, accessed 31/05/07. [NPR]
UNFCCC, 1995: Report of COP-1, Actions Taken. UN Framework Convention on Climate Change, <http://unfccc.int/resource/docs/cop1/07a01.pdf>, accessed 31/05/07. [NPR]
UNFCCC, 1997: Kyoto Protocol. UN Framework Convention on Climate Change, <http://unfccc.int/resource/docs/convkp/kpeng.pdf>, accessed 31/05/07. [NPR]
UNFCCC, 2001: The Marrakech Accords. UN Framework Convention on Climate Change, <http://unfccc.int/resource/docs/cop7/13a02.pdf>, accessed 31/05/07. [NPR]
UNFCCC, 2002: Good practices. Policies and measures among parties included in Annex I to the Convention in their Third National Communications - Report by the Secretariat. UN Framework Convention on Climate Change. [NPR]
UNFCCC, 2004: Results of the survey on effectiveness of the use of the UNFCCC technology information. UN Framework Convention on Climate Change clearinghouse (TT:CLEAR). [NPR]
UNFCCC, CDM, n.d.: UN Framework Convention on Climate Change, Clean Development Mechanism. <http://cdm.unfccc.int/Statistics>, accessed 31/05/07. [NPR]
UNIDO, 2001: Africa industry and climate change project proceedings. UN Industrial Development Organization, UNIDO publication, Vienna. [NPR]
UNIDO, 2002: Developing national capacity to implement Clean Development Mechanism projects in a selected number of countries in Africa-A case study for Zambia and Zimbabwe UNIDO publication. UN Industrial Development Organization. [NPR]
UNIDO and IFDC, 1998: Fertilizer manual. UN Industrial Development Organization and International Fertilizer Development Center, Dordrecht, The Netherlands, Kluwer Academic Press, 515 pp. [NPR]
US DOE, 2002: Steam system opportunity assessment for the pulp and paper, chemical manufacturing, and petroleum refining industries - Main Report. US Department of Energy, Washington, D.C. [NPR]
US DOE, 2003: Energy matters, the best practices quarterly newsletter for the US Department of Energy’s Industrial Technologies Program. US Department of Energy, Summer 2003 issue, pp. 6-7. [NPR]
US DOE, 2004: 20 ways to save energy now. US Department of Energy, <www.eere.energy.gov/industry/bestpractices/energymatters/pdfs/em_volume29.pdf>, accessed 31/05/07. [NPR]
US DOE, 2006: Industrial Technologies Program: Summary of Program results for CY 2004. US Department of Energy, <www1.eere.energy.gov/industry/about/pdfs/impacts2006.pdf>, accessed 31/05/07. [NPR]
US DOE, n.d.-a: Industrial Technologies Program. US Department of Energy, <http://www1.eere.energy.gov/industry>, accessed 31/05/07. [NPR]
US DOE, n.d.-b.: Solicitations. US Department of Energy, <www1.eere.energy.gov/industry/financial/solicitations.html>, accessed 31/05/07. [NPR]
US Dept. of State, 2004: Bilateral and regional partnerships. <http://www.state.gov/g/oes/ris/fs/2004/39438.htm>, accessed 31/05/07, <www.state.gov/g/oes/rls/fs/2004/39438.htm>, accessed 31/05/07. [NPR]
US EPA, 1999: International efforts to reduce perfluorocarbon (PFC) emissions from primary aluminium production. United States Environmental Protection Agency Washington, DC. [NPR]
US EPA, 2001: Non-CO2 greenhouse gas emissions from developed countries: 1990-2010. United States Environmental Protection Agency, Washington, DC. [NPR]
US EPA, 2002: Solid waste management and greenhouse gases: A life-cycle assessment of emissions and sinks. United States Environmental Protection Agency, EPA # 530-R-02-006. [NPR]
US EPA, 2003: International analysis of methane and nitrous oxide abatement opportunities: report to energy modelling forum. Working Group 21, Appendix C. United States Environmental Protection Agency, <www.epa.gov/nonco2/econ-inv/pdfs/methodologych4.pdf >, accessed 31/05/07. [NPR, MoS]
US EPA, 2005: Landfill methane outreach program. United States Environmental Protection Agency, <www.epa.gov/lmop>, accessed 31/05/07. [NPR]
US EPA, 2006a: Global mitigation of non-CO2 greenhouse gas emissions (EPA Report 430-R-06-005). United States Environmental Protection Agency Office of Air and Radiation. Washington, D.C., USA. [NPR]
US EPA, 2006b: Global anthropogenic emissions of non-CO2 greenhouse gas 1990-2020 (EPA Report 430-R-06-003). United States Environmental Protection Agency Office of Air and Radiation. Washington, D.C., USA. [NPR]
US EPA, n.d.: Energy STAR for Industry. United States Environmental Protection Agency, <www.eere.energystar.gov/index.cfm?c=industry.bus_industry>, accessed 31/05/07. [NPR]
USGS, 2005: Minerals Yearbook 2004. US Geological Survey Reston, VA, USA. <http://minerals.usgs.gov/minerals/pubs/myb.html>, accessed 31/05/07. [NPR]
USGS, 2006: Minerals commodity summary: cement. Reston, VA, USA. US Geological Survey, <http://minerals.usgs.gov/minerals/pubs/commodity/cement/cemenmcs06.pdf>, accessed 31/05/07. [NPR]
UTC, 2003: United technologies steps up global conservation efforts. United Technologies Corporation <http://www.utc.com/press/releases/2003-04-22.htm>, accessed 31/05/07. [NPR]
Vaccari, G., E. Tamburini, G. Sualdino, K. Urbaniec. and J. Klemes. 2005: Overview of the environmental problems in beet sugar processing: Possible solutions. Journal of Cleaner Production, 13, pp. 499-507. Clean
Verduijn, W.D. and J.J. de Wit, 2001: Energy conservation: Key to survival for fertiliser producers? Proceedings No. 479, International Fertiliser Society, York, UK, 24 pp. [NPR]
Wartmann, S., and J. Harnisch, 2005: Reduction of SF6 emissions from high and medium voltage equipment in Europe. For the Coordinating Committee for the Associations of Manufacturers of Industrial Electrical Switchgear and Control Gear in the EU (CAPIEL) <www.capiel-elec-tric.com/publicats/Ecofys%20SF6%20Study%20Final%20Report%2022%20Nov%202005.pdf>, accessed 31/05/07. [NPR, SRC]
WBCSD, 2002: The cement sustainability initiative: Our agenda for action. World Business Council for Sustainable Development, <www.wbcsd.org/web/publications/cement-action-plan.pdf>, accessed 31/05/07. [NPR]
WBCSD, 2005: The Cement Sustainability Initiative: Progress Report. World Business Council for Sustainable Development, <www.wbcsdcement.org/pdf/csi_progress_report_2005.pdf>, accessed 31/05/07. [NPR]
WEC, 2001: Energy efficiency policies and indicators policies, World Energy Council London. [NPR]
WEC, 2004: Energy efficiency: A worldwide review - Indicators, policies, evaluation. World Energy Council, London. [NPR]
WEF, 2005: Global Greenhouse Gas Register. World Economic Forum. <www.weforum.org/en/iniatives/ghg/GreenhouseGasRegister/index.htm>, accessed 31/05/07. [NPR]
WWF, n.d.: Climate change featured projects: Climate savers. World Wildlife Fund, <www.worldwildlife.org/climate/projects/climateSavers.cfm>, accessed 31/05/07. [NPR]
Worrell, E., D. Smit, K. Phylipsen, K. Blok, F. van der Vleuten, and J. Jansen, 1995: International comparison of energy efficiency improvement in the cement industry. Proceedings ACEEE 1995 Summer Study on Energy Efficiency in Industry, Volume II, pp. 123-134. [NPR, SRC]
Worrell, E., D. Phylipsen, D. Einstein, and N. Martin, 2000a: Energy use and energy intensity of the US chemical industry. Berkeley, CA, Lawrence Berkeley National Laboratory (LBNL-44314). [NPR, SRC]
Worrell, E., N. Martin, and L.K. Price, 2000b: Potentials for energy efficiency improvement in the us cement industry. Energy, 25, pp. 1189-1214. [SRC]
Worrell, E., N. Martin, and L.K. Price, 2001a: Energy efficiency and carbon dioxide emissions reduction opportunities in the US iron and steel sector. Energy, 26, pp. 513-36. [SRC]
Worrell, E., L.K. Price, N. Martin, C. Hendriks, and L. Ozawa Meida, 2001b: Carbon dioxide emissions from the global cement industry. Annual Review of Energy and Environment, 26, pp. 303-29. [SRC]
Worrell, E., N. Martin, L. Price, M. Ruth, N. Elliott, A. Shipley, and J. Thorne, 2002: Emerging energy efficient technologies for industry. Energy Engineering Journal, 99, pp. 36-55. [SRC]
Worrell, E., J.A. Laitner, M., Ruth, and H. Finman, 2003: Productivity Benefits of Industrial Energy Efficiency Measures. Energy, 28, pp. 1081-1098. [SRC]
Worrell, E. and C. Galitsky, 2004: Energy efficiency improvement opportunities for cement making - An ENERGY STAR Guide for Energy and Plant Managers. Berkeley, CA: Lawrence Berkeley National Laboratory, (LBNL 54036). [NPR, SRC]
Worrell, E. and G. Biermans, 2005: Move over! Stock Turnover, Retrofit and Industrial Energy Efficiency. Energy Policy, 33, pp. 949-962. [SRC]
Worrell, E. and C. Galitsky, 2005: Energy efficiency improvement opportunities for petroleum refineries - An ENERGY STAR Guide for Energy and Plant Managers. Berkeley, CA: Lawrence Berkeley National Laboratory (LBNL 56183). [NPR, SRC]
WRI/WBCSD, 2004: The Greenhouse Gas Protocol: A corporate accounting and reporting standard (revised edition). World Resources Institute/World Business Council for Sustainable Development, 112 pp. [NPR]
WRI/WBCSD, 2005: GHG Protocol initiative/GHG calculation tools. World Resources Institute/World Business Council for Sustainable Development, <www.ghgprotocol.org>, accessed 31/05/07. [NPR]
Xenergy, Inc., 1998: Evaluation of the US Department of Energy Motor Challenge Program. <www.eere.energy.gov/industry/bestpractices/pdfs/mceval1_2.pdf>, accessed 31/05/07. [NPR]
Yamba, F. D. and E.M. Matsika, 2003: Assessment of the potential of the state of the art biomass technologies in contributing to a sustainable SADC removal mitigation energy scenario. Energy technologies for post Kyoto targets in the medium term. Proceedings of RISO International Energy Conference (19-23 May 2003). [NPR, SRC]
Yamba, F.D. and E.M. Matsika, 2004: Factors and barriers influences the transfer of technologies with particular reference to Southern Africa. Presented at the IPCC Expert Meeting on Industrial Technology Development, Transfer and Diffusion, Tokyo (21-23 September 2004). [NPR, SRC]
Yeoh, B.G., 2004: A technical and economic analysis of heat and power generation of palm oil mill effluent (POME) <www.cogen3.net/doc/countryinfo/malaysia/TechnicalEconomicAnalysisCHPPalmEffluent_BG.pdf >, accessed 31/05/07. [NPR]
Zhang, Z. X., 2003: Why did energy intensity fall in China’s industrial sector in the 1990s? The relative importance of structural change and intensity change. East-West Center Working Papers. Environmental Change, Vulnerability and Governance Series. [NPR]
Zhou, D., Y. Dai, C. Yi, Y. Guo, and Y. Zhu, 2003: China’s Sustainable Energy Scenarios in 2020. China Environmental Science Press, Beijing. [NPR, ARC]
Ziebek, A. and W. Stanek, 2001: Forecasting of the energy effects of injecting plastic wastes into the blast furnace in comparison with other auxiliary fuels. Energy, 26, pp. 1159-1173. [MoS]
| Tag | Explanation | Occurrences | Percentages |
|---|---|---|---|
| PoC |
Person of Concern Key individual involved in CRU emails as defined in this spreadsheet. |
1 | 0.2% |
| JoC |
Journal of Concern A Journal which has published articles by one or more PoCs (Person of Concern) |
4 | 1% |
| MoS |
Model or Simulation Reference appears to be a model or simulation, not observation or experiment. |
21 | 5% |
| NPR |
Non Peer Reviewed Reference has no Journal or no Volume or no Pages or it has Editors. |
274 | 77% |
| SRC |
Self Reference Concern Author of a chapter containing references to own work. |
43 | 12% |
| ARC |
Paper authored or co-authored by person who is also in list of Authors of another chapter. |
19 | 5% |
| Clean |
The reference was probably peer reviewed. |
44 | 12% |
NOTE: In the Summary of Reviewer Comments,
Accepted includes only those responses that were unambiguously Accepted.
There are other categories of responses that we have not yet quantified
due to inconsistencies of usage.
| Reviewer | Total Comments | Accepted | IPCC Roles | Papers |
|---|---|---|---|---|
| Government of U.S. Department of State | 104 | 52 | 0 | 0 |
| Government of Australia | 38 | 22 | 0 | 1 |
| Olivier, JGJ | 33 | 14 | 1 | 7 |
| Kessels, John | 30 | 24 | 0 | 0 |
| Government of European Community / European Commission | 24 | 10 | 0 | 0 |
| Gielen, D | 23 | 8 | 1 | 3 |
| Braathen, Nils-Axel | 21 | 6 | 0 | 0 |
| Marks, Jerry | 17 | 5 | 0 | 0 |
| Government of China Meteorological Administration | 12 | 8 | 0 | 0 |
| Nyboer, J | 12 | 3 | 0 | 5 |
| Debruxelles, Jean-Pierre | 11 | 1 | 0 | 0 |
| Muirheid, Ben | 10 | 2 | 0 | 0 |
| Ploger, Friedrich | 10 | 1 | 0 | 0 |
| Campbell, Nick | 9 | 3 | 0 | 0 |
| Gardiner, A | 9 | 2 | 0 | 2 |
| Novotny, Vladimir | 8 | 1 | 0 | 0 |
| Wang, Yanjia | 8 | 5 | 0 | 0 |
| Bonduelle, Antoine | 6 | 3 | 0 | 0 |
| Nziramasanga, Norbert | 6 | 2 | 0 | 0 |
| Aguayo, F | 6 | 2 | 0 | 1 |
| Government of India | 5 | 1 | 0 | 1 |
| Mizuno, Koichi | 5 | 1 | 0 | 0 |
| Lorea, Claude | 5 | 3 | 0 | 0 |
| Government of Belgium | 5 | 4 | 0 | 0 |
| McCulloch, Archie | 5 | 1 | 0 | 0 |
| Juhler-Kristoffersen, Helle | 4 | 0 | 0 | 0 |
| IPCC | 4 | 2 | 0 | 50 |
| Government of Japan | 4 | 2 | 0 | 2 |
| Chase, Robert | 4 | 1 | 0 | 0 |
| Government of Sweden | 3 | 0 | 0 | 0 |
| Taylor, MAP | 3 | 1 | 0 | 3 |
| Okayama, Takayuki | 3 | 1 | 0 | 0 |
| Miner, R | 3 | 1 | 1 | 2 |
| Mollersten, K | 3 | 0 | 0 | 2 |
| Akimoto, K | 2 | 2 | 0 | 3 |
| Klages-Buechner, Sabine | 2 | 1 | 0 | 0 |
| Poot, Brigitte | 2 | 1 | 0 | 0 |
| Fenhann, J | 2 | 2 | 1 | 3 |
| Rothermel, Joerg | 2 | 0 | 0 | 0 |
| de T'Serclaes, Philippine | 2 | 0 | 0 | 0 |
| Onuma, E | 2 | 0 | 0 | 1 |
| Macey, Kirsten | 2 | 0 | 0 | 0 |
| Berntsson, T | 2 | 0 | 0 | 1 |
| Izumi, Y | 2 | 0 | 0 | 1 |
| Caneill, Jean-Yves | 1 | 0 | 0 | 0 |
| Park, Jacob | 1 | 0 | 0 | 0 |
| Beerkens, Ruud | 1 | 1 | 0 | 0 |
| Jefferson, M | 1 | 0 | 0 | 2 |
| Government of Finland | 1 | 0 | 0 | 0 |
| Yanagisawa, Y | 1 | 0 | 0 | 1 |
| Jones, RJA | 1 | 0 | 0 | 2 |
| Koch, Lorenz | 1 | 1 | 0 | 0 |
| Maeda, A | 1 | 0 | 0 | 1 |
| Masui, T | 1 | 0 | 0 | 7 |
| Van Houte, Frederic | 1 | 0 | 0 | 0 |
| Hein, Joachim | 1 | 0 | 0 | 0 |
| Pitz-Paal, Robert | 1 | 1 | 0 | 0 |
| Oren, Kjell | 1 | 0 | 0 | 0 |
| Bogner, J | 1 | 0 | 2 | 11 |
| Botschek, Peter | 1 | 1 | 0 | 0 |
| Totals | 489 | 202 | 6 | 112 |
| Reviewer Type | Total Comments | Accepted | % Accepted |
|---|---|---|---|
| Govt | 149 | 74 | 49% |
| IPCC | 159 | 62 | 38% |
| Indep | 181 | 66 | 36% |
| Total | 489 | 202 | 41% |
Main AR4 Index | Working Group WG3 Index | Table of Contents | Authors | Executive Summary | Annotated Text | References | Reviewer Comments